Aims and objectives
PI-RADS is a guide for making a structured report and decreasing inter-observer variability
Management of PI-RADS 3 category patients is not well defined,
leaving the indication for biopsy solely to the urologist’s decision.
The positivity rate for clinically significant prostate cancer in PI-RADS 3 patients has been reported as 24.8%.
Fig. 1
Prostate cancer can be classified according to its histological pattern in low,
intermediate and high risk,
where the latter two are categorized as clinically significant.
Fig. 2
PSA values have been considered an...
Methods and materials
Retrospective study,
approved by the ethics committee of our institution.
All multiparametric prostate MRI (mpMRI) reports were identified in our PACS between January 2016 and December 2017 (48 months).
Inclusion criteria for the analysis were:
1) Radiological report with a category PI-RADS 3.
2) Histological result of transrectal biopsy available
Data such as age,
prostate-specific antigen and prostatic volume (calculated at the time of the radiological report) were extracted.
Prostate-specific antigen density was calculated (PSA / Prostatic Volume = PSA Density).
All biopsies results were...
Results
The group of institutional patients (267 patients,
134 biopsied and 133 non-biopsied) was characterized according to age,
prostate-specific antigen,
and prostatic volume.
Mean age of the biopsied group was 60 years and that of the non-biopsied group was 62 years.
(p = 0.03).
There were no significant differences between the patients biopsied and non-biopsied in regards to prostate-specific antigen,
prostate volume and PSA-density. Fig. 5
PSA-density distribution of the biopsied patients showed skewed shape with a greater number of patients with prostate-specific antigen density close...
Conclusion
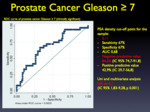
PSA density is a variable that can help guide management of these cases.
In our population,
a PSA density greater than 0.11 ng/ml/cc had an OR of 4.1 for a clinically significant cancer.
This threshold is lower than the one previously described by the literature (between 0.15 and 0.20 ng/ml/cc).
As a result of our work,
we propose a lower cut-off value for the indication of biopsy in our PI-RADS score 3 patients.
It should be noted that in the period of time studied,
a...
Personal information
A.
Labra Weitzler,
F.
Tapia,
C.
Silva,
J.
P.
Olivares,A.
Zuñiga;
Facultad de medicina Clinica Alemana de Santiago - Universidad del Desarrollo.
Department of Images,
CT - MRI Service.
Vitacura 5951,
Vitacura,
Region Metropolitana - Santiago - Chile.
Postal Code: 7650568
Phone number +56 2 22101111
email:
[email protected] /
[email protected]
References
Mehralivand S,
Bednarova S,
Shih JH et al (2017) Prospective Evaluation of PI-RADS™ Version 2 Using the International Society of Urological Pathology Prostate Cancer Grade Group System.
The Journal of Urology 198:583-590
Hassanzadeh E,
Glazer DI,
Dunne RM,
Fennessy FM,
Harisinghani MG,
Tempany CM (2017) Prostate imaging reporting and data system version 2 (PI-RADS v2): a pictorial review.
Abdominal radiology (New York) 42:278-289
Distler FA,
Radtke JP,
Bonekamp D et al (2017) The Value of PSA Density in Combination with PI-RADS for the Accuracy of...