While,
in recent years,
survival rates for children with cancer increased,
imaging technologies available have evolved to include a wide array of modalities.
Ultrasound is the first-line imaging modality in pediatrics,
particularly for patients presenting with a palpable abdominal mass,
therefore being the most common method of diagnosis.
On ultrasound,
neuroblastomas are generally solid,
occasionally with small punctate echogenic areas.
However,
the echogenicity on ultrasonography varies; some may be evenly echogenic,
whereas others are highly heterogeneous.
Acoustic shadowing behind the areas of calcification may or may not be present.
The tumor mass exceptionally invades the kidney,
but when it does this makes differentiation from a Wilms tumor even more difficult.
Flow is often detected on color Doppler,
aiding in the differential.
Doppler is essential to evaluate encased and displaced IVC,
aorta,
celiac axis and mesenteric vessels.
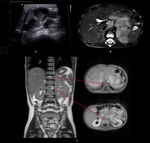
Lymphadenopathy is often a feature of the tumoral mass,
sometimes being difficult to discriminate between the two and furthermore misdirecting the diagnosis by suggesting a lymphomatous pathology (Fig.
1).
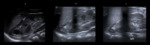
Although occasionally difficult,
distinguishing abdominal neuroblastoma from Wilms tumor at presentation is critical,
as surgical management differs significantly (Fig.
2).
Both neuroblastoma and Wilms tumour occur in early childhood and typically present as a large abdominal mass closely related to the kidneys.
Distinguishing between the two is essential,
and a number of features are helpful,
albeit none are pathognomonic.
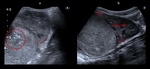
Calcification is one of the features of neuroblastoma that helps differentiate it from a Wilms tumor.
The presence of fine pinpoint calcification favors neuroblastoma and is seen in 80-90% of cases.
While neuroblastoma is likely to displace,
compress and rotate the kidney,
Wilms’ tumor arises from the kidney (Fig.
3).
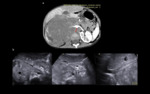
Neuroblastoma tends to be a mass crossing the midline,
encasing and displacing vessels,
rather than infiltrating them,
while a tumor thrombus in the renal vein or inferior vena cava is highly predictive,
but not invariably diagnostic,
for Wilms tumor (Fig.
4).
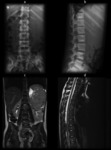
A paravertebral mass with calcifications and intra-spinal extension advocate for a neuroblastoma.
Heterogenous lesions,
the presence or absence of calcifications,
displacement or invasion of vessels suggest the pathology.
However,
the diagnosis may be problematic by reason of often overlapping imaging features leading to misdiagnosis and,
therefore,
may not be established only on the initial imaging findings,
often requiring follow-up imaging and correlation with clinical history.
Although widely used and unquestionably beneficial,
ultrasound as a sole imaging method is generally insufficient.
The role of ultrasound is to provide the initial diagnosis,
which will thus direct further imaging and investigation.
CT and MRI provide better assessment regarding the local extent of the tumor and are required as a guide to staging,
resectability,
prognosis and follow-up.
On CT,
neuroblastomas tend to appear lobulated and to grow in an invasive pattern,
surrounding and engulfing,
rather than invading,
vessels.
The masses are often inhomogeneous secondary to hemorrhage,
necrosis and calcifications.
Calcifications are seen by CT in 80-90% of cases.
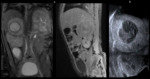
On MRI,
neuroblastoma,
like most other malignancies,
appears of high signal on T2-weighted images,
but can also be heterogeneous (Fig.
5c).
MRI is superior in detecting neuroforaminal involvement and intraspinal extension (Fig.
5d).
Typically obtained in the occurrence of skeletal pain or limping due to skeletal metastases,
conventional radiographs may show a number of fortuitous findings,
leading to the diagnosis.
An intra-abdominal soft-tissue mass with fine calcifications may be seen (Fig.
5a) with evidence of lucent,
ill-defined bony metastases (Fig.
5b).
Proximity to adjacent bones may cause remodelling of vertebral bodies and,
furthermore,
pedicle erosion from the potential intraspinal extension may be detected,
a hallmark of neuroblastoma.
Nuclear medicine studies are essential for metastatic surveillance.
MIBG labeled iodine-123 shows uptake both in primary tumor and metastases.
Evaluation of skeletal lesions is routinely done using technetium-99m MDP,
being much more sensitive than conventional radiography in the detection of bone disease.
The role of positron emission tomography (PET) and PET/CT is still being defined.