IMAGING TECNIQUES
CHEST RADIOGRAPH
Initial and sometimes only study to provide information regarding structure and function of the cardiovascular system.
Allows assessing size of cardiac chambers,
calcifications,
left and right cardiac dysfunction,
pulmonary circulation,
pulmonary artery hypertension and congenital heart abnormalities.
Calcifications are better depicted in an overexposed radiograph and with a tangential plane,
though superposition of other structures (i.e.
heart or spine) may hamper its visualization.
MULTISLICE CT
Very sensitive technique for detection,
localization and quantification of cardiovascular calcifications.
Particularly useful for coronary disease evaluation.
ECHOCARDIOGRAPHY
Allows initial evaluation of cardiovascular diseases related to the presence of calcifications though it is not the most appropriate imaging technique for its assessment.
MRI
Allows a functional approach to the evaluation of cardiovascular diseases related to calcifications.
Most useful for assessing perfusion and myocardial viability in ischemic heart disease.
CARDIAC CALCIFICATIONS
PERICARDIAL CALCIFICATIONS
Clinical data: Inflammatory and fibrotic areas.
Most frequent causes: Heart surgery,
traumatism with hemopericardium and radiotherapy.
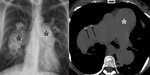
Other causes: Infections including tuberculosis (Fig. 1),
rheumatic disease,
collagen diseases (lupus),
uremic pericarditis.
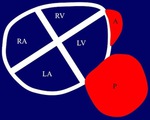
Imaging diagnosis: Most frequent location: Right side,
lower and diaphragmatic portions and atrioventricular grooves.
Curvilinear or plaques following the cardiac contour.
Thicker calcifications in long-term disease.
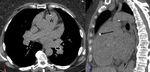
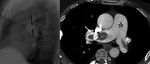
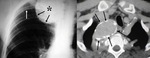
Chronic hematoma (Fig. 2 and Fig. 3): Calcium deposits in residual intrapericardial blood.
Constrictive pericarditis in 0.2-0.3%.
Also heart failure may appear.
Ranging from 2 weeks to 30 years after surgery.
Underlying factors: Traumatic surgery,
hemopericardium,
infection and post-pericardiectomy inflammation.
Calcification may occur in chronic stage.
Differential diagnosis with coronary or ventricular pseudoaneurysms and neoplasms.
Lack of contrast enhancement on MSCT and MRI.
MYOCARDIAL CALCIFICATIONS
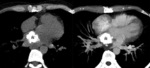
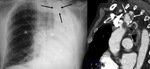
Papillary muscle: Uncommon.
After myocardial infarction.
Related to: coronary disease,
dilated cardiomyopathy,
hypercalcemia,
mitral valve insufficiency and chronic renal insufficiency.
Not visible due to its tiny size at chest radiograph.
Located in the left ventricle (Fig. 4).
Contrast enhanced MSCT shows the location in the papillary muscles (Fig. 5).
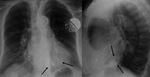
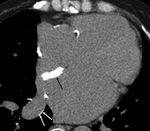
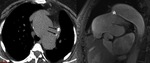
Cardiac hydatid cyst (Fig. 6 and Fig. 7): Echinococcus granulosus infection.
Endemic in countries with important ovine cattle industry.
Transmission through domestic cats and dogs.
Heart involvement in 0.5-2%.
Localization: Left ventricle (60%),
right ventricle (10%),
pericardium (7%),
pulmonary artery (6%),
left atrium appendage (6%) and interventricular septum (4%).
Myocardial invasion through coronary and pulmonary venous circulation.
The presence of calcifications may render the cyst visible at chest radiograph.
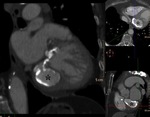
At MSCT,
precise location of the cyst and detection of calcifications or other minor associated lesions.
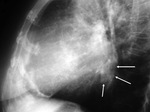
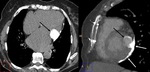
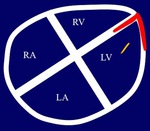
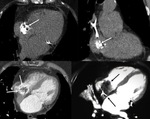
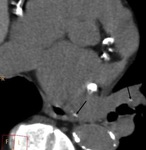
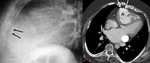
Left ventricle pseudoaneurysm (Fig. 8 and Fig. 9): Myocardial tear on the left ventricle wall contained by fibrous tissue and pericardial adherences preventing lethal outcome secondary to hemopericardium and cardiac tamponade.
Usually found after infarction.
Risk factors: elders,
females,
systemic arterial hypertension,
previous transmural myocardial infarction and lack of collateral coronary vascularization at the infarction area.
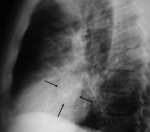
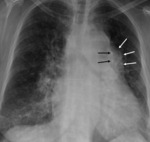
At chest radiograph,
focal posterior and lateral bulging in left ventricle,
thin and curvilinear calcifications.
Increase in size in consecutive radiographs should raise concerns of rupture.
MSCT allows differentiation of true aneurysm (Fig. 10) and assessment of size,
thrombus presence,
left ventricular function,
myocardial viability and coronary arteries stenosis.
VALVULAR CALCIFICATIONS
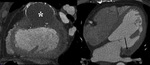
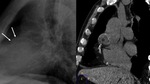
Caseous mitral (Fig. 11 and Fig. 12): Rare variant of the anular mitral calcification (present in 2.7% of patients over 50 in necropsy studies).
Calcium deposits on periannular tissue with central liquefaction containing calcium,
fatty acids and cholesterol.
Asymptomatic elders.
Initial diagnosis with echocardiography.
Chest radiograph shows peripherally calcified oval lesion projected on mitral valve zone.
At MSCT,
hyperattenuating mass,
well defined,
oval shaped with peripheral calcification.
Nonenhancing after intravenous contrast administration and no soft tissue densities (in contrast to cardiac tumors).
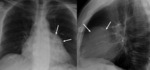
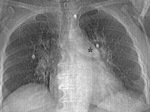
Pulmonary valve (Fig. 13 and Fig. 14): Unusual finding.
In adult patients with pulmonary valve stenosis,
tetralogy of Fallot,
pulmonary arterial hypertension and after bacterial endocarditis.
Pulmonary valve is more cephalic than the rest of cardiac valves.
On posteroanterior view of chest radiograph is seen below and medial to the edge of the main pulmonary artery between spine and left atrial appendage.
On lateral view in the upper zone of the heart posterior to sternum.
MSCT allows detection of small calcifications and findings related to pulmonary arterial hypertension.
INTRACAVITARY CALCIFICATIONS
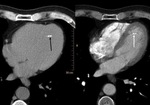
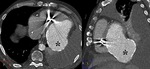
Cardiac tumors (Fig. 15 and Fig. 16): Myxoma is the most frequent among calcified cardiac tumors.
Usually located in left atrium,
myxomas may provoke mitral regurgitation or stenosis.
Other tumors: teratomas,
fibromas,
rabdomyosarcomas,
carcinoid tumors,
endotheliomas,
sarcomas and metastasis.
Calcifications present on chest radiograph in 5-10%.
Large calcifications may be seen in cardiac osteosarcomas.
MSCT allows assessment location and extension of the tumor.
Myxomas usually appear as low-attenuating masses in left atrium with or without calcifications.
Cardiac osteosarcomas may have large calcifications.
Primary osteosarcomas are located in left atrium whereas secondary tumors are usually located in right cardiac chambers.
Cardiac thrombus (Fig. 17): Very uncommon.
Location: Left ventricle (thrombus post-infarction with risk of embolization),
left atrium (usually in left appendage or surrounding posterior wall) and right atrium (in relation to central venous catheters).
At chest radiograph,
very difficult to identify even if large in size.
MSCT allows location and depiction of the thrombus and associated findings (central venous catheter).
VASCULAR CALCIFICATIONS
AORTIC CALCIFICATIONS
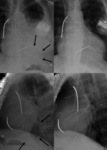
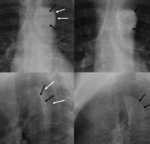
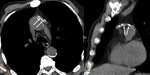
Dissection (Fig. 18 and Fig. 19): The most frequent cause of acute aortic emergency.
Arterial hypertension is the most prevalent risk factor (60-90%).
Chest radiograph is normal in 10-40% of patients.
Findings: Medial shift of calcification over 10 mm (14%),
superior mediastinal widening (61%),
aortic arch haziness or double aortic contour,
changes in aortic configuration in consecutive chest radiographs.
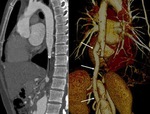
MSCT allows differentiation between type A and type B dissection with implications in treatment.
Also depicts entrance tear in tunica intima and possible involvement of visceral branches and its relationship with true and false lumen.
Intimal flap in approximately 70%.
Calcified false lumen may be seen in chronic dissections.
Takayasu (Fig. 20): Inflammatory vascular disease affecting aorta and main branches and pulmonary arteries.
More frequent among Asian women.
The only type of aortitis causing thoracic aortic stenosis.
At chest radiograph,
aortic calcification in approximately 18% of cases.
MSCT is particularly useful in early-stage disease when aortic wall thickening may be seen.
As disease progresses stenosis,
occlusions and aneurysms may appear.
In late-stage disease extensive aortic calcification may develop.
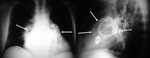
Aortic pseudoaneurysm (Fig. 21 and Fig. 22): Causes: Thoracic trauma or cardiac surgery (valve replacement or coronary bypass grafting).
Ascending aorta pseudoaneurysm appears in less than 0.5% of cardiac surgical procedures.
Chest radiograph: Focal or diffuse mediastinal widening.
Calcifications may be seen in chronic pseudoaneurysms.
Typically located at the isthmus in cases with previous trauma.
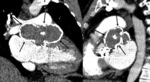
MSCT evaluates surrounding structures,
detects complications and defines surgical planning.
PULMONARY ARTERY CALCIFICATIONS
Uncommon.
Chronic pulmonary arterial hypertension as the main cause of pulmonary artery calcifications.
Chest radiograph is the initial study.
Findings of chronic pulmonary arterial hypertension: pulmonary trunk dilatation,
peripheral arteries pruning,
right interlobar artery dilatation and retrosternal space occupation due to right ventricular dilation.
At MSCT,
pulmonary trunk diameter over 29 mm.
Congenital left-to-right shunts may be seen causing chronic pulmonary hypertension (atrial and ventricular septal defects,
patent ductus arteriosus and anomalous pulmonary venous return).
Eisenmenger syndrome if bidirectional or reverse shunt.
Findings include aneurysmatic dilatation of central segments of pulmonary arteries,
thrombus with or without calcification and mural atheromatous calcification of pulmonary arteries (Fig. 23).
Chronic thromboembolism: Lung hyperlucency on chest radiograph.
Pulmonary artery calcification may be seen in some cases.
MSCT findings: thrombus in contact with vascular wall that may become calcified (Fig. 24),
vascular stenosis and signs of pulmonary arterial hypertension.
SUCLAVIAN ARTERIES CALCIFICATIONS
Subclavian arteries may show calcifications under different circumstances: atherosclerosis,
vasculitis,
infections,
iatrogenic and post-trauma.
Aneurysms (Fig. 25): Atherosclerosis the main cause.
May be associated with aortic coarctation and mural calcifications secondary to atherosclerosis peripherally visible on chest radiograph.
MSCT depicts extension of the aortic coarctation and the presence of collateral pathways.
BRONCHIAL ARTERIES CALCIFICATIONS
Bronchial arteries are responsible for hemoptysis in most cases.
Bronchial arteries dilatation if caliber exceeds 2 mm.
Aneurysms (Fig. 26): Present in less than 1% of selective bronchial arteriographies.
Predisposing factors: silicosis,
bronchiectasis,
lung neoplasm,
recurrent infections,
cystic fibrosis and atherosclerosis.
Two types: pulmonary (massive or intermittent hemoptysis) and mediastinal (mediastinal mass,
superior vena cava obstruction,
dysphagia,
hemothorax,
hemomediastinum and hematemesis.
Aneurysm may be seen in chest radiograph if large enough and present atheromatous mural calcifications.
MSCT allows definitive diagnosis.
PULMONARY VEINS CALCIFICATIONS
Described in two settings: mitral valve disease of rheumatic origin (Fig. 27) and chronic renal insufficiency (Fig. 28).
In cases of rheumatic disease is associated with atrial fibrillation,
left atrium dilatation,
dyspnea and female predominance.
In cases of chronic renal insufficiency is associated with cardiac arrhythmia.
At chest radiograph,
signs of left atrium enlargement may be seen.
MSCT may show left atrium dilatation and endocardial calcifications.
In some cases moldlike calcifications extending to the interatrial septum and distal segment of pulmonary veins.
VALSALVA SINUS CALCIFICATIONS
Aneurysms (Fig. 29): 0.09% of patients in necropsy series.
Three to four times as frequent in men as in women.
Congenital (Marfan and Ehlers-Danlos syndromes; related to other cardiac abnormalities) or acquired (infections,
atherosclerosis,
cystic medial necrosis and trauma).
Symptoms secondary to rupture or compression of surrounding structures.
Location: Right coronary sinus (72%),
non-coronary sinus (22%) and left coronary sinus (6%).
At chest radiograph,
usually undetected.
Depending on the localization of the aneurysm heart contour abnormalities may be seen.
Hemodynamic abnormalities related to the aneurysm are more frequently seen than aneurysm itself (aortic valve insufficiency or rupture).
Calcification may develop secondary to atherosclerosis.
At MSCT,
these are the diagnostic criteria: origin above aortic annulus,
saccular shape and aortic root and ascending aorta with normal caliber.
Excellent anatomic localization and detection of rupture.
DUCTUS ARTERIOSUS CALCIFICATIONS
Closure of the ductus arteriosus usually soon after birth becoming ligamentum arteriosus.
This ligament may calcify with a higher incidence in women.
Prevalence increases with age and atherosclerosis.
In chest radiograph,
focal small calcification in cases with normal closure of the ductus arteriosus.
In cases of persistence of ductus arteriosus curvilinear calcifications may be seen (Fig. 30).
MSCT allows visualization of the patent ductus arteriosus,
assess the size and morphological type and evaluate the extension of the calcifications (Fig. 31).
Calcification of the ligamentum arteriosus seen on CT examinations in healthy children reaches 13%.
POST-SURGERY CALCIFICATIONS
FALLOT SURGERY
Reparative surgery of Fallot usually performed during the first year.
It consists of closure of the ventricular septum defect and widening of the right ventricle outflow tract after partial resection of the myocardial muscle or the pulmonary valve using a patch to increase the outflow area or connecting the right ventricle with the main pulmonary artery.
Calcifications and aneurysmatic dilatation of the right ventricle outflow tract may be seen (Fig. 32).
Calcifications may also be seen in the connection between right ventricle and main pulmonary artery in patients with atherosclerosis.
SAFENOUS VENOUS GRAFT
Safenous venous grafts are used in coronary revascularization surgery.
They may become arterialized with changes including: intimal fibrous thickening,
medial hypertrophy and lipid deposits.
The most important clinical marker for safenous graft calcification is time since surgery.
Calcifications are more frequent on safenous graft wall than on atheroma plaque.
Faster deterioration than native coronary arteries with average time of arterialization since surgery of 10.5 years.
Visible in both chest radiograph and MSCT (Fig. 33).
Intravascular ultrasound has also been used in the assessment of the extent of calcification.