Aims and objectives
To evaluate the value of MR imaging features
in predicting pancreatic neuroendocrine tumors (PNETs) with somatostatin receptor-2(SSTR2) expression.
Methods and materials
Between January 2010 and September 2018,
65 patients of PNETs with preoperative MR imaging were included.
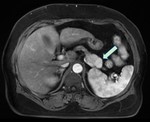
Tumor grading was based on the new 2017 World Health Organization classification of PNETs.MR imaging featuresincluded size,
shape,
consistency,
contrast enhancement pattern,
and Contrast-to-noise ratios (CNRs) of enhancement.
SSTR2 expression of all the resected specimens were assessed by immunocytochemistry.
Results
13 tumors with negative SSTR2 and 52 tumors with positive SSTR2 wereevaluated.
The positive SSTR2 group showed male(48.1% vs 84.6%,P < 0.05 ),
hypointense signal on arterial phase (23.1% vs 76.9%,
P < 0.05),
portal venous phase (13.5% vs 61.5%,
P < 0.05),
and delayed phase (9.6% vs46.2%,
P < 0.05) less frequently than the negative SSTR2 group,
as opposed to the G1(34.6 % vs 7.7%,P < 0.05 ).
However,
The CNRs of arterial phase of the positive SSTR2 group were larger than the negative...
Conclusion
The hyper/isointense signal of arterial phase may help to predict PNETs with SSTR2 expression.
Personal information
Yabin Hu,Department of Radiology,
Zhongshan Hospital,
Fudan University,
and Shanghai Institute of Medical Imaging,
Shanghai,
China
References
de Herder W W.
When and How to Use Somatostatin Analogues[J].
Endocrinology and Metabolism Clinics of North America,
2018,47(3):549-555.
Werner R A,
Ilhan H,
Lehner S,
et al.
Pre-therapy Somatostatin Receptor-Based Heterogeneity Predicts Overall Survival in Pancreatic Neuroendocrine Tumor Patients Undergoing Peptide Receptor Radionuclide Therapy[J].
Molecular Imaging and Biology,
2018.
Cives M,
Strosberg J R.
Gastroenteropancreatic Neuroendocrine Tumors[J].
CA Cancer J Clin,
2018,68(6):471-487.
Carmona-Bayonas A,
Jiménez-Fonseca P,
Custodio A,
et al.
Optimizing Somatostatin Analog Use in Well or Moderately Differentiated Gastroenteropancreatic Neuroendocrine Tumors[J].
Current Oncology...