Aims and objectives
Between August 2009 and December 2013,
Guerbet conducted a national non-interventional post-marketing surveillance (PMS) study with iobitridol (Xenetix®) in Germany.
The aim of the PMS study was to gain additional information about the safety of iobitridol as well as its efficacy in daily practice using the most up-to-date imaging and administration techniques.
Methods and materials
A two-page report form was used to record the data.
The report form included 9 sections: patient information (age,
sex,
height,
weight),
known risk factors (bronchial asthma,
CNS disorder,
coronary heart disease,
diabetes mellitus,
diuretics,
heart failure,
history of a reaction to a contrast agent,
hypertension,
known allergies,
pheochromocytoma,
poor general condition,
severe liver dysfunction,
sickle cell anemia,
thyroid dysfunction,
water balance/electrolyte metabolism disturbed),
impaired renal function,
premedication,
type of examination,
injection of contrast agent,
imaging quality,
diagnostic value and safety.
The data was descriptively...
Results
A total of 94,960 examinations with iobitridol (53.1% male,
46.9% female,
mean (±SD) age 58.8 ± 15.6 years) were carried out in 168 centres,
predominantly CTexaminations (49.3%) or IV urography (48.8%).
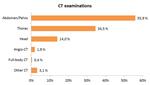
The most common CT examinations were of the abdomen/pelvis (55.9%),
thorax (34.5%) and head (14.0%) [Fig.
1].
For the CT examinations,
the mean (±SD) volume of contrast agent administered was 89.9 ± 19.9 mL with an injected volume between 50 and 120 mL in 90% of the patients [Fig.
2].
Patients who underwent IV...
Conclusion
Adverse events occurred in 0.5% of patients.
The most common adverse events were nausea,
urticaria,
dizziness and rash.
Serious adverse events were observed in 0.03% of patients.
Examination with iobitridol enabled a diagnosis to be made in 99.6% of all cases (CT examination 99.9%,
IV urography 99.3%),
with excellent or good imaging quality (96.6%).
The results demonstrate that iobitridol (Xenetix®) is a safe contrast agent that enables diagnosis with optimal imaging quality.
References
Maurer M.,
et al.
Safety and tolerability of iobitridol in general and in patients with risk factors: Results in more than 160 000 patients.
Eur J Radiol 80,
357-362 (2011)
Wendt-Nordahl G.,
et al.
Intravenous contrast media in uroradiology: Evaluation of safety and tolerability in almost 50,00 patients.
Med Princ Pract 15 (5),
358 – 361 (2006)
Vogl TJ.,
et al.
Safety of iobitridol in the general population and at-risk patients.
Eur Radiol 16 (6),
1288-1297 (2006)
Petersein J.,
et al.
Results of the safety...