This poster is published under an
open license. Please read the
disclaimer for further details.
Type:
Educational Exhibit
Keywords:
Extremities, Genital / Reproductive system female, Musculoskeletal soft tissue, MR, Ultrasound, Diagnostic procedure, Removal, Treatment effects, Foreign bodies
Authors:
X. Tomás1, A. Rodriguez-Veret2, S. Orlando3, E. Guisantes2, C. Yanguas4, J. C. Vilanova5, S. Baleato González6; 1Barcelona/ES, 2Terrassa/ES, 3Sant Cugat del Vallès/ES, 4Manresa/ES, 5Girona/ES, 6Santiago de Compostela/ES
DOI:
10.1594/ecr2015/C-1244
Background
In 1983,
the first subdermal contraceptive implant,
Norplant®,
was approved by the Finnish national drug regulatory authority.
Since then,
several more implants such Jadelle® (Norplant II) and Implanon®,
the most commonly used in Spain,
have been registered.
A large body of evidence demonstrates the high contraceptive effectiveness and safety of the 5-year levonorgestrel releasing implants Norplant® and Jadelle® (1).
Implanon® is a nonbiodegradable,
progestogen only single-rod based on an ethylene vinylacetate carrier of desogestrel metabolite etonogestrel (ENG) which is released for a duration of at least 3 years.
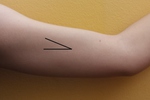
Subdermal implants should ideally be inserted into the subdermal medial aspect of the nondominant upper arm,
6-8 cm above the elbow (2) (Figure 1).
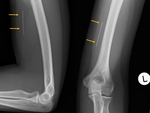
Norplant® is a six-capsule preparation easily visualized with X-Ray (3),
Jadelle® is a two-rod preparation,
with a size of 43 x 2.5 mm.
whereas newer Implanon® is a single-rod preparation with a size of 40 x 2 mm.,
visualized with X-Ray since 2013 (4) (Figure 2),
easier to insert and remove from subdermal target than Norplant® (2,
5-8).
Imaging techniques such as digital fluoroscopy have been used to remove subdermal implants in the past (9),
but ultrasound is considered the “gold-standard” imaging technique to assess the localization for the removal of subdermal nonpalpable implants (10).
An implant located just under the skin,
under the fascia muscularis or one located deep in the muscle can most often be localized with ultrasound,
with a characteristic pattern of an echogenic focus producing a posterior acoustic shadow.
Specific settings of the ultrasound machine,
such as a high-resolution linear array transducer (10-15 MHz),
cancelling of all image improvement processing (multiple focusing,
image compounding,
adaptive post-processing) or positioning of the focus in the near field are mandatory (11).
In a previous study from Singh M et al,
positive identification of the implants was achieved in 21 of 23 cases using ultrasound.
All 21 Implanon® rods identified by ultrasound were successfully removed.
No implant was detected in two cases and etonogestrel was not demonstrated serologically in either woman,
suggesting non-insertion (12).
Perhaps in both patients an MRI might have been an additional option to exclude insertion of nonfunctional rods.
Measurements of serum etonogestrel levels may be necessary to confirm the presence or absence of the implant when it cannot be visualized by either of the two imaging methods (10).
In those patients and in other rare cases,
subdermal implants could not be localized due to previous scars,
major obesity,
deep implantation or migration.
Under these circumstances,
MRI might have been considered the best imaging technique for the unequivocal localization of nonpalpable,
ultrasonographically not detectable implant rods (10,
13).
In another study,
Westerway SC et al published that MRI imaging requires caution when differentiating blood vessels and fibrous septae from the Implanon® rods (14).
We report the MRI findings of two patients carrying nonpalpable Jadelle® subdermal implants. To the best of our knowledge,
this is the first study which describes those findings.