A-Vascular complications
Occur in less than 10% of kidney transplantations but remain a major cause of graft dysfunction [1].
Although angiography is the gold standard technique to evaluate these complications,
Doppler-ultrasound (US) offers an excellent noninvasive modality whether in the early or late postoperative period.
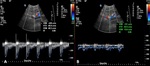
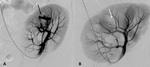
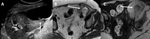
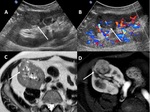
A-1-Renal artery stenosis Fig. 2 Fig. 3 :
-Reported in up to 10% of kidney transplantations [2].
-Occurs in the first year following transplantation [3].
-Alerting symptoms are severe hypertension refractory to medical treatment,
audible bruit over the graft or unexplained graft dysfunction [4].
-Half of the arterial stenosis are located at the anastomosis site,
with end-to-end anastomosis having a threefold greater risk of stenosis than the end-to-side anastomosis [5].
-Doppler US will show focal color aliasing at the stenotic site,
which can be graded and characterized by the duplex technique.
-Doppler criteria for significant stenosis include [5,
6]:
- Velocities greater than 2m/s
- Velocity gradient of more than 2:1 between prestenotic and stenotic segments
- Marked distal flow disturbance
- Parvus tardus waveforms in distal renal graft artery
-Magnetic resonance angiography (MRA) is a good alternative technique and can be perform with or without intravenous gadolinium chelate injection [3].
-Treatment of choice is percutaneous transluminal angioplasty with or without stent placement [7].
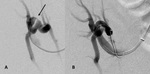
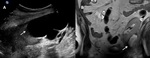
A-2-Arterial thrombosis and renal infarction Fig. 4 :
-Rare complication occurring in the early postoperative period and generally leads to graft loss [8].
-May be due to hyperacute rejection,
anastomotic occlusion,
arterial kinking or flap [3].
-Infarcts in the renal transplant may be focal or diffuse,
as a result of rejection or vascular thrombosis [3].
-Patients will present with anuria and eventually swelling and tenderness over the graft [3].
Note that the graft itself is denervated but pain can result from peri-renal or visceral peritoneum inflammation.
-In case of renal infarct,
US will show a poorly marginated hypoechoic focus,
with no color flow on Doppler examination that can also be seen in severe pyelonephritis.
-In case of complete vascular obstruction,
absent perfusion of the whole graft will be noted at US-Doppler examination.
-If the diagnosis is uncertain,
angiography or MRA may be performed.
-Early diagnosis is mandatory to preserve graft viability.
Percutaneous angiographic interventional techniques are usually the first line treatment.
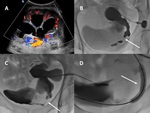
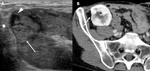
A-3-Renal vein stenosis and thrombosis Fig. 5 :
-Uncommon complication occurring in less than 5% of renal transplant.
-The renal vein thrombosis occurs usually in the first postoperative week [3].
-Patients with renal vein thrombosis will present with anuria,
swelling and tenderness over the graft,
and renal dysfunction.
-US of renal vein stenosis will show similar pattern than those for arterial stenosis,
including Doppler-color aliasing and high velocities (threefold or fourfold increase in velocity is usually considered as a significant stenosis) [9].
-US of renal vein thrombosis will show an enlarged kidney,
reduced or absent venous flow and increased resistance of the arterial flow with reversed diastolic flow [3].
-Early recognition of this condition may bring to prompt thrombectomy,
although graft infarction is frequent and will end by transplant nephrectomy in order to prevent secondary infection [9].
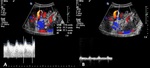
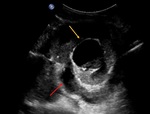
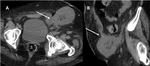
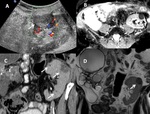
A-4-Intrarenal arteriovenous fistulas and pseudoaneurysms Fig. 6 Fig. 7 :
-Occur as a complication of percutaneous renal biopsy.
-The majority of these lesions are small and clinically insignificant,
resolving spontaneously; therefore,
the exact frequency of this complication is unknown [10].
-When large,
they may result in renal ischemia,
hematuria or peri-renal hematoma (in case of rupture).
-At US-Doppler,
arteriovenous fistulas will present as focal disorganized flow around the normal renal vasculature.
The feeding artery will show a high-velocity and low-resistance waveform,
whereas the draining vein will display a pulsatile and arterialized flow [11].
-Pseudoaneurysms mimic renal cysts on gray-scale US images but with evidence of a vascular flow inside it.
Those with a narrow neck and no venous communication will show a “to-and-fro” Doppler waveform at their neck site while the ones with a broad necks may show a high-velocity and low-resistance waveform.
-Most of these complications will resolve spontaneously and are treated conservatively.
However,
when a progressive enlargement or an unusual size (larger than 2 cm in diameter) is noticed,
treatment is warranted using selective transvascular catheterization.
B-Non-vascular complications
B-1-Urologic complication (4%-8%) [12,
13] :
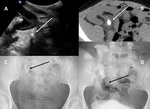
B-1-a-Urine leaks and urinomas Fig. 8 :
-Extravasation of urine due to ureteral (or caliciel) necrosis caused by vascular insufficiency or increased urinary pressure (obstruction).
-Usually occur in the first two weeks after the surgery [3].
-Urine leak may also be due to post-instrumentation complication or trauma.
-Urine extravasation may be encapsulated,
forming a urinoma.
-Urinoma may initially be clinically occult with delayed complication such as hydronephrosis,
paralytic ileus,
abscess or electrolyte imbalances.
-US will show evidence of a well-defined anechoic fluid collection with no septations (thinned wall for urinoma) that increases in size rapidly.
The collection is usually contouring any portion of the renal tracts.
-Small urinomas are usually treated conservatively.
For larger,
persistent or symptomatic urinomas,
imaging-guided treatment can be managed with a combination of percutaneous drainage catheter,
percutaneous nephrostomy catheter,
ureteral stent,
and bladder drainage.
-Antegrade pyelography is necessary to provide detailed information about the origin site of the urinoma [14].
B-1-b-Urinary obstruction Fig. 9 Fig. 10 :
-Occurs in about 2% of transplantations,
always within the first 6 months after the procedure [3].
-The narrowing is usually at the ureterovesical junction and may be caused by scaring secondary to ischemia or rejection,
by technical issues during ureteroneocystostomy or by kinking.
The patient does not feel the typical renal colic pain because the transplanted kidney is denervated.
-US must be performed with an empty bladder to search for (uretero) hydronephrosis.
A distended urinary bladder may cause dilatation of the pelvicalyceal system,
wrongly interpreted as hydronephrosis.
-Hydronephrosis remains a nonspecific finding because it is also seen in cases of diminished ureteral tone from denervation of the transplant [11].
-US may also demonstrates direct causes of urinary obstruction as calculi,
or indirect causes as lymphoceles,
hematomas,
abscesses,
and urinomas.
Any echogenicity within a dilated collecting system is usually clinically significant [15,
16].
-Treatment of urinary obstruction consists of stent placement,
balloon dilatation,
or correction of extrinsic compression of the collecting system.
Surgical reconstruction may be required for long or recurrent strictures.
B-2-Peritransplant fluid collections:
-Occur in up to 50% of renal transplantations.
-Urinomas and hematomas develop usually immediately after the surgery whereas lymphoceles are most likely to appear 4-8 weeks after [3].
B-2-a-Hematomas Fig. 11 :
-Blood collections that are common in the immediate post-operative period but may also develop as a consequence of trauma or biopsy.
-Usually small and resolve spontaneously.
-On US,
acute hematomas are echogenic and become less echogenic with time.
Older hematomas even appear anechoic and septations may develop.
-On computed tomography (CT),
acute hematoma presents high-attenuation components and older ones contain liquefied and serous portions of intermediate attenuation.
-Immediate post-operative small hematomas are not worrisome [11].
-Size,
location and growth should be documented on the baseline US.
B-2-b-Lymphoceles Fig. 12 :
-Caused by lymph leakage from surgically disrupted lymphatic channels along the iliac vessels or from the lymphatics of the graft.
-US will show an anechoic collection with or without septations.
-Hydronephrosis can eventually be caused by extrinsic compression from the lymphocele.
-CT-scan will usually show a sharply circumscribed collection with attenuation values identical to those of water.
-Most lymphoceles are discovered incidentally,
are asymptomatic and do not require therapy.
-In case of complications,
they can be treated by percutaneous or surgical technique (i.e.
aspiration or placement of a drain ± sclerotherapy).
B-3-Infection and abscesses Fig. 13 :
-Infection will affect more than 80% of renal transplant recipients within the first year after the transplantation [3].
-Early diagnosis is mandatory to prevent loss of graft function.
-Pyelonephritis may be caused by bacterial seeding from a lymphocele,
a hematoma,
or a urinoma.
-Any complex collection identified in the late postoperative period with clinical evidence of infection may represent an abscess.
-On US,
focal pyelonephritis may appear as focal areas of increased or decreased parenchymal echogenicity with bulging of the renal capsule and hyperechoic infiltration of the peri-renal fat.
-Abscesses will show non-specific appearance at US (hypoechoic/heterogeneous),
depending on their content.
-On CT-scan,
gas within the collection is an evidence of abscess formation.
-Abscess are generally treated by image guided percutaneous drainage.
B-4-Gastrointestinal/herniation complications Fig. 14 :
-The most common gastrointestinal complication in the transplanted patient is hemorrhage due to peptic ulcer [17].
-Intestinal obstruction and urinary obstruction may be caused by postoperative adhesion or herniation of bowel or ureter through a post-surgical peritoneal defect [18].
-The prevalence of these complications varies depending on the surgical approach (intra or extra peritoneal) [19].
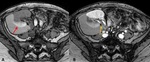
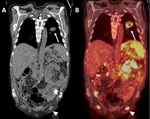
B-5-Neoplasms Fig. 15 Fig. 16 :
-Kidney transplant recipients have up to 100 times the normal risk for developing cancer,
due to prolonged immunosuppression.
-The average reported prevalence of malignancy in transplanted patients is 6% [20].
-Prevalence of renal adenocarcinoma may be increased,
with 90% of these tumors occurring in the native kidneys and 10% in the transplanted one [21].
-A solid heterogeneous renal mass will be found on US or CT examinations.
-Malignancy in the transplanted kidney is treated by total removal of the graft and immunosuppression cessation.
B-6-Post-transplantation lymphoproliferative disorder (PTLD) Fig. 17 :
-PTLD develops in 8% of transplantations.
-Caused by impaired immune surveillance and immunosuppressive therapy.
-Range from low-grade/benign lymphoid hyperplasia to high grade malignant non-Hodgkin lymphoma.
-The liver,
brain and lungs are involved more frequently than the gastrointestinal tract [3].
B-7-Renal failure:
B-7-a-Acute tubular necrosis:
-Defined as the need for dialysis in the first week following transplantation [22].
-Due to prolonged ischemia and reperfusion injury.
-Occurs in most cadaveric grafts and resolves spontaneously over the first 2 weeks,
depending on the degree of ischemia.
B-7-b-Rejection:
-Fifty percent of transplanted patients develop one episode of acute rejection within the first year.
-Chronic rejection is the most common cause of late graft loss [22].
B-7-c-Drug nephrotoxicity:
-Many drugs are potentially nephrotoxic and their effects are increased by dehydration and decreased renal perfusion with cyclosporine having the greatest nephrotoxic potential.
-US findings are nonspecific,
showing renal enlargement,
increased cortical thickness,
loss of corticomedullary differentiation,
prominent pyramid,
collecting system thickening and loss of central sinus echo complex.
-The final diagnosis is made by histologic analysis.