Learning objectives
Aims
What is LI-RADS and what are it's clinical applications?
What has changed in LI-RADS 2017?
Outline
Clear explanation of how to use LI-RADS
Multiple cases illustrating both the major and ancilliary features using MRI and CT
Interactive - self test with answers provided in the concluding section
Background
Introduction
Liver cancer represents a major health burden,
it is the fifth commonest cancer in men and the ninth commonest in women and represents the second most common cause of death from cancer worldwide with an estimated 746,000 deaths in 20121.
HCC represents the commonest primary liver malignancy.
The commonest risk factor for HCC is cirrhosis,
regardless of the underlying cause.
HCC in the cirrhotic liver has specific imaging characteristics and increasingly treatment is planned based on non-invasive criteria using MRI and CT characteristics.
While...
Findings and procedure details
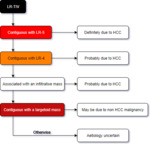
Using LI-RADS 2017
LI-RADS should be used in patients at high risk of HCC,
this includes patients with cirrhosis,
those with chronic hepatitis B infection and finally those with current or previous HCC,
whether or not there is underlying cirrhosis/chronic hepatitis B infection.
LI-RADS should not be used in patients below the age of 18,
those with cirrhosis secondary to an underlying vascular disorder or those with congenital hepatic fibrosis.
In addition,
it should not be used to assess lesions which have a pathological diagnosis...
Conclusion
Case
LI-RADS
Notes
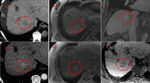
Case 1 - Fig. 4
LR-4
The lesion has inherent high T1 signal making the multiphase CT invaluable to assess for arterial phase enhancement.
Case 2 - Fig. 5
N/A
While the imaging features are of an LR-4 lesion the patient has Budd-Chiari and therefore LI-RADS should not be used.
Case 3 - Fig. 6
LR-2/3 initially then LR-3
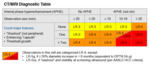
On the first study the diagnostic table would assign LR-3 however the concentration of the hepatobiliary contrast agent would allow adjustment to LR-2....
References
1http://globocan.iarc.fr/old/FactSheets/cancers/liver-new.asp
2https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS
3https://www.acr.org/-/media/ACR/Files/RADS/LI-RADS/LIRADS_2017_Core.pdf