Multi-parametric MRI (mpMRI) represents a breakthrough in the diagnosis and management of prostate cancer with a sensitivity and specificity dramatically superior to TRUS [1-2-3].
Notwithstanding the accuracy of mpMRI,
prostate biopsy is still considered the gold standard for the histological characterization and clinical management of prostate cancer.
Currently,
systematic TRUS guided biopsy is accepted as the technique of choice as it is friendly,
well-tolerated and inexpensive.
As it consists of a randomized sampling,
the major limit is represented by the possibility of missing clinically significant tumors especially located in “difficult” prostatic areas and,
on the other hand,
the risk of overdiagnosis of clinically insignificant tumors or precancerous lesions [1].
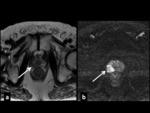
The introduction of mpMRI with its outstanding sensitivity and specificity (Fig 1) has introduced the necessity of performing targeted biopsies on areas with a pathological signal.
In fact,
there are increasing evidences that targeted biopsies have a better cancer detection rate than systematic biopsies alone [4-5-6].
Targeted biopsies can be carried out in three different ways
a) MRI guided in bore biopsy;
b) TRUS guided biopsy with cognitive approach;
c) TRUS guided biopsy with TRUS-mpMRI fusion.
At first sight,
MRI guided biopsy could appear the best solution,
but we need to consider many disadvantages and limits.
The procedure is expensive,
time consuming,
less tolerated and is performed mainly with a transperineal approach that is more painful and requires extensive local anesthesia or sedation [1-7].
Furthermore,
the procedure cannot be performed in a true real-time modality.
TRUS-guided biopsy with cognitive approach is intuitive,
inexpensive and preserve all the advantages of TRUS guided biopsy but is susceptible to human errors [8].
TRUS-guided biopsy with TRUS-mpMRI fusion is the most promising as it combines all the advantages of TRUS guided biopsy,
included the possibility of systematic sampling when necessary,
together with the sensitivity of mpMRI.
Virtual Navigator by image fusion techniques (also known as interactive localizing techniques) has been proposed in the last two decades in order to spatially coregistrate a real-time modality (i.e.
US) with high resolution isotropic 3D CT and MRI or functional images (i.e.
PET-CT).
In this way the operator is allowed to use,
at the same time,
the information from multiple imaging modalities which enhances diagnostic and localization capability.
Image fusion can be achieved by Electro-Magnetic (EM),
optical or mechanical devices.
There are some advantages of EM device over optical and mechanical device:
a) The tracked device can reside out of generator sight;
b) Multiple devices can be tracked at the same time;
c) Devices can be tracked also within the patient’s body without signal attenuation;
d) There are no significant limitation to operator position and movements.
These advantages make EM tracking systems flexible and suited for multipurpose applications.
The main technical limitation of EM tracking systems is that the ferromagnetic environment can cause interference and distortion of magnetic coordinates.
However,
a new generation of EM tracking systems has reduced susceptibility to the effects of the metal hardware [9-10-11].
Other possible drawbacks are the necessity of expensive dedicated image-fusion hardware and software,
and the difficulty of obtaining a precise image fusion considering the anatomical distortion that can be induced by physiological changes or by the TRUS probe itself.
The aim of our study is to evaluate the feasibility of TRUS guided biopsy with TRUS-mpMRI fusion using an ultrasound scanner equipped with a multipurpose Electro-Magnetic (EM) tracking system.