Keywords:
Interventional vascular, Emergency, Catheter arteriography, Fluoroscopy, Embolisation, Haemorrhage
Authors:
F. Giurazza, F. Corvino, G. Cangiano, E. Cavaglià, F. Amodio, R. Niola; Naples/IT
DOI:
10.1594/ecr2018/C-0158
Methods and materials
In the last two years,
ten patients have been treated for arterial embolization using MVP: 6 males and 4 women,
and mean age 55.1 years (range 20–80 years).
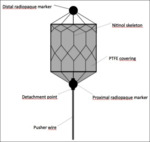
The MVP is a self-expanding device composed of a detachable ovoid-shaped embolic plug; the skeleton is made of Nitinol and it is covered with a Polytetrafluoroethylene (PTFE) coating (Fig.
1).
At moment,
three sizes are available (Table 1): 3,
5 and 7; MVP3 and MVP5 are released through a 2.7 Fr microcath while MVP7 is released through a 4 Fr catheter.
All MVPs present two radiopaque markers at proximal and distal extremities.
After acquiring a thin slice (0.65mm) contrast enhanced Computed Tomography (CECT) to plan the embolization, a diagnostic digital subtraction angiography (DSA) confirmed the site to embolize and the size of the vessel to occlude.
The catheter tip was positioned in correspondence of the target and the MVP was unsheathed using a pull-back technique,
retiring the catheter while gently advancing with the pusher wire; once the proximal marker exits from the catheter tip and appeared stable,
it was detached with anticlockwise torque.
The MVP was employed even in cases when other embolizing agents failed to properly occlude an arterial vessel.