The clinical spectrum of childhood TB reflects differences in the balance between the pathogen and the host immune response,
with more severe disease resulting from either poor or ‘over-exuberant’ attempts to contain the disease.
It remains largely unknown what determines the differences in the host/pathogen interactions that leads to successful containment as opposed to progressive disease,
however age and immunodeficiency represent important factors,
strictly correlated each other.
Two different types of PTB have been described in the extremes of childhood according to differences in host immunity: infant disease and adult-type disease.
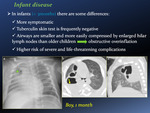
The former (typical of infants,
0-3 months) is due to poor cell mediated immunity which leads to unrestrained proliferation of bacilli with progressive parenchymal lung damage (with or without cavity formation) and dissemination [Fig. 2].
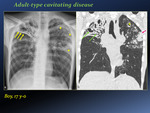
The latter,
adult-type disease,
occurs in children major than 10 years old,
representing the dominant disease manifestation following recent primary infection.
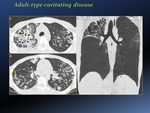
This is due to an exuberant immune response in immunocompetent adolescents which tends to result in cavitating disease [Fig. 3, Fig. 4].
The initial presentation of adult-type disease may be with cloudy opacification in apical lung zones,
before coalescence and parenchymal breakdown.
Most commonly the apical and posterior segments of the upper lobes and the apical segment of the lower lobes are affected.
Complications may result from progressive cavity formation and intra-bronchial spread with bronchopneumonic consolidation.
Apart from these particular types of PTB,
radiologic manifestations of other forms of disease affecting children with normal immune function can be categorized into the two distinct forms of primary and post-primary disease,
in individuals without and with prior exposure and acquired specific immunity,
respectively.
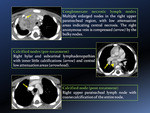
The most common form in childhood is primary,
whose hallmark is represented by lymphadenopathies [Fig. 5].

Fig. 6: TB lymph nodes features. *[3,4]
Lymphadeopathies may be accompanied by parenchymal involvement,
with age-related differences in pattern of presentation: children from 0 to 3 years old usually have higher prevalence of lymphadenopathy and lower prevalence of parenchymal involvement,
whereas children 4-15 years old present lower prevalence of lymphadenopathy and higher prevalence of parenchymal involvement [5].
Parenchymal involvement in primary pulmonary TB most commonly appears as an area of homogeneous consolidation,
though patchy,
linear,
nodular and masslike forms have also been described.
Consolidation typically occurs in a segmental or lobar distribution.
Less common findings of PTB are represented by multifocal parenchymal involvement,
pleural effusion,
obstructive atelectasis and obstructive overinflation (resulting both from compression by adjacent enlarged nodes).
Right side is more commonly affected than left side (reflecting presumably the greater statistical probability of an airborne infection involving the right lung),
without relevant differences between upper and lower pulmonary zones.
Although CXR is the first exam performed in children with respiratory symptoms and/or suspicion of PTB,
CT is the imaging of choice to diagnose the presence,
location and characteristics of mediastinal adenopathy,
to detect PBT in patients with normal CXR (higher incidence of lymphadenopathies on CT scans - 85% - compared to CXR - 70%) [6],
to identify miliary forms and eventual complications.
Finally,
CT allows the classification of TB disease severity.
CLASSIFICATION OF PTB SEVERITY
One of the obstacles in childhood TB is the lack of standard descriptive terminology to classify the diverse spectrum of disease.
Definite disease classification is important to allow an accurate evaluation of patient prognosis [7].
Conventionally PTB has been classified into pulmonary and extrapulmonary forms (which include miliary disease and TB meningitis),
without correlation with clinical disease severity.
Although no approach to comprehensively classify the observed clinical disease spectrum and severity in children has been developed yet,
Wiseman CA et al [2] have recently proposed a new classification that more accurately reflect the clinical severity of TB in children.
Clinical severity is important to determine,
as it strictly correlates to bacterial load (thus infectivity),
patient’s prognosis and treatment response.
Based on the site primarily involved,
on the extent of disease and on the presence of complications,
the different disease entities may be classified at first as intrathoracic or extrathoracic and then as either severe or non severe.
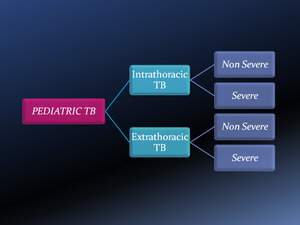
Fig. 7: Classification of PTB.
Severe disease implies either poor host control of Mycobacterium tuberculosis (uncontrolled disease) or some forms of complicated disease manifestation,
often with severe sequelae.
Non Severe disease implies that the host has managed to control the pathogen so that the disease pattern is limited (controlled),
non-disseminated and uncomplicated.
INTRATHORACIC PTB

Fig. 8: Classification of intrathoracic PTB.
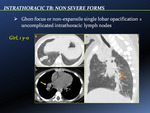
Non severe forms
- Ghon focus or non-expansile single lobar opacification,
which may be accompanied by uncomplicated intrathoracic lymph nodes (Ranke complex) [Fig. 9]
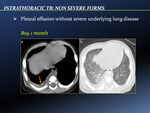
- Pleural effusion without severe underlying lung disease [Fig. 10]
Pleural effusion may be due to a hypersensitivity phenomenon after rupture of uni- or bilateral subpleural foci,
or from hematogenous spread.
It is classified as non severe if there is no evidence of severe underlying lung disease. Large effusions in young children are uncommon,
usually occurring in adolescence.
Pleural effusions complicate 2–38% of cases of pulmonary TB in children,
usually unilaterally [8].
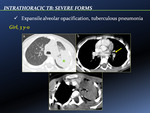
Severe forms
- Expansile alveolar opacification,
tuberculous bronchopneumonia [Fig. 11]
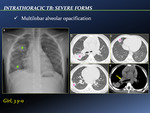
- Multilobar alveolar opacification [Fig. 12]
Cavitation in single or multiple sites is evident radiographically in 40%–45% of cases of postprimary TB.
Walls of cavities may range from thin and smooth to thick and nodular; air-fluid levels have been reported to occur in 9%–21% of tuberculous cavities [5].
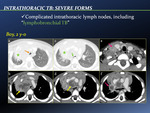
- Complicated intrathoracic lymph nodes,
including “lymphobronchial TB” [Fig. 15]
The wide spectrum of endobronchial or “lymphobronchial” TB includes different entities: extrinsic bronchial compression,
actively caseating lesion in the tracheobronchial tree,
granuloma formation,
polypoid mass lesions and lymph node protrusion through mucosal erosion with ulceration.
Although these lesions are observed during bronchoscopy,
large airway compression can often be suspected based on CXR findings.
CT scans may show irregular or smooth circumferential bronchial narrowing associated with mural thickening [Fig. 4-d],
airway compression by lymphadenopathies,
polypoid masses on the wall of the tracheobronchial tree and also lymph nodes into the bronchial lumen.
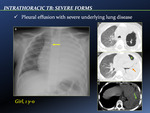
- Pleural effusion with severe underlying lung disease [Fig. 16]
Occasionally,
an air-fluid level may be also demonstrable within the pleural cavity,
indicating the presence of a broncho-pleural fistula.
Contrast-enhanced CT evaluation of post-primary tuberculous effusions typically reveals smooth thickening of visceral and parietal pleural surfaces separated by a variable amount of fluid: the ‘‘split pleura’’ sign [5].
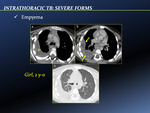
Tuberculous effusions are typically loculated and may be stable in size for years; detection of persistent fluid within a calcified fibrothorax at CT should raise concern for active disease and chronic tuberculous empyema.
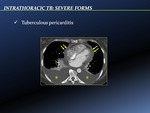
Usually develops when a subcarinal lymph node erupts into the pericardial space.
However,
it is also the result of hematogenous bacillary spread.
If unrecognized,
it may have severe sequelae,
including death,
due to its mechanical effects.
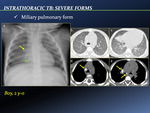
Innumerable,
1–3-mm non-calcified nodules scattered throughout both lungs,
with mild basilar predominance,
often associated with intra and interlobular septal thickening.
Typical miliary lesions may not be visible until 3–6 weeks after hematogenous dissemination.
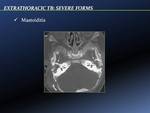
EXTRATHORACIC PTB

Fig. 20: Classification of extrathoracic PTB in children.
Non severe forms
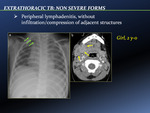
- Peripheral lymphadenitis,
without infiltration/compression of adjacent structures [Fig. 21]
No evidence of contiguous spread and infiltration or compression of adjacent neural,
vascular,
lymphatic duct,
or osteal tissues.
Isolated sinus or fistula formation from a diseased node,
although reflecting uncontrolled disease,
is classified as non severe,
as its complications do not usually result in any functional deficits.
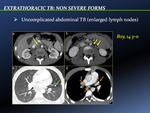
- Uncomplicated abdominal TB [Fig. 22]
It is thought to be due to retrograde lymphatic spread from a pulmonary focus,
hematogenous spread,
or ingestion of organisms during primary infection or disease.
- Otitis media (if not complicated by deafness)
- Hypersensitivity forms of arthritis,
synovitis and osteitis in isolation
Severe forms
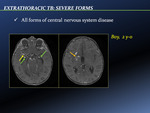
- All forms of central nervous system disease [Fig. 23]
They are represented by tuberculous meningitis,
vasculitis,
granulomata and brain abscess,
reflecting hematogenous organism dissemination.
- Complicated forms of abdominal TB
These forms include enteritis,
solid organ disease,
and peritoneal spread,
which reflect either bowel wall infiltration from ingested organisms at primary infection,
hematogenous organism dissemination,
or rupture of abdominal nodes into the peritoneal cavity.
It may occur from hematogenous dissemination or contiguous spread from uncontrolled otitis media.
It is assumed to arise from hematogenous spread of bacilli.
- Spondylitis,
arthritis,
synovitis and osteitis
They are the result of hematogenous TB dissemination.
This classification seems to correlate quite directly with clinical disease severity,
allowing a more rapid prognostic evaluation.
TREATMENT RESPONSE
The aim of anti-TB treatment in children is to cure the patient of TB,
reduce spread to others and avoid the development of drug resistance within the community.
Although national recommendations still vary considerably in treatment duration and drug regimens used,
TB treatment consists generally of two phases: an intensive phase,
using a combination of bactericidal drugs to kill the rapidly growing bacilli,
and a continuation phase,
using fewer drugs to eradicate the slower growing persistent bacilli [9].
Owing to the lack of bacteriologic confirmation in most cases,
radiologic and clinical evaluation become the major indicators of response to anti-TB therapy in children.
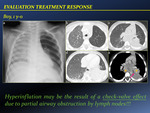
Regression of radiologic abnormalities is a slow process,
involving in parallel parenchymal consolidations and lymphadenopathies [Fig. 25, Fig. 26].
Resolution of parenchymal abnormalities may require from 6 months to 1 year.
Lymphadenopathy may persist for several years after treatment.
During the first 3 months of therapy,
worsening of radiologic signs (i.e.
extension of parenchymal involvement,
development or enlargement of existing nodes) may be observed in up to 1/3 of patients.
It has been attributed to an hypersensitivity reaction which normally occurs 2-10 weeks after initial infection.
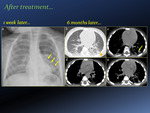
After treatment,
lymph nodes in regression usually appear discrete,
reduced in size,
homogeneous,
with visible surrounding perinodal fat and sometimes with calcifications [Fig. 5].
However,
residual sizable nodes after 6 months of therapy do not necessarily indicate disease activity: it’s the clinical scenario along with the trend of regression of nodes which suggest if the disease is active or not [10].

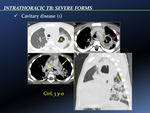
![Fig. 14: CXR anteroposterior view (e-f) 1 week after CT scan [Fig. 13] shows reduction of consolidation and cavitation of LUL (arrow), even more reduced 1 month later.](https://epos.myesr.org/posterimage/esr/ecr2013/115121/media/490977?maxheight=150&maxwidth=150)