Purpose
The purpose of this study is to develop a multi-dimensional tissue marker to mark lesion location after biopsy.
This marker is deployed during biopsy or surgery and can be used in breast and other soft tissue organs.
The requirements of the marker include detectable (MRI,US,
x-ray) and deliverable through a trocar of less than 2 mm and the marker must be visible by a magnetic susceptibility using FDA and C Mark approved materials.
The new molecular marker must do the following:
1. Deployed during biopsy...
Methods and Materials
The breast marker consists of hermetically-sealed solid vial containing mixture of FDA-approved contrast agents to optimize MRI,
ultrasound and x-ray-based imaging using superior biocompatibility and imaging characteristics.
We optimize the liquid core using FDA-approved gadolinium and iodine solutions.
Using relaxivity mapping with 3T MRI we optimize gadolinium,
iodine,
and water for optimal imaging under MRI clinical sequences.
Our solution is as follows:
1.Embed a drop of Gd-doped water in the tissue
- bright signal on T1w images
2.
Enclose this liquid drop in a container...
Results
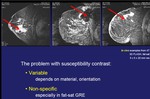
Excellent MR visibility in all tissue types.
Minimal interference with MRI and MRS.
Imaging is at least at good or better as available markers for x-ray and ultrasound.
The marker is independently optimized for MRI,
ultrasound and x-ray and shows excellent resolution in all three imagingmodalities.
The new molecular marker shows the following advantages on phantom MRI imaging:
1. Excellent MR visibility
2. Permanent
3. Can be made completely frombiocompatible materials
4. Visible on all 3 major modalities
5. Minimal interference with MRI and MRS
Conclusion
Novel MRI marker is visible under all conventional imaging modalities.
Marker has superior MRI visualization allowing for molecular imaging spectroscopy and DCE uptake.
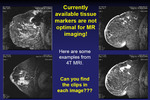
This multimodal design can outperform existing tissue markers,
especially in MRI.
The marker is currently under further development to optimize contrast and size.
Patent pending.
Acknowledgements: This work was supported under NIH grants R01CA092004,
P41-RR008079,
the MIND institute,
and the Keck Foundation
References
Saslow D,
Boetes C,
Burke W,et al.
American Cancer Society guidelines for breast screening with MRI as adjunct to mammography.
Cancer J Clin 2007; 57:75–89.
Manton DJ,
Chaturvedi A,
Hubbard A,
et al.
Neoadjuvant chemotherapy in breast cancer: early response prediction with quantitative MR imaging and spectroscopy.
Br J Cancer 2006; 94:427–435.
Kvistad KA,
Bakken IJ,
Gribberstad IS,
et al.
Characterization of neoplastic and normal human breast tissues with in vivo 1H spectroscopy: initial results.
Radiology 2001; 220:40–46.
Jagannathan NR,
Kumar M,
Seanu V,
et...
Personal Information
Michael T.
Nelson,
M.D.
Professor of Radiology
University of Minnesota Medical Center-Fairview and Fairview Breast Center,
Department of Radiology and Center for Magnetic Resonance Research