Learning objectives
Brief notes about the pathophysiology-imaging correlation of neuroendocrine tumor (NET)
to discuss the role of morphologic imaging in patients with suspicion of NET.
CT appearance of NET
Background
NETs are a rare and heterogeneous group of neoplasms,
which are derived from APUD cells,
the so-called amine precursor uptake and decarboxylation properties system,
ubiquitously found in the digestive tract,
in pancreas,
respiratory system and producing serotonin and histamine-like substances.
It was O.
Lubarsch,
a German pathologist native of Berlin,
et al [1] in 1888,
at the University of Munich,
who provided the first detailed description of a carcinoid tumor.
He gave the description of ileal carcinoids,
called “little carcinomata”,
originating in the intestinal crypts...
Findings and procedure details
Epidemiology and presentation
The majority NETs are sporadic,
with a median age at diagnosis of 63 years [3],
but they could,
sometimes,
occur in patients with complex tumor susceptibility genetic syndromes.
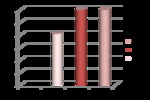
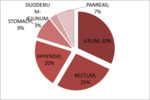
The tumor distribution varies in different parts of the world [3,4] (Fig.1,
Graphic 1).
These tumors occur most frequently in the gastrointestinal tract (67% of cases),
followed by the tracheobronchial system (24%).
In the gastrointestinal tract 30% of neuroendocrine tumors (NETs) occur in the ileum (5),
followed by the rectum (21%–27%) and the appendix...
Conclusion
NETs are a heterogeneous and complex group of neoplasms with a wide spectrum of clinical manifestations.
Morphologic imaging,
such as multidetector CT, is the most widely used technique for the initial evaluation and for exclusion of metastatic disease,
while functional imaging techniques (e.g.,
somatostatin receptor scintigraphy) are useful both for detecting tumors and selecting patients for receptor-targeted therapy.
Surgery is the potentially curative treatment for NETs and their metastases.
References
1.Lubarsch O.
Ueber den primären krebs des ileum nebst bemerkungen über das gleichzeitige vorkommen von krebs und tuberculose.Virchows Arch Pathol Anat1888;111:280–317.
2.Feyrter F.
Über diffuse endokrine epitheliale Organe.Zentralblatt Innere Medizin 1938;59:546–556.
3.Yao JC,
Hassan M,
Phan A et al.
One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States.
J Clin Oncol 2008;26:3063–3072.
4.Ito T,
Sasano H,
Tanaka M et al.
Epidemiological study of gastroenteropancreatic neuroendocrine tumors in Japan.
J Gastroenterol 2010;45:234–243.
5.Heller MT,Shah AB.Imaging...