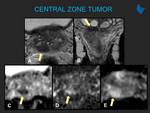
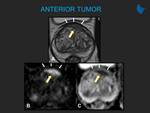
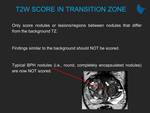
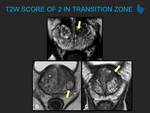
Learning objectives
To highlight the Prostate Imaging Reporting and Data System (PI-RADS) version 2.1 (v2.1) changes in interpretation criteria and technical specifications of image data acquisition;
To illustrate the clarifications and revisions in image interpretation criteria;
To discuss the role of dynamic contrast-enhanced magnetic resonance imaging (DCE MRI) and the implications of biparametric magnetic resonance imaging (bpMRI) for PI-RADS assessment categories.
Background
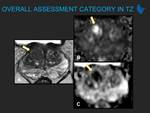
Prostate Imaging Reporting and Data System (PI-RADS) was first released in 2012 and designed to promote global standardization and diminish variation in the acquisition, interpretation, and reporting of prostate multiparametric magnetic resonance imaging (mpMRI) examination in assessing categories that summarize levels of suspicion or risk of clinically significant prostate cancer (csPCa). Since then, numerous studies have validated the value of PI-RADS, but, as expected, they have also shown some inconsistencies and limitations, what, added the advances in technology (both in software and hardware), guided the...
Findings and procedure details
TECHNICAL SPECIFICATIONS OF IMAGE DATA ACQUISITION
PI-RADS v2.1 presents some updates on technical specifications of image data acquisition:
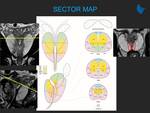
recommended T2-weighted (T2W) imaging planes to be performed;
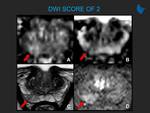
clarification of b-values to use for purposes of diffusion-weighted imaging (DWI) acquisition and apparent diffusion coefficient (ADC) map calculation;
advice about dynamic contrast-enhanced (DCE) temporal resolution and preferred imaging sequence.
T2-weighted images
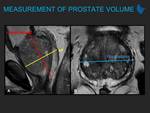
PI-RADS version 2 (v2) stated multiplanar (axial, coronal, and sagittal) T2W images are usually obtained. Instead, PI-RADS v2.1 recommends that T2W images should always be obtained in...
Conclusion
PI-RADS system is a living document with adaptions being made as evidence accumulates in its use in practice. The evolution of PI-RADS has led to version 2.1, whose adoption will probably improve inter-reader variability and further simplify PI-RADS evaluation of prostate MRI. Research on the value and limitations on all components of PI-RADS v2.1 will eventually lead to PI-RADS 3 and it is anticipated it will include further investigations in bpMRI, applications of artificial intelligence, and potential integration of circulating tumor cells or serum and/or...
Personal information and conflict of interest
Rodrigo Bartels - nothing to disclose
Radiology resident at Hospital Moinhos de Vento, Porto Alegre/BR
E-mail:
[email protected]
Louise Torres - nothing to disclose
Radiology resident at Hospital Moinhos de Vento, Porto Alegre/BR
E-mail:
[email protected]
Ivan Morzoletto Pedrollo - nothing to disclose
Abdominal radiologistat Hospital Moinhos de Vento, Porto Alegre/BR
E-mail:
[email protected]
References
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, Thoeny HC, Verma S. Weinreb JC. PI-RADS Prostate Imaging-Reporting and Data System:2015,Version 2. EurUrol 2016;69(1):16-40
Turkbey B, Rosenkrantz AB, Haider MA, Padhani AR, Villeirs G, Macura KJ, Tempany CM, Choyke PL, Cornud F, Margolis DJ, Thoeny HC, Verma S, Barentsz J, Weinreb JC. Prostate Imaging Reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur Urol. 2019....