Aims and objectives
Nowadays immunotherapy is widely used in oncology for different kind of primary tumous and it has the main effect to improve the immune system in identifying and fighting tumour cells.
Nivolumab is a PD-1 (Programme Death cell-1) check-point inhibitor that distrupts PD-1 mediated signalling and restores anti-tumour immunity.
Nivolumab is the only PD-1 inhibitor approved for a broad range of patients with previously treated metastaticNon Small Cell Lung Cancer (NSCLC,
stage IIIb-IV)regardless of PD-L1 expression [1,2].
Nivolumab,
like others Immune Check-Point Inhibitors,
may cause atypical...
Methods and materials
In this retrospective study,
toghether with the Departments of Oncology and Radiotherapy of our Institution,
we evaluated patients with NSCLC stage IIIb/IV receiving second-line Nivolumab therapy.
Period: January 2016- March2018.
Patients:n=56,
all with the following demographic data: age,
gender,
stage of the disease,
tumour histology, Pack-years (if smokers).
Inclusion criteria:
patientswith NSCLS stageIIIb–IVreceivingNivolumab in 2°linetherapy
onetargetlesionatleast
followup > 3months
2or morefollow upCT
failure of 1° linechemotherapy.
Exclusion criteria:36 patients were excluded because of:
absence of targetlesions (n=11)
relapsing lesions after radiotherapy (n=2)
previous radiotherapy (n=4)...
Results
Patients were 46 males and 10 females (aged 48-84,
mean age 68 years).
All were smokers and Median Pack-year was 48.2 (range 6-120 p/y).
Tumour stage was IIIb in 16/56 (28.6%) and IV in 40/56 (71.4%).
Tumour histology was adenocarcinoma in 27/56 (48.2%) and squamous-cell carcinoma in 29/56 (51.8%).
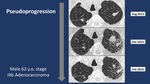
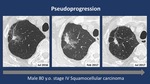
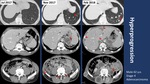
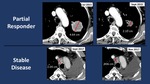
Using iRECIST criteria,
radiological analysis of CT scans demonstrated:
Pseudoprogression patternin 3/56 (5.3%)
Hyperprogression pattern in 5/56 (8.9%)
Other Responders (OR):
- CR: in 0 patients
- PR: in 6/56 (10.7%)
- SD: in 17/56 (30.3%)...
Conclusion
Althought having experienced differences in the demographic data of the three responders groups,
regarding expecially patients age and tumor hystology,
these differences were not statistically significant and we were not able to find a useful clinical or histological index suggesting us the most probable of CT response in patients undergoing Nivolumab therapy.
Our results highlight the necessity of identifying predictors,
in particular of Hyperprogression,
in order to avoid Nivolumab treatments in patients at risk.
We also think that a wider population may will possibly help...
Personal information
Dr.
Chiara Moroni
Department of Emergency Radiology
Careggi University Hospital
L.go Brambilla,
3
50134 Florence
Italy (IT)
Dr.
Diletta Cozzi: Department of Emergency Radiology - Careggi University Hospital,
Florence
Dr.
Maurizio Bartolucci: Department of Emergency Radiology - Careggi University Hospital,
Florence
Dr.
Federico Giannelli: Department of Emergency Radiology - Careggi University Hospital,
Florence
Dr.Viola Bonti:Section of Respiratory Medicine - Careggi University Hospital,
Florence,
Italy.
Dr.Vieri Scotti:Department of Oncology,
Radiation Therapy Unit - Careggi University Hospital,
Florence,
Italy.
Dr.
Francesca Mazzoni: Department of Oncology - Careggi...
References
Rizvi NA et al.
Activity and safety of nivolumab,
an anti-PD-1 immune checkpoint inhibitor,
for patients with advanced,
refractory squamous non-small-cell lung cancer (CheckMate 063): a phase 2,
single-arm trial.
Lancet Oncol. 2015;16(3):257–65.
Ellis PM et al.
Immune Checkpoint Inhibitors for patients with advanced non-small cell lung cancer: a systematic review.
Lung Cancer. 2017; 18(5):444-459.
Kwak JJ et al.
Cancer immunotherapy: imaging assessment of novel treatment response patterns and immune-related adverse events.
Radiographics.
2015;35(2):424–37.
Borghaei H et al.
Nivolumab versus docetaxel in advanced non squamous...