Learning objectives
Over the last years we have developed a security program at the Magnetic Resonance Imaging (MRI) area in the radiology department to improve the quality of care and safety of the patients.
The special conditions of security on MRI make necessary to have a strict control of patient’s devices compatibility before performing an MRI study.
Specific training and involvement of all personnel involved in the MRI studies is needed to guarantee patient’s safety and offer a high quality service.
Background
-To ensure the completion of the Magnetic Resonance studies with optimal safety conditions for the patient.
-To identify in advance patients with patients with non-compatible MRI devices to reduce the number of rescheduling and/or cancellations
Findings and procedure details
We review the MRI request to avoid request or appointment errors.
The patient's clinical history is then reviewed to evaluate if he or she has any device or implant that contraindicates the MRI examination.
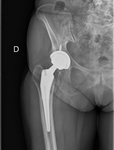
Joint prostheses and common surgical materials are usually compatible.
We pay special attention to devices such as pacemakers,
implantable cardioverter defibrillator (ICD),
cardiac valves,
cochlear implants,
neurostimulators and any other devices that may be incompatible with MRI.
In the case of pacemakers and ICD we need to know if the device...
Conclusion
With the systematic review of the medical records in patients with an MRI appointment,
it has been possible to minimize the number of recitations in cases with contraindications or in those who require a specific device preparation before and after the study.
Thought this systematic review of the clinical records in advance has significantly improved the quality of care and safety provided to our patients.