Learning objectives
To illustrate 10 most frequently made mistakes in radiologic tumor response monitoring using RECIST 1.1 criteria.
To demonstrate possible negative consequences caused by these mistakes and to provoke alertness to avoid them.
Background
Over the past three decades there has been a divergence of imaging-based tumor-specific response criteria with the purpose to achieve objective assessment of treatment response in oncologic clinical trials.
In 2009 a revised RECIST 1.1 (Response Evaluation Criteria in Solid Tumors) was developed to address the pitfalls and limitations of previously applied response systems such as WHO and the initial version of RECIST [1].
The major changes included the number of lesions to be assessed,
which has been reduced from a maximum of 10 to...
Findings and procedure details
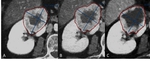
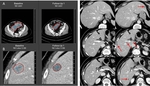
The most frequent mistakes in RECIST 1.1 application are related to inaccurate measurements,
inappropriate lesion definition or inadequate imaging techniques stated below:
1.
Assignment of non-qualifying target lesions (number,
size,
pseudolesions)
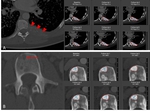
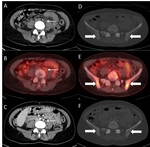
Despite a clear definition of target lesion number and size to be measured in RECIST 1.1,
assignment of non-qualifying lesions may occur systematically,
such as incorrect inclusion of too small lesions or non-metastatic lesions (Fig.
1A/B).
Definition of non-malignant masses as target lesions,
e.g.
hemangiomas and benign cystic lesions at baseline measurementsresults in incorrect...
Conclusion
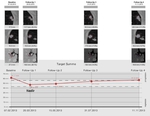
Radiologists should be aware that even minor mistakes at RECIST 1.1 application can dramatically influence treatment decision.
Proper application of RECIST 1.1 criteria with thorough knowledge of possible pitfalls and limitations can lower the negative implications for patient care and increase the efficiency of imaging based tumor response evaluation using RECIST 1.1.
Personal information
Maka Kekelidze,
MD,
PhD
Clinic of Radiology and Nuclear Medicine
University Hospital of Basel,
Switzerland
[email protected]
Pitro Lodise,
MD
Department of Radiological,
Oncological and Anatomopathological Sciences
"Sapienza" University of Rome,
Italy
[email protected]
Magdalini Tozakidou,
MD
Clinic of Radiology and Nuclear Medicine
University Hospital of Basel,
Switzerland
[email protected]
Mathias Seitel,
MD
Mint Medical GmbH,
Heidelberg,
Germany
[email protected]
Georg Bongartz,
MD,
PhD
Clinic of Radiology and Nuclear Medicine
University Hospital of Basel,
Switzerland
[email protected]
References
1.Eisenhauer,
E.A.,
et al.,
New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer,
2009.
45(2): p.
228-47.
2.Hamaoka,
T.,
et al.,
Tumour response interpretation with new tumour response criteria vs the World Health Organisation criteria in patients with bone-only metastatic breast cancer. Br J Cancer,
2010.
102(4): p.
651-7.
3.Choi,
H.,
et al.,
Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new...