Cardiac rupture is a life-threatening event.
The most common cause of myocardial rupture is a recent myocardial infarction.
Other causes include cardiac trauma,
endocarditis,
cardiac tumors and takotsubo cardiomyopathy.
The incidence of cardiac rupture has decreased in the era of urgent revascularization and aggressive pharmacological therapy to treat acute myocardial infarction1.
However,
it is still a common cause of in-hospital death in STEMI.
According to a recent report from the Global Registry of Acute Coronary Events,
myocardial rupture occurs in 0.45% of all myocardial infarctions2,
but its incidence is probably underestimated because an imprecise number of patients may present self-limited forms.

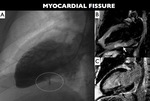
Fig. 1: A) Left ventriculogram showing a diverticulum-like outpouching of the left ventricular inferior wall suggesting impending cardiac rupture. B) STIR two-chamber image and C) delayed contrast-enhanced two-chamber image two days later demonstrating focal edema (white arrow) with a transmural serpiginous hyperenhancement (black arrow) in the inferior wall due to a self-limited myocardial tear (see Fig.2: video 1)

Fig. 2: video 1: Short-axis cine-MRI after gadolinium injection showing a transmural serpiginous hyperenhancement signal consistent with healed myocardial fissure in the LV inferior wall
Cardiac rupture can be classified as:
1- Free-wall rupture (FWR),
the most common type and associated with high mortality
2.- Ventricular septal rupture,
causing a ventricular septal defect
3.- Papillary muscle rupture,
resulting in acute mitral regurgitation

Fig. 3: Axial and coronal CT images demonstrating a ventricular septal defect (arrow) due to a ventricular septal rupture after RCA infarct
According to the type of presentation,
FWR can be considered:
1.- acute: presenting with sudden death caused by severe hypotension and electromechanical dissociation secondary to acute pericardial tamponade
2.- subacute: an increasingly recognized form,
presenting with hypotension of differing severity without cardiac arrest by electromechanical dissociation.
This definition implies that the subacute form is clearly less severe and more likely to recover with successful medical management.
According to time of occurrence,
FWR can be considered:
1.- early: when it occurs within the first 48 hours.
Factors such as persistent strain in the infarcted area due to sustained arterial hypertension or maintenance of ambulatory activities appear to be of importance.
2.- late: when it occurs beyond the second day.
Autopsy studies have indicated that infarct extension may be a trigger of late FWR.
DIAGNOSIS
Cardiac rupture should be suspected when a patient with a high-risk clinical profile suddenly presents hypotension or electromechanical dissociation associated with moderate to severe pericardial effusion
High-risk clinical profile of patients with FWR3:
- Age >55 years
- First transmural myocardial infarction
- Killip class I or II
- Persistent ST segment elevation
- Persistent or recurrent chest pain
- Sudden or progressive hypotension or sudden electromechanical dissociation
MRI is not the investigation of choice in acute patients with hemodynamic instability.
In the acute phase,
the diagnosis is often made by echocardiography,
but MRI plays an important role in the subacute phase when the patient is stable.
Acute findings:
Blowout ruptures manifest with a catastrophic presentation and death within minutes.
Echocardiography is the procedure of choice for the diagnosis of cardiac rupture in patients who are hemodynamically unstable.
Emergent bedside echocardiography showing a pericardial effusion is highly suggestive of FWR.
If the pericardial effusion is hemodynamically compromising,
the suspicion of FWR increases,
especially if electromechanical dissociation occurs.

Fig. 4: Transthoracic echocardiogram shows severe apical hypokinesis with large pericardial effusion, suggesting a cardiac rupture in a patient with AMI who suddenly presents hypotension
Sometimes heart rupture is not sudden and fatal because it is incomplete or contained; ventriculography may outline the rupture. In these cases,
the rupture does not traverse the myocardial layers,
epicardial fat,
and visceral pericardium.

Fig. 5: Acute left ventriculogram showing an apical defect suggesting impending cardiac rupture and subacute contrast-enhanced two-chamber image demonstrating a transmural infarction with an apical thrombus (white arrow) sitting in the myocardial defect
Subacute findings:
When cardiac rupture does not result in immediate fatal pericardial tamponade with hemodynamic collapse,
the definite diagnosis is difficult and often based on subtle MRI findings:
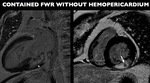
- A subset of patients with subacute FWR may present with moderate to severe hemopericardium and no apparent cardiac rupture. It is likely that some self-limited FWRs are rather small and open temporarily only during bursts of arterial hypertension or increased myocardial strain.
Some may even consist of a stuttering oozing through a narrow fissure.
Some necropsy studies of AMI with hemopericardium have found FWRs that are not apparent macroscopically5,6.

Fig. 6: A) Short-axis cine-MRI showing a moderate hemopericardium (asterisks) with heterogeneous signal intensity due to due to its hemorrhagic contents. B) Delayed contrast-enhanced 3-chamber image demonstrating an apical transmural necrosis without mural thrombus or defects
- Although free wall rupture occurs in infarcts of variable size,
it is not uncommonly found in association with a rather small area of lateral necrosis.

Fig. 7: A) Four-chamber cine image shows bilateral pleural effusion and circumferential heterogeneous pericardial effusion (asterisks), consistent with hemopericardium. B ) Delayed contrast-enhanced short-axis image demonstrates small lateral transmural necrosis (arrow)
- Smaller or incomplete tears may be sealed by a thrombus sitting at the orifice or pericardial adhesions; thus,
massive blood extravasation is prevented7.
Identification of an intramyocardial hypoenhanced dot within the infarcted area,
suggesting a clot,
in the absence of a severe hemopericardium enables a diagnosis of contained rupture.

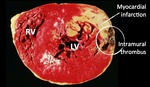
Fig. 8: Gross specimen of a FWR with subacute clinical presentation after myocardial infarction, showing healed myocardial infarct with intramural thrombus

Fig. 9: Two-chamber and short-axis delayed contrast-enhanced images demonstrating a transmural inferolateral infarction (arrowheads) with a intramyocardial clot (arrow) and mild pericardial effusion (asterisk)

Fig. 10: A) Four-chamber cine image showing a myocardial tear (arrow) contained by a small thrombus (arrowhead) within the epicardial fat (asterisks). B) Delayed contrast-enhanced two-chamber image demonstrating a large transmural infarction

Fig. 11: A) Four-chamber cine image showing a complex pericardial effusion with stranding (arrowheads) and focal pericardial apical thickening due to pericardial adherences (see Fig.12: video 2). B) Delayed contrast-enhanced four-chamber image demonstrating a transmural apical infarction with enhancement of the adjacent pericardium

Fig. 12: video 2: Four-chamber cine image showing pericardial effusion with focal pericardial apical thickening and pericardial adherences
- Large areas of no-reflow and myocardial hemorrhage are often seen in cases of cardiac rupture.
Lack of contrast enhancement within the infarction due to no-reflow phenomenon may obscure identification of mural clots.

Fig. 13: A) Four-chamber cine image showing pericardial effusion in a patient with subacute FRW. B) Delayed contrast-enhanced four-chamber image demonstrating a large transmural infraction with hypoenhancement wihtin the infarct core due to no-reflow areas that obscure identification of mural clots
Chronic findings:
A subacute FWR containing thrombus and pericardial adhesions would progressively evolve into definite wall healing or pseudoaneurysm formation.

Fig. 14: short-axis delayed contrast-enhanced image 3 days after AMI shows a transmural inferolateral infarction with a mural clot (arrow) in a patient with subacute FWR. 9 months later short-axis delayed contrast-enhanced image shows a healed myocardial infarct in the inferolateral wall without pseudoaneurysm formation
The ventricular pseudoaneurysm wall consists only of epicardium and pericardium.
In contrast,
a true ventricular aneurysm contains myocardial tissue.
Absence of a myocardial layer makes the pseudoaneurysm susceptible to rupture.

Fig. 15: Two-chamber cine-MRI showing a defect in the inferior wall of left ventricle with a pseudoaneurysm contained by a thin layer of visceral pericardium
Differentiation between ventricular pseudoaneurysms and aneurysms can be challenging.
One way to assess them is to check the diameter of the aneurysm neck.
Typically,
pseudoaneurysms have a narrower neck than the diameter of the aneurysm.

Fig. 16: PSEUDOANEURYSM: Two-chamber delayed contrast-enhanced image showing necrotic tissue at the edge of the psuedoaneurysm (arrow) and large mural thrombus in the psuedoaneurysm wall (arrowheads).
TRUE ANEURYSM: Two-chamber delayed contrast-enhanced image demonstrating necrotic myocardial tissue (arrows) in the aneurysmal wall
Late gadolinium enhancement may also help in their differentiation.
Theoretically,
a true aneurysm will show late enhancement,
whereas a pseudoaneurysm will not because its wall is not comprised of necrotic myocardium.
However,
late gadolinium enhancement of the pericardium is a common finding in contained LV rupture8.
In our experience,
we have noticed that pseudoaneurysms sometimes show small foci of late gadolinium enhancement at the edge of the neck due to myocardial discontinuity at the infarcted region.
On rare occasions,
a true aneurysm can be associated with midventricular or apical hypertrophic cardiomyopathy with a narrow neck,
mimicking a pseudoaneurysm.

Fig. 17: A) Four-chamber cine image showing a true aneurysm with a narrow neck, mimicking a pseudoaneurysm in a patient with midventricular hypertrophic cardiomyopathy. B) Delayed contrast-enhanced four-chamber image demonstrating a diffuse apical enhancement with myocardial thinning in the aneurismatic region