Clinical findings:
Initial diagnosis of GTD is based on a combination of history,
examination,
quantitative β-hCG titers,
and pelvic sonography.
Diagnosis of hydatiform mole based on clinical features may be difficult due to non-specificity of signs and symptoms.
Patients present with irregular vaginal bleeding,
excessive vomiting,
transvaginal expulsion of grape-like vesicles,
abnormally enlarged uterus,
and features of preeclampsia,
anemia,
or hyperthyroidism. β-hCG levels show large variation in normal,
multiple,
and abnormal gestations and when considered in isolation may be misleading for diagnosis of hydatiform mole.
Hence,
early first trimester sonography remains the investigation of choice for initial diagnosis of hydatiform mole (3,4).
The standard treatment for hydatiform form is suction evacuation,
resulting in approximately 84% cure rate for complete moles and 99.5% for partial moles.
It is difficult to predict patient outcome at the time of initial diagnosis.
Hence,
all patients are followed up with serial quantitative serum β-hCG measurements following suction evacuation,
to allow early diagnosis of persistent trophoblastic neoplasia. β-hCG levels should decline following suction evacuation.
As per the latest FIGO guidelines,
post-hydatiform mole GTN may be diagnosed based on any of the following criteria:(1) β-hCG level plateau for 4 measurements over a period of 3 weeks or longer,
that is,
for days 1,7,14,
and 21.
(2) A rise in β-hCG levels for 3 consecutive measurements or longer over a period of at least 2 weeks or more,
that is,
on days 1,
7,
and 14.
(3) Histological diagnosis of Choriocarcinoma. (4) Elevated β-hCG levels for 6 months or more after evacuation.
Although β-hCG is useful for diagnosis of PTN,
imaging studies may play a confirmatory role in early disease or patients with confusing clinical picture.
The most important role of ultrasound in patients with suspected PTN is to exclude a normal gestation as a cause of elevated β-hCG level.
Non-metastatic GTN presents as localized uterine tumor with abnormal vaginal bleeding.
Hematogenous dissemination is the principal route of spread,
most commonly to lung (80%) followed by vagina (30%),
brain (10%),
and liver (10%).
It may also metastasize to kidney,
gastrointestinal tract,
skin,
or fetus (from Choriocarcinoma).
Isolated metastasis to other sites is rare in the absence of lung and vaginal metastases.
Hypervascular metastases may present with features of hemorrhage.
Lung metastases may be asymptomatic or present with hemoptysis,
dyspnea,
chest pain,
or pulmonary artery hypertension.
Vaginal metastases may result in torrential vaginal bleeding.
Hence,
a biopsy should be avoided.
Brain metastases may present with headache,
seizures,
motor,
or sensory deficit.
Liver metastases are usually asymptomatic (1).
Procedure details:
1.
Ultrasound
Grey-scale ultrasound with color and spectral Doppler imaging is a very useful tool for diagnosing GTD.
Ultrasound can be done by transabdominal or transvaginal approach.
Transvaginal sonography provides better details of the lesion due to its superior spatial resolution and proximity to the anatomy of interest.
On the contrary,
transabdominal approach needs full bladder resulting in patient discomfort and provides fewer details.
Determining presence of invasive disease,
predicting response to chemotherapy,
post-chemotherapy follow-up,
and detection of recurrence (2,6).
1.1. Ultrasonographic Features of Hydatiform Mole
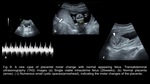
On first trimester ultrasound,
hydatiform mole which constitutes 80% cases of GTD will be seen most frequently as an enlarged uterus with a heterogeneous endometrial mass of variable echogenicity (predominantly echogenic).
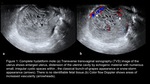
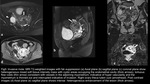
Ultrasound appearance of the lesion has been classically described earlier as “snow-storm”,
“bunch of grapes” or “granular” appearance due to multiple echogenic foci (figure1).
A fetus or fetal parts are not seen in a complete hydatiform mole except in 1-2% of cases with coexistent dizygotic diploid twin pregnancy (1).
In contrast,
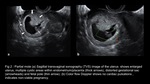
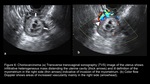
partial mole is usually associated with a fetus which is growth retarded or anomalous and an enlarged,
thickened placenta with numerous anechoic cystic lesions (figure2).
Partial molar pregnancy with coexisting normal living fetus is a rare complication with the incidence of 0.005%–0.01% of all the pregnancies.
US findings include a greatly enlarged placenta relative to the size of the uterine cavity associated with cystic spaces “molar placenta” (figure9) (13).
Differentiation of the complete and partial moles can be difficult but is of limited clinical significance,
as the management is similar.
Although ultrasound is very useful for suggesting a molar pregnancy,
final diagnosis still rests with the pathology (5).
1.2.
Ultrasonographic Features of Invasive Disease (GTN)
Myometrial invasion is best appreciated on TVS due to superior demonstration of the interface between trophoblastic tissue and myometrium.
Invasive mole,
Choriocarcinoma,
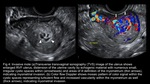
and PSTT are seen on grey-scale ultrasound as nonspecific focal masses with myometrial epicenter and are sonographically indistinguishable from one another.
The mass may be echogenic,
hypoechoic,
complex,
or multicystic.
It may show anechoic spaces which represent hemorrhage,
necrosis,
cysts,
or vascular spaces (figure4,6).
More extensive disease may appear as a heterogeneously enlarged uterus with lobulated contour or large pelvic mass which may extend to involve other pelvic organs.
These masses may be potentially confused with fibroids or adenomyosis.
Adenomyosis typically appears as a diffuse disease process causing enlarged uterus with a diffuse heterogeneous echo texture and indistinct endometrial-myometrial junction.
Typical fibroid usually presents as a well-circumscribed hypoechoic myometrial lesion,
although echogenicity may vary.
The various ultrasound presentations of gestational trophoblastic neoplasia may overlap with these imaging findings of fibroids and adenomyosis.
However,
correlation with serum β-hCG levels,
clinical history,
and lack of extreme vascularity on Doppler sonography aid in their differentiation (1).
Persistent trophoblastic neoplasia is presumed to be invasive mole unless there is presence of metastases to suggest Choriocarcinoma.
Since both invasive mole and Choriocarcinoma are usually treated with chemotherapy,
histological differentiation of the two types is not needed routinely (2).
2.
Magnetic Resonance Imaging (MRI)
MRI appearance of molar pregnancy can be relatively nonspecific and difficult to distinguish from retained products of conception or ectopic pregnancy (7,8).
First trimester MRI may reveal little or no abnormality.
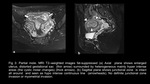
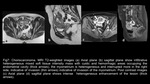
In the second trimester,
hydatiform mole is visualized as a heterogeneously hyper intense tumor distorting the normal zonal architecture on T2-weighted images which may show numerous characteristic cystic spaces.
It causes uterine enlargement and distension of the endometrial cavity with indistinct boundary between the endometrium and myometrium,
in partial mole gestational sac is seen (figure3).
On T1-weighted images,
it is isointense or mildly hyper intense to the myometrium with areas of hemorrhage,
seen as focal signal hyper intensity (7,8).
Diffuse myometrial involvement by the tumor is seen as diffuse myometrial signal hyper intensity with obliteration of the normal zonal anatomy.
Invasive GTN has similar signal characteristics however with a myometrial epicenter,
invasion into parametrium,
and more frequent hemorrhage and necrosis (figure5) (8).
MRI is superior to ultrasound for identification of parametrial invasion,
which is seen as heterogeneous T2 hyper intense masses beyond the confines of the uterus (2,8).
Choriocarcinoma in MR appears as heterogeneous,
Hypervascular mass that distort the normal zonal anatomy; extra uterine extension seen as abnormal signal intensity in myometrium or parametrium (figure7).
On contrast enhanced dynamic MRI,
viable trophoblastic tissue with surrounding inflammatory response is seen as marked enhancement on early arterial phase (figure5) (9).
Tumor hyper vascularity is also seen as prominent tortuous flow voids in the tumor and adjoining myometrium,
parametrium,
and adnexa on both T1 and T2-weighted images with engorgement of internal iliac and arcuate vessels with respect to the external iliac artery (10).
However,
the hypo vascular form of PSTT has been demonstrated to be hyper intense to myometrium on both T1- and T2-weighted images with absence of prominent vascularity or flow voids and poor enhancement on post contrast images (10).
With successful treatment,
uterine volume and tumor hyper vascularity decrease with restoration of normal zonal anatomy on T2-weighted images and reduction of heterogeneous appearance (11).
Intra-lesional hemorrhage may occur.
MRI findings usually normalize at 6–9 months following effective chemotherapy.
Persistent uterine vascular malformations are seen as residual tortuous and coiled vessels within a thickened myometrium (12).
3.
Metastatic Work-Up
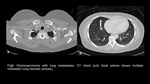
CT scan is more sensitive than chest radiograph for diagnosing lung metastases which appear as multiple,
rounded,
soft tissue density lesions,
usually up to 3 cm in size (figure8).
The lesions may be solitary,
miliary and may rarely show cavitation.
MRI or CT done to exclude brain metastases,
brain metastases are usually multiple,
located at grey-white matter junction,
and associated with hemorrhage and surrounding edema.
Due to hemorrhage,
the lesions appear hyper dense on unenhanced CT and show variable signal intensity on MRI depending on the age of the intra-lesional hemorrhage.
Vaginal disease usually results from contiguous spread from the uterine lesion.
Vaginal metastases are usually evaluated with MRI which demonstrates T2 hyper intense vaginal wall lesion with indistinct margins.
Ultrasound screening of the liver may be done at the time of pelvic assessment.
Liver metastases are typically seen as multiple Hypervascular lesions on both CT and MRI,
showing avid contrast enhancement in the arterial phase and sometimes hemorrhagic transformation (11,12).