Learning objectives
To review types of subdermal contraceptive implants (SCI)
To show MRI normal appearance of SCI Jadelle®.
To distinguish MRI findings of potential pathology: misplacement/migration,
neurovascular injury,
infection.
Background
In 1983,
the first subdermal contraceptive implant,
Norplant®,
was approved by the Finnish national drug regulatory authority.
Since then,
several more implants such Jadelle® (Norplant II) and Implanon®,
the most commonly used in Spain,
have been registered.
A large body of evidence demonstrates the high contraceptive effectiveness and safety of the 5-year levonorgestrel releasing implants Norplant® and Jadelle® (1).
Implanon® is a nonbiodegradable,
progestogen only single-rod based on an ethylene vinylacetate carrier of desogestrel metabolite etonogestrel (ENG) which is released for a duration of at...
Findings and procedure details
In this section we will show two patients with nonpalpable Jadelle® subdermal implants,
retrospectively selected from our institutions.
Case 1.
A Jadelle® subdermal implant was inserted four years ago in the upper non-dominant left arm of a 29 year old female.
One year ago she presented vaginal bleeding disturbances (prolonged and irregular bleeding),
and underwent local removal.
Unfortunately,
only one rod was removed and surrounding soft-tissue was infected (cellulitis).
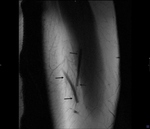
Ultrasound (figure 3) and MRI techniques (figures 4,
5) of the left arm were obtained before...
Conclusion
Little attention has been focused on MRI findings of subdermal contraceptive implants in radiology literature.
Radiologists and gynecologists must be aware of theirtypes:
Norplant®: six-capsule preparation
Jadelle®: two-rod preparation,
with a size of 43 x 2.5 mm.
Implanon®: single-rod preparation with a size of 40 x 2 mm.
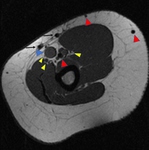
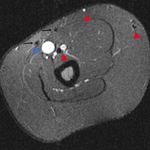
and MRI findings:hypointense signal on all sequences,
sharp margins.
Caution is needed:
When insertion is made so close to basilic or brachial vein in the medial aspect of the arm.
In our opinion an anterior location farther...
Personal information
First author:
Xavier TOMAS MD PhD
ESR / SERME Member
ESSR Councillor Executive Committee
Staff Consultant Radiology Dpt (MSK unit)
Hospital Clinic Villarroel 170 Barcelona 08036 (Spain)
University of Barcelona (UB)
Phone+34932275412
Cell :+34606837805
Emails:
[email protected]
[email protected]
References
Meirik O,
Fraser IS,
d'Arcangues C; WHO Consultation on Implantable Contraceptives for Women.
Implantable contraceptives for women.Hum Reprod Update.
2003 Jan-Feb;9(1):49-59.
Mascarenhas,
L.
Insertion and removal of Implanon.
Contraception.
1998; 58: 79S–83S
Lantz,
A,
Nosher,
J.L,
Pasquale,
S,
and Siegel,
R.L.
Ultrasound characteristics of subdermally implanted Implanon® contraceptive rods.
Contraception.
1997; 56: 323–327
Case courtesy of Dr Amit Tripathi,
<ahref="http://radiopaedia.org/">Radiopaedia.org</a>.
From the case <a href="http://radiopaedia.org/cases/misplaced-implanon">Misplaced Implanon</a>
Sarma,
S.P,
Wamsher,
J.G,
and Whitlock,
S.W.
Removal of deeply inserted nonpalpable levonorgestrel (Norplant®) implants.
Contraception.
1996; 53: 159–161...