Purpose
Apical hypertrophic cardiomyopathy (ApHCM) represents a relatively uncommon phenotype of hypertrophic cardiomyopathy (HCM),
affecting mostly the apex of the left ventricle.
This disease was underdiagnosed due to variable onset and clinical course.
Furthermore,
before the advent of cardiac MRI,
the diagnosis were hard because of the low spatial resolution of echocardiography compared to MRI,
and the insufficient capacity of the former to identify focal areas of hypertrophy,
particularly in the apex,
compared to the latter.
In the past,
the prognosis were considered to be more...
Methods and Materials
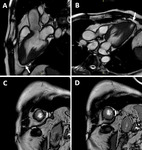
We discuss a case of seventy-ninth years old Caucasian man,
who underwent CMR on December 2016,
with suspect of cardiomyopathy.
He performed an ECG that showed some alteration of repolarization (T wave inversions in precordial leads) and echocardiography,
which demonstrate a mild wall thickening in the left ventricle.
In order to improve the assessment of patient,
the cardiologist recommended CMR.
We used a 1,5 T scanner (Achieva D-Stream,
Philips,
Best,
The Netherlands) and ECG triggering.
A multi-phase steady-state free precession sequence was applied to obtain...
Results
Genetics,
Clinical andPathophysiology
Apical hypertrophic cardiomyopathy (ApHCM) was first described in Japan by Sakamoto et al.
as a novel cardiac condition characterized by an ace-of-spades configuration of the left ventricular cavity,
apical hypertrophy,
and giant negative T waves.
ApHCM is seen as a variant of global hypertrophic cardiomyopathy in which act particular genetic factors.
It presents often a congenital autosomal dominant aetiology.
Recent studies suggest the association with genetic mutations in ACTC1,
TPM1,
MYBPC3 and MYH7.
Symptoms include chest pain,
palpitations,
dyspnea,
light-headedness and syncope,...
Conclusion
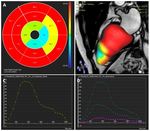
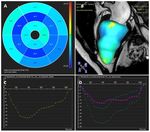
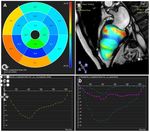
CMR play an important role in the management of apical hypertrophic cardiomyopathy,
because it can detect an unusual phenotype with thin-walled aneurysms and its complications,
as thromboembolic phenomenon,
that need a systemic anticoagulant for stroke prevention.
LGE may be reliable regional areas of myocardial fibrosis,
especially in thin-walled of aneurysms,
composed mainly of fibrosis,
which extends into the septum and free wall.
This condition represents a substrate for malignant ventricular arrhythmias and risk of sudden death,
and also is an important factor for implantable cardioverter...