A prospective longitudinal study was carried out from September 2012 to February 2016 in which 60 consecutive recipients of liver transplant for any etiology were included in our hospital.
Of these,
we selected the 15 patients who presented reinfection of the graft by HCV during the first year after the transplant and who received treatment with the new antivirals.
The hospital ethics committee considered that the approaches were correct and gave its approval to do so.
The patients were informed and gave their consent.
The recurrence of post-transplant HCV infection was defined by detection of viremia (HCV RNA) in peripheral blood by polymerase chain reaction technique.
The recurrence of HCV disease on the graft was defined by biochemical criteria (elevation of transaminases,
bilirubin and cholestasis enzymes not attributable to other causes) and histological criteria in 11 of the 15 patients.
One of the patients was excluded from the analysis because during follow-up he had a serious complication (Budd-Chiari syndrome due to thrombosis of the hepatic veins).
Table 1 shows a chronogram showing when the examinations,
the biopsies and the elastography measurements were performed,
the timing of the antiviral treatment,
and the therapeutic regimen.
| Patient |
Recurrence |
Biopsy |
Start AV |
Completed AV |
E1 |
E2 |
Antiviral Theraphy |
| 1 |
4 |
4 |
13 |
19 |
12 |
21 |
Sofobuvir+ribavirin |
| 2 |
1 |
1 |
3 |
9 |
3 |
10 |
Sofobuvir+ribavirin |
| 3 |
5 |
6 |
24 |
30 |
23 |
31 |
Sofosbuvir+ledipasvir |
| 4 |
1 |
NA |
18 |
24 |
16 |
26 |
Sofosbuvir+ledipasvir |
| 5 |
1 |
1 |
1 |
7 |
6 |
8 |
Sofosbuvir+ledipasvir |
| 6 |
7 |
8 |
12 |
18 |
12 |
19 |
Sofosbuvir+simeprevir |
| 7 |
6 |
6 |
12 |
18 |
12 |
21 |
Sofosbuvir+simeprevir |
| 8 |
3 |
3 |
4 |
10 |
4 |
11 |
Sofosbuvir+daclatasvir |
| 9 |
12 |
NA |
13 |
19 |
12 |
19 |
Sofosbuvir+ledipasvir |
| 10 |
2 |
2 |
9 |
15 |
9 |
18 |
Sofosbuvir+daclatasvir |
| 11 |
2 |
2 |
9 |
15 |
9 |
17 |
Sofosbuvir+daclatasvir |
| 12 |
12 |
NA |
13 |
19 |
12 |
20 |
Sofosbuvir+ledipasvir |
| 13 |
10 |
NA |
12 |
18 |
12 |
18 |
Sofosbuvir+ledipasvir |
| 14 |
1 |
1 |
2 |
8 |
1 |
8 |
Sofosbuvir+daclatasvir |
Table 1 Chronology of the study in months with respect to the transplant date (time 0).
For example: patient 1 had a recurrence at 4 months,
a biopsy was performed at 4 months,
antiviral treatment (AV) was started at 13 months and it was completed at 19 months; the pretreatment elastography (E1) was performed at 12 months and the aftercare (E2) at 21 months.
NA: Not available.
In some patients there was a delay of several months between the diagnosis and the administration of antiviral treatment; the cause was that the new antivirals were authorized in Spain in 2015,
so in the first patients they were authorized as compassionate use,
a process that required several months.
All 15 patients underwent baseline abdominal ultrasound,
liver transplant Doppler and hepatic ARFI elastography in the 3 months prior to initiating the antiviral treatment and in the 3 months after its completion.
Also in the same week of the ultrasound examination,
an analytical study with a complete liver profile was performed and the viral load of HCV was determined.
ARFI elastography was performed using a convex multifrequency probe (4C1) with an ACUSON S2000 device (Siemens Medical Solutions,
Mountain View,
CA,
USA) with the specific software for generating and tracking shear waves Virtual Touch Tissue Quantification.
All studies were performed by two radiologist with more than 8 years of experience in performing ARFI.
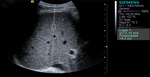
SWV was measured in 10 regions of interest (ROI) of the right hepatic lobe (figure 1).
A rectangular ROI with fixed dimensions (1 × 0.5 cm) was adjusted under ultrasound control to avoid identifiable blood vessels and biliary structures.
The main data provided in the team's software as a table for the subsequent analysis are the mean and the standard deviation (figure 2).
Biopsy was performed in 11 of the 15 patients,
and the anatomopathological data were collected.
The METAVIR classification (10) was used to semiquantitatively assess the degree of necroinflammatory activity and the degree of fibrosis.
Necroinflammatory activity was classified as: A0,
without necroinflammatory activity; A1,
scarce; A2,
moderate; and A3,
important.
Fibrosis was stratified on a scale of 0 to 4: F0,
without fibrosis; F1,
portal fibrosis without septa; F2,
portal fibrosis with some septa; F3,
numerous septa without fibrosis; and F4,
cirrhosis.
Significant fibrosis was defined as a degree greater than or equal to F2.
Statistical analysis: to estimate differences between the parameters measured before and after treatment,
the Wilcoxon test was used for paired data and the paired Student's t test.
The level of statistical significance was established at 0.05.
The software used was Stata / IC v.1.1 (StataCorp,
2015.
Stata Statistical Software: Release 14.
College Station,
TX: StataCorp LP).