Purpose
Patients with stage IV A colorectal cancer still have possibilities of treatment and disease control.
This is particularly true in those subjects with resectable liver metastases or with liver metastases becoming resectable after neoadjuvant chemotherapy (ChT).
In patients with resectable liver lesions,
it has been shown that ChT improves the overall treatment results.
ChT can be carried out as a neoadjuvant or as an adjuvant treatment,
with the choice between the two is still debated.
Antiangiogenetic drugs offer new possibilities for the neoadjuvant therapy.
However,...
Methods and Materials
Patient Population
From January 2007 to October 2010 39 patients with liver-limited metastases (synchronous or metachronous to a colorectal cancer,
M1a stage) were enrolled as part of a phase II study.
These subjects underwent neoadjuvant therapy with the anti-VEGF agent bevacizumab and with the Folfiri protocol (folinic acid,
fluorouracil,
and irinotecan).
The primary purpose of the study was to determine the percentage of progression-free patients at 12 months from neoadjuvant therapy and surgery.
As an under project of the phase II study,
we meant to...
Results
Results
CEUS was always completed,
with an adequate assessment of all lesions.
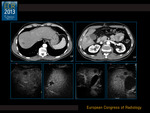
At time 0 there were 1-8 lesions (mean 3) per patient.
On a patient basis,
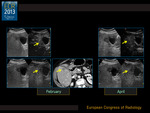
CEUS correlated with MDCT in all cases (100%) (Fig. 2).
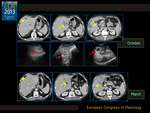
Thirty-five subjects (89.7%) were diagnosed as responders (20 completely and 15 partially) both at CEUS and MDCT,
and consequently underwent surgery.
Four patients showed disease progression and were regarded as not responders (10.3%).
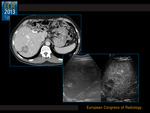
The correlation between pre-surgical imaging and post-surgical pathology is shown in Fig. 3.
Responding lesions...
Conclusion
CEUS is feasible and effective in the assessment of patients undergoing antiangiogenetic drugs-based neoadjuvant therapy for colorectal liver metastasis.
CEUS is comparable to MDCT and it may represent a valuable alternative/combination for patients monitoring.
CEUS could be considered in an alternative combination with CT,
to monitorize the neoadjuvant therapy (closer assessment?),
and/or as a problem-solver tool in the case of indeterminate findings at CT or PET or in the case of discrepancy between the CT and PET findings.
References
Anzidei M et al. Liver metastases from colorectal cancer treated with conventional and antiangiogenetic chemotherapy: evaluation with liver computed tomography perfusion and magnetic resonance diffusion-weighted imaging.
J Comput Assist Tomogr 2011;35:690-6
Bertolini F et al. FOLFOX6 and bevacizumab in non-optimally resectable liver metastases from colorectal cancer.
Br J Cancer 2011;104:1079-84
Chaudhury P et al. Perioperative chemotherapy with bevacizumab and liver resection for colorectal cancer liver metastasis.
HPB (Oxford) 2010;12:37-42
Eisenhauer EA et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1)....
Personal Information
Orlando Catalano,
MD,
Dept of Radiology I,
National Cancer Institute “Pascale Foundation”,
Naples,
Italy.
[email protected]
Antonio Nunziata,
MD,
Dept of Radiology,
P.
“Stefania Bellone”,
DSB30,
ASL Napoli 1 Centro,
Naples,
Italy.
[email protected]
Elisabetta de Lutio di Castelguidone,
MD,
Dept of Radiology I,
National Cancer Institute “Pascale Foundation”,
Naples,
Italy.
Guglielmo Nasti,
MD,
Dept of Colorectal Oncology,
National Cancer Institute
“Pascale Foundation”,
Naples,
Italy.
Fabiana Tatangelo,
MD,
Dept of Pathology,
National Cancer Institute “Pascale Foundation”,
Naples,
Italy.
Antonella Petrillo,
MD,
Dept of Radiology I,
National Cancer...