Aims and objectives
To assess venous thromboembolism incidence in patients with port-systems.
To identify VTE risk factors and to develop management strategy
Port-systems are known to create the high riskfor the development of venous thromboembolism (VTE).
Specifically,
around 15% and 4.5% of the patients have been reported to suffer from thrombotic complications and pulmonary embolism respectively [1,
2].
Taking these facts into consideration,
certain hospitals elaborate and implement the active thrombosis monitoring programs [3].
Therefore,
the objective of the present study was to evaluate the frequency of venous...
Methods and materials
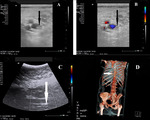
In our study 289 patients with implanted for long-term chemotherapy central venous port-systems (devices insertion commonly by subclavian access under CT-navigation,
in 5 cases by femoral access) were included (Figure 1).
Fig. 1
Mean patients' age was 45 y.o.
(27 - 61).
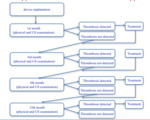
After intervention patients were involved in observational program: visits to surgeon (1,
3,
6 and 12 months),
venous ultrasound examination and echocardiography.
In case of pulmonary embolism suspicion - chest CT angiography.
Results
In 39 patients (13.5%) thrombotic device occlusion or subclavian vein thrombosis were detected.
Thromboses were treated by low-molecular weight heparins.
In 7 cases local thrombolysis was effective.
In 2 patients we have performed device explantation due to risk of thrombosis progression.
Mean time for venous thrombosis development was 3 months after intervention.
In 1 case non-massive pulmonary embolism has revealed.
Based on risk analysis male sex (p<0.05) and head and neck cancer (p=0.03) were justified as predictors for thrombotic complications (Figure 1).
Fig. 1
Additionally...
Conclusion
Active VTE monitoring in patients with implanted central venous access devices is the important part of disease management (Figure 3).
Aggressive strategy with device recanalization allows to continue chemotherapy.
We suggest male sex,
head and neck cancer and device malposition as additional independent risk factors for VTE.
References
1.Whitman ED.
Complications associated with the use of central venous access devices.Curr Probl Surg.
1996;33(4):309-378.
2.
Poorter RL,
Lauw FN,
Bemelman WA,
Bakker PJ,
Taat CW,
Veenhof CH.
Complications of an implantable venous access device (Port-a-Cath) during intermittent continuous infusion of chemotherapy.Eur J Cancer.
1996;32A(13):2262-2266.
3.
Marcy P,
Thariat J,
Lacout A,
Sanglier J,
Figl A,
Chamorey E.
Further data about catheter related venous thrombosis in oncology.Thromb Res.
2014;134(1):207-209.
doi: 10.1016/j.thromres.2014.04.003