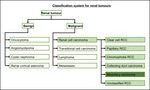
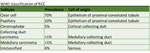
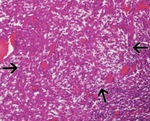
Classification system for renal tumours
Renal tumours are common,
and the incidence has increased greatly since the 1990s (1).
This in part due to an ageing population but also as a result of incidental findings on increasingly-used cross-sectional imaging.
Renal lesions should be classified when possible by the radiologist or histopathologist to inform the referring clinician of the expected clinical course,
prognosis,
and appropriate management options.
[Figure 1].
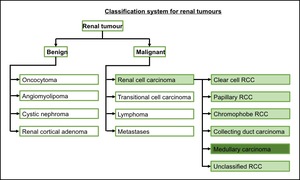
Fig. 1: A simple classification system for renal tumours.
Solid renal tumours can be benign or malignant.
The likelihood of malignancy varies depending on size at diagnosis.
Of tumours <1cm in diameter,
46% have been shown to be benign whilst of those >7cm only 7% are (3).
Of those that are benign,
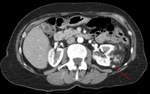
angiomyolipoma [Figure 2],
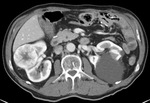
oncocytoma [Figure 3],
and benign adenomas are the commonly seen solid tumours.
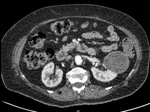
Cystic tumours (i.e.
cystic nephroma [Figure 4]) should be classified using the Bosniak classification (4, 5).
Of malignant tumours,
renal cell carcinoma contributes over 80% of cases [Figure 5].
~8% are transitional cell carcinoma [Figure 6].
Of the remainder,
there are cases of renal lymphoma and metastases (1).
In children,
a Wilms’ tumour is the most common,
however discussion of this is beyond the scope of this poster.
Renal cell carcinoma (RCC) is comprised of several subtypes.
The most common is clear cell RCC,
arising from epithelium of the proximal convoluted tubule.
Rarer subtypes include papillary RCC,
which is commonly seen in those with end-stage renal failure,
and collecting duct carcinoma (6). Tumours are staged according to the TNM classification.
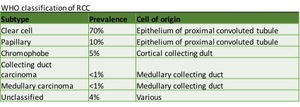
Fig. 5: Subtypes of RCC. Adapted from Prasad et al, 2006 (6).
References: Prasad et al. (2006). Common and uncommon histologic subtypes of renal cell carcinoma: imaging spectrum with pathologic correlation. Radiographics. 26(6):1795-1806
Epidemiology & pathophysiology of medullary renal carcinoma
RMC is incredibly rare,
with the majority of published reports being only case reports or small case series.
It was first described as a distinct entity in 1995 after 34 cases of serving US military personnel developing RMC over a 22 year period (7).
Only 220 cases have been described worldwide (2,
8).
Sickle cell haemoglobinopathy can cause renal disease.
For some time,
it was felt that intermittent macroscopic haemauturia in patients with sickle cell was a benign finding,
caused by the partial pressure of oxygen within blood at the renal medulla being below the sickling threshold (9).
However,
those with sickle cell have an increased risk of developing end stage renal failure (10).
RMC occurs in the calyceal epithelium,
where sickling is most pronounced.
It is hypothesised that this is due to repeated localised hypoxic injury,
although this is not explained by organs such as the spleen,
which more frequently suffer infarction form sickling,
not developing tumours for a similar reason (2).
The estimated lifetime prevalence of RMC among those with sickle cell trait is ~5 cases per 100,000 individuals (11).
RMC is almost always seen in those with sickle cell disease or sickle cell trait.
A few cases have been described in other haemoglobinopathies and 5 cases in patients with normal haemoglobin (2).
The median age at diagnosis is 24 years (8) and 96% of cases are less than 40 years old (2),
which is in contrast to other renal tumours.
RMC occurs more commonly in males (ratio 2.4:1).
Interestingly and inexplicably,
tumours are more often right-sided (2,
12).
RMC usually presents with at least one of the triad of signs & symptoms associated with a renal tumour: haematuria; flank pain; or a palpable mass.
The disease course is invariably aggressive.
The primary tumour is seen as an infiltrative mass arising close to the collecting system.
Most tumours are large at presentation,
measuring on average 6cm (2) with 88% of patients being Stage IV.
Survival rates are dismal: median survival is 8-13 months from diagnosis; 5 year survival rate just 9%; no patients with metastatic disease survived more than 3 years (8,
13).
Imaging features of primary and metastatic disease
General imaging features
Few articles on RMC have focussed on the imaging appearances however given the aggressive nature of RMC,
any radiological features that suggest the possibility of RMC may help inform management.
In general,
RMC is macroscopically seen as an infiltrative,
poorly-marginated mass,
arising from or abutting the pelvicalyceal and invading the renal sinus (14).
The primary lesion is often large: median size at diagnosis is 6cm but 11% are >10cm (7,
8).
The great majority are solid tumours,
although rare cases have been predominantly cystic (14).
As the tumour is generally located centrally,
it can cause obstruction of the renal pelvis or caliectasis.
The kidney can be enlarged by the tumour but without disruption of the normal reniform contour (15). Typically,
tumours are not calcified.
[Figure 7].
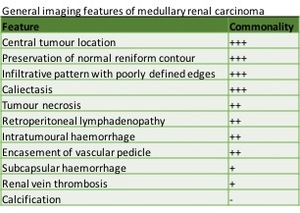
Fig. 7: General imaging features seen in renal medullary carcinoma, with approximation of how commonly findings are seen. Adapted from Blitman et al (12)
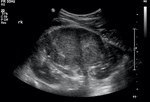
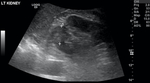
Ultrasound
Ultrasound is a useful screening test for patients with loin pain although in those with a palpable mass or high suspicion of renal tumour normal ultrasound should not be considered definitive.
In the few cases of RMC with described ultrasound findings,
an infiltrative mass has been seen within an enlarged kidney.
Areas of necrosis correspond to absent Doppler flow (15).
In several cases,
ultrasound has been falsely reassuring seeing either no abnormality or simply an enlarged kidney.
This can be due to preservation of the normal reniform contours (unlike with classical exophytic RCC) (12,
15).
Lymphadenopathy can sometimes be seen [Figure 8,
9].
The imaging appearances on contrast-enhanced ultrasound have thus far not been described.
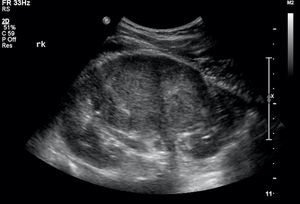
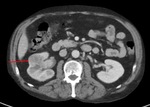
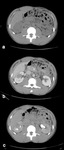
Fig. 8: Large right MRC seen arising close to the collecting system and growing outwards. There is still some preservation of the normal reniform contour despite the tumour's large size.
CT
CT scanning is the predominant modality for assessment of potential renal cancer.
CT can determine the extent of the primary tumour,
and the degree of local and distant invasion.
At our institution,
haematuria is investigated with cystoscopy and cross-sectional imaging of the abdomen and pelvis (non-contrast and split-bolus contrast enhanced imaging).
On non-contrast CT,
RMC is a heterogeneous mass that is predominantly isodense to the parenchyma and arises from the renal medulla.
Following contrast administration,
the lesion enhances but to a lesser degree than normal cortex or medulla on all phases (16).
Following contrast-administration,
the extent of the primary lesion can be seen,
although the margin is often indistinct.
The hypovascular nature of RMC can help differentiate it from other renal tumours.
Regions of necrosis or intra-tumoral haemorrhage are well-demonstrated on CT. RMC,
which grows into the renal pelvis,
often invades locally.
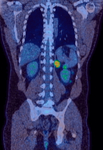
It can encase the renal vein or inferior vena cava or cause tumour thrombus (15) [Figure 10,
11].
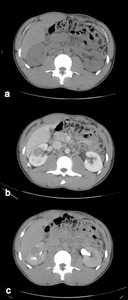
Fig. 10: CT images of a right lower pole RMC: a) non-contrast; b) portal-venous phase contrast-enhanced; c) delayed imaging.
The small tumour is isodense to the renal parenchyma on non-contrast imaging, then enhances but to a lesser degree than surrounding parenchyma which corresponds with its hypovascular nature.
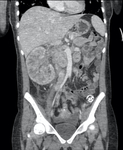
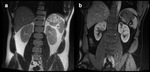
MRI
MRI is less commonly used to assess renal tumours,
however it offers excellent soft-tissue contrast.
The general features are similar to those seen on CT: a centrally-located primary tumour with indistinct margins,
which can be better defined following Gadolinium administration (12,
15).
MRI is thought to be as good as CT at defining the primary tumour and extent of local invasion (12) [Figure 12].
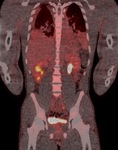
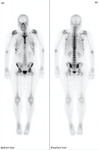
Nuclear Medicine
There are no specific molecular targets for RMC currently used in nuclear medicine.
18F-fluorodeoxyglucose (18F-FDG) PET/CT finding have only been described in 2 of the 217 published reports.
At our centre,
all 5 patients have underone PET/CT.
The primary lesions show very high tracer uptake,
far in excess of the adjacent normal renal collecting system.
PET/CT is particularly strong at looking for metastatic disease or local recurrence following radical nephrectomy [Figure 13].
Differentiating RMC from other renal tumours
Most imaging features of RMC are non-specific,
however a combination of the previously mentioned features in a young patient with sickle cell haemoglobinopathy should raise suspicion.
Other renal tumours can involve the medulla so differentiating purely on location can be difficult (14-16).
- 94% of clear cell RCCs have a typical,
expansile appearance with exophytic growth that disrupts the renal contour.
Contrast enhancement of clear cell RCCs is usually greater that the surrounding renal cortex.
- TCCs can be seen arising from the collecting system,
however these are often associated with gross hydronephrosis rather than minor caliectasis seen in RMC,
and patients are usually much older.
- Renal lymphoma can have an infiltrative appearance,
although this is rarely the presenting feature of non-Hodgkins lymphoma.
Usually renal lymphoma is usually bilateral and multi-focal; a feature that has not yet been described in RMC.
- Whilst most cases of RMC are solid,
a few cystic tumours have been seen.
However,
these still maintain an infiltrative appearance,
which can help differentiate from benign cystic tumours (e.g.
cystic nephroma),
which display a pseudocapsule or well-defined margins
- Collecting duct carcinoma has particularly similar imaging appearances to RMC as it too arises from the collecting system.
Both are biologically aggressive.
However,
collecting duct carcinoma has not been linked to haemoglobinopathy.
Metastatic disease
RMC has an aggressive course.
Often even radical nephrectomy with apparently clear surgical margins is not able to stop disease progression.
Of the 217 described cases in Alvarez et al’s systematic review,
158 patients had metastatic disease (73%) (2).
Even within a paediatric population,
88% were Stage IV at diagnosis (14).
In those with metastatic disease,
the most common sites are lymph nodes (68%),
lung (44%),
liver (28%),
adrenals (11%),
bone (6%) (2).
There are also case reports of metastatic brain and bone marrow deposits.
The retroperitoneal lymphadenopathy can be very marked.
Varied lung metastatic appearances from RMC have been described: small nodules; cannonball metastases; and thick pleurally-based rind (12).
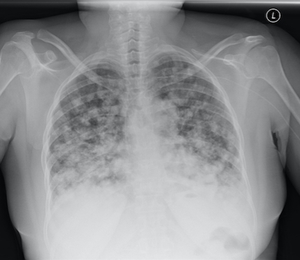
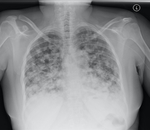
Fig. 14: PA chest radiograph. Innumerable small nodules throughout both lungs consistent with pulmonary metastases from MRC.
CT and MRI are both excellent modalities for identifying abdominal and pelvic metastases.
CT can characterise disease within the lungs.
For those areas of disease 1cm or larger,
18F-FDG PET/CT can be useful.
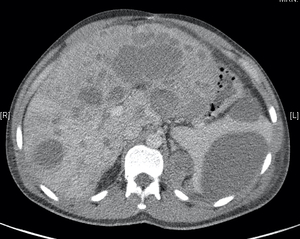
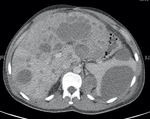
Fig. 17: Non-contrast axial CT scan. Splenic collection with widespread hepatic metastases in addition to metastatic nodules abutting the left diaphragmatic crus.
Histopathological correlation
RMC and collecting duct carcinoma have similar imaging appearances.
Even histologically,
they were once considered to be part of the same spectrum
Initially RMC was considered to be part of collecting duct carcinoma,
although now it is known to be clinically and pathologically distinct (8).
RMCs are a high-grade epithelial tumour with features of poorly-differentiated adenocarcinoma (17).
A variety of morphological appearances have been described,
primarily a diffuse infiltrative pattern arising the medulla and growing towards the renal sinus.
There can be acute and chronic inflammatory change,
haemorrhage,
necrosis,
and sickled erythrocytes (18).
Both RMC and collecting duct carcinoma display dense stromal desmoplasia and positive results on mucicarmine staining (12).
Presence of a cystic or papillary appearance on histopathology suggests collecting duct carcinoma over RMC (6).
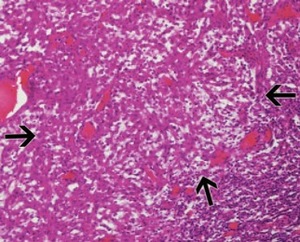
Fig. 18: Sheets of high-grade tumour cells with a prominent lymphocytic response. Taken from Prasad et al, 2006 (6).
References: Prasad et al. (2006). Common and uncommon histologic subtypes of renal cell carcinoma: imaging spectrum with pathologic correlation. Radiographics. 26(6):1795-1806
Loss of SMARCB1/INI1 gene expression on immunohistochemistry has been shown to assist histopathological assessment,
however arguably knowledge of sickle cell status is more useful (11,
17).
RMC samples have shown variable antibody expression,
however immunohistochemistry profiling consistently includes positive expression of low molecular weight cytokeratin,
epithelial membrane antigen and vimentin,
with absence of cytokeratin 34βE12 (which is seen in renal collecting duct carcinomas) (19).
Whenever RMC is found,
the RMC Working Group recommend radical nephrectomy with lymph node dissection and enrolment in clinical trials for chemotherapy.
If not possible,
other traditional chemotherapy combinations should be considered (20).
In spite of this aggressive management approach,
even radical nephrectomy with clear surgical margins does not consistently stop disease progression (8).