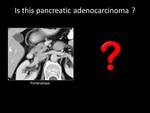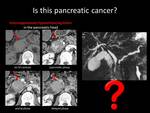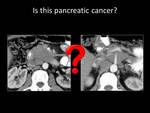
Fig. 1: QUIZ no1
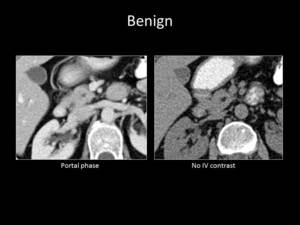
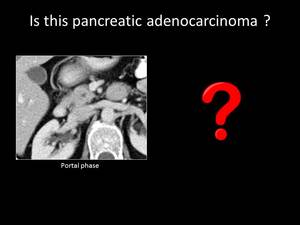
Fig. 2: QUIZ no1
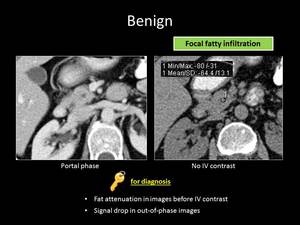
Fig. 3: ANSWER QUIZ no1: Focal fatty infiltration
BENIGN CONDITIONS
FOCAL FATTY INFILTRATION
Fatty infiltration of the pancreas can be focal or diffuse.
It is a benign procedure related with diabetes,
obesity,
focal pancreatitis,
steroid therapy,
cystic fibrosis and other conditions.
Focal fatty infiltration is usually most prominent in the head of the pancreas and on contrast-enhanced CT it usually appears as an hypoenhanced region compared to the normal pancreatic tissue,
simulating PA.
The key finding that leads to the correct diagnosis is the presence of fat density within the lesion on non-contrast-enhanced CT or signal drop on opposed phase chemical shift MRI.
Additional helpful findings in favor of focal fatty infiltration are the preserved lobulated pancreatic outline,
the absence of pancreatic or biliary duct dilatation,
the absence of encasement of adjacent vessels,
and the absence of lymphadenopathy.
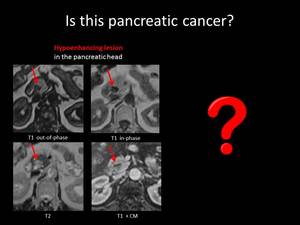
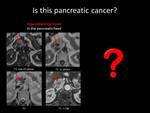
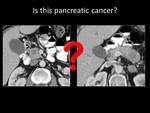
Fig. 4: QUIZ no2
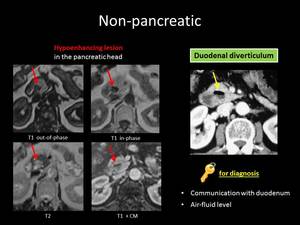
Fig. 5: ANSWER QUIZ no2: Duodenal diverticulum
DUODENAL DIVERTICULUM
Duodenum is the second most frequent GI tract part after colon,
in which diverticula appear,
usually in the periampullary region.
They are typically acquired and,
in most cases,
they are not difficult to diagnose,
as they demonstrate an air-fluid level or hyperattenuating content due the presence of oral contrast agent.
Nevertheless,
sometimes diverticula have been misdiagnosed as pancreatic tumors.
A collapsed diverticulum or one with a totally fluid content could masquerade to a suspicious lesion of the pancreas.
This ambiguous finding can be overcome by sufficient dilatation of the duodenal loop, in order to demonstrate the communication of the lesion with the duodenal lumen.
Cases of diverticulitis or diverticular rupture are even harder to correctly diagnose.
In such cases,
endoscopy can contribute in making the correct diagnosis.
Alternatively,
CT can be repeated within a few days,
after remission of the clinical and radiological findings.
__________________________________________________________________
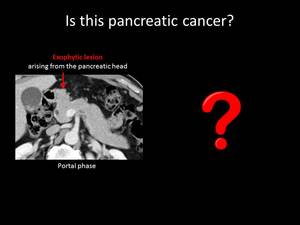
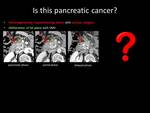
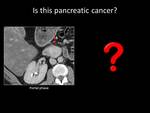
Fig. 6: QUIZ no3
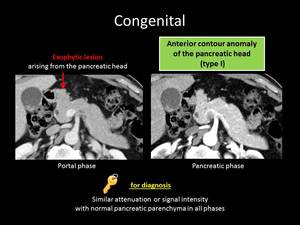
Fig. 7: ANSWER QUIZ no3: Normal variation of pancreatic head contour
CONGENITAL
NORMAL VARIATION OF THE PANCREATIC HEAD CONTOUR
Normal variation of the pancreatic head contour occurs in 35% of the population and can sometimes imitate malignancy.
When normal pancreatic tissue extends over 1cm relatively to the anterior superior pancreaticoduodenal artery (ASPD),
it is referred as lobulation of the head of the pancreas.
There are three types of parenchymal bulging: Type I,
II and III refer to anterior,
posterior and horizontal bulging relatively to the ASPD respectively.
Familiarity with these alterations is important in order to overcome their misdiagnosis as PA.
The key finding suggestive of the presence of a normal variation of the pancreatic contour is that this outpouching has similar enhancement patterns with the rest of the pancreatic parenchyma in all phases (nonenhanced,
pancreatic,
portal and venous phase).
Coronal and sagittal reformations can supplementary assist to the visualization of the lobulated contour.
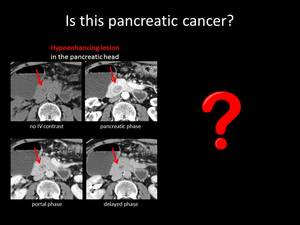
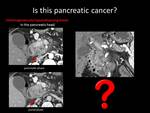
Fig. 8: QUIZ no4
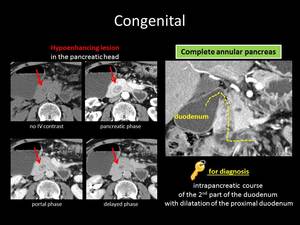
Fig. 9: ANSWER QUIZ no4: Annular pancreas
ANNULAR PANCREAS
Annular pancreas is a rare variant (1/20000 prevalence) in which pancreatic parenchyma surrounds the second portion of the duodenum completely or partially.
When symptomatic in children,
it usually presents with gastric outlet obstruction symptoms.
In adults,
it is often an incidental finding or it occurs with obstruction symptoms,
peptic ulcers or pancreatitis.
When the intrapancreatic portion of the descending duodenum is not clearly opacified,
it can be mistaken for an hypoenhancing suspicious lesion.
Following the course of the duodenum carefully can help us overcome this deception.
Another key finding in favor of annular pancreas is the detection of Santorini’s duct in the normal pancreatic tissue that surrounds the duodenum.
In some cases,
stenosis of the descending duodenum can be also demonstrated.
In uncertain cases,
secretin- enhanced MRI can help us make the correct diagnosis.
__________________________________________________________________
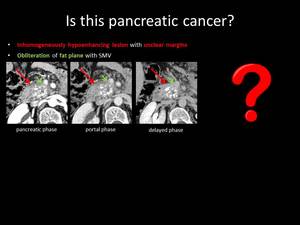
Fig. 10: QUIZ no5
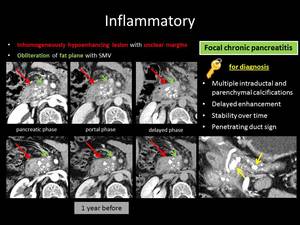
Fig. 11: ANSWER QUIZ no5: Focal chronic pancreatitis
INFLAMMATORY
FOCAL CHRONIC PANCREATITIS
On imaging,
chronic pancreatitis is characterized by parenchymal atrophy,
dilatation of the main pancreatic duct and presence of calcifications.
Nearly 20% of chronic pancreatitis cases develop a focal form,
which can simulate PA and is usually associated with increased alcohol consumption.
Furthermore,
the high incidence of PA in chronic pancreatitis patients makes the differential diagnosis between these two entities even more challenging.
On CT,
focal chronic pancreatitis appears as a low attenuating mass that causes peripheral dilatation of the main pancreatic duct and atrophy of the peripheral pancreatic parenchyma.
The depiction of a penetrating pancreatic duct within the suspicious lesion is indicative of an inflammatory pseudotumor,
as well as the absence of encasement of the peripancreatic vessels. Diffusion weighted MRI provides additional assistance in equivocal cases as it demonstrates lower apparent diffusion coefficient (ADC) values in PA compared to the rest pancreatic tissue,
but not significant differences of ADC values in focal chronic pancreatitis with the rest of the pancreas.
Nevertheless,
according to NCCN guidelines,
in patients with chronic pancreatitis and increased likehood of PA,
resection is strongly recommended even without histological proof.
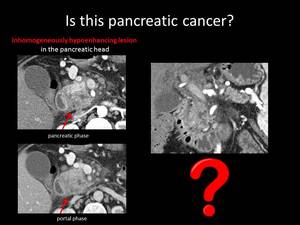
Fig. 12: QUIZ no6
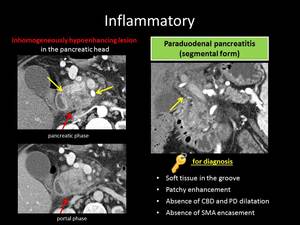
Fig. 13: ANSWER QUIZ no6: Paraduodenal pancreatitis
PARADUODENAL PANCREATITIS
Paraduodenal (or groove) pancreatitis (PP) forms another distinct entity of chronic pancreatitis with debatable prevalence.
Although the precise cause is not known,
it is typically presented in middle aged men with a history of alcohol consumption with durable (3-6 months) episodes of jaundice and weight loss or with symptoms of acute pancreatitis.
It is divided into two types: inflammatory tissue and fibrotic change can affect either only the groove region (pure) or can also affect the pancreatic head (segmental).
On CT,
the pure form usually appears as a soft tissue mass in the groove region,
which is described as sheet-like tissue that shows late enhancement due to fibrosis.
Additional pancreatic head involvement appears in the segmental form.
Both types may mimic PA and,
unfortunately,
even negative FNA results cannot exclude malignancy with absolute certainty.
The particular location of PP is an important clue to the diagnosis as is the displacement of the vasculature with absence of obstruction.
Οther characteristics of PP are duodenal wall thickening,
and patchy heterogeneous enhancement unlike PA which is usually hypoenhancing homogeneously.
Moreover,
the presence of small cysts in the duodenal wall is another demonstration of PP that is unusual in PA.
Additionally,
the smooth,
long narrowing of the pancreatic and the common bile duct favors PP.
Conversely,
marked pancreatic duct dilatation or sharp pancreatic duct interruption are suspicious findings that indicate the presence of malignancy.
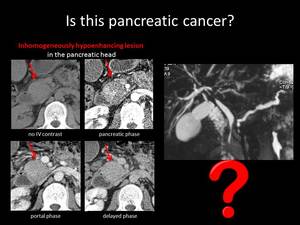
Fig. 14: QUIZ no7
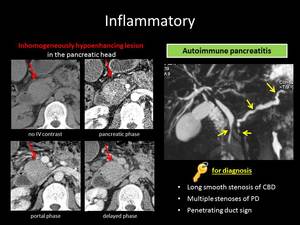
Fig. 15: ANSWER QUIZ no7: Autoimmune pancreatitis
AUTOIMMUNE PANCREATITIS
Autoimmune pancreatitis (AIP) is one of the most frequent misdiagnoses that lead to pancreatic surgery with the suspicion of PA.
It is a distinct type of chronic pancreatitis that benefits impressively from steroids and it is divided into two types.
Type 1 AIP is an IgG4 related disease with extra-pancreatic manifestations (sclerosing mesenteritis,
Sjogren-like syndrome,
retroperitoneal fibrosis,
interstitial nephritis),
whereas type 2 AIP coexists with IBD in 30% of cases.
Diffuse “sausage-like” parenchymal enlargement with effacement of the lobular contour of the pancreas and capsule-like rim with delayed enhancement are typical radiologic features of autoimmune pancreatitis.
Nevertheless,
in 30–40% of patients with AIP a focal masslike enlargement of the pancreas is seen,
which simulates PA.
Imaging findings suggestive of focal AIP are patchy inhomogenous enhancement in the pancreatic phase and homogeneous enhancement in the late phase,
unlike PA which shows decreased enhancement in both phases.
Another differential clue of high specificity for AIP is the “capsule-like” rim of low attenuation in contrast to the high attenuation rim that surrounds PA in some cases.
Additionally,
segmental narrowing of the main pancreatic duct (MPD) is usually multifocal and milder in AIP,
whereas dilatation of the MPD more than 5mm distally to the lesion is suggestive of malignancy.
Some other key imaging findings in favor of AIP are the duct penetrating sign and the delayed MPD enhancement,
as well as presence of extrapancreatic manifestations (sclerosing cholangitis,
interstitial nephritis,
sclerosing mesenteritis,
retroperitoneal fibrosis in type 1 or ulcerative colitis in type 2).
High suspicion of AIP should be confirmed by high serum IgG4 levels and an EUS guided biopsy.
In case of ambiguous biopsy results,
a 4-6 weeks steroid treatment is recommended,
for therapeutic and diagnostic purposes.
__________________________________________________________
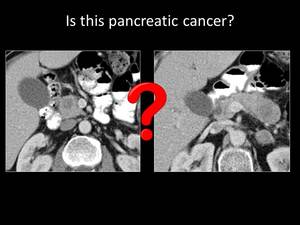
Fig. 16: QUIZ no8
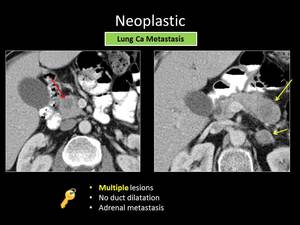
Fig. 17: ANSWER QUIZ no8: Metastases
NEOPLASTIC
METASTASIS
Pancreatic metastases are quite uncommon and account for 1-2% of all pancreatic neoplasms.
The most common primaries that can metastasize to the pancreas are renal,
lung,
breast,
melanoma,
colon,
gastric,
ovary and prostate cancer.
Patient’s history of malignancy indicates increased suspicion for metastatic disease.
Metastases are often multifocal and usually well circumscribed,
unlike PA which is typically solitary and tends to have irregular borders.
In most cases of pancreatic metastases the main pancreatic duct and the common bile duct is not dilated, in contrast to PA.
Another key finding is the presence of contemporary metastases in organs that pancreatic cancer is unlike to spread (bones,
adrenals).
It should be noted that hyperenhancing lesions usually originate from an hypervascular neoplasm,
such as renal cancer,
and hypoenhancing lesions originate from hypovascular tumors,
like lung cancer.
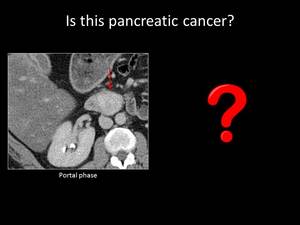
Fig. 18: QUIZ no9
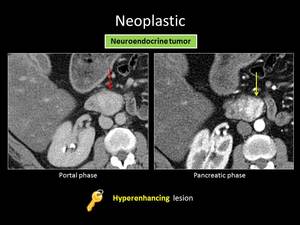
Fig. 19: ANSWER QUIZ no9: Neuroendocrine tumor
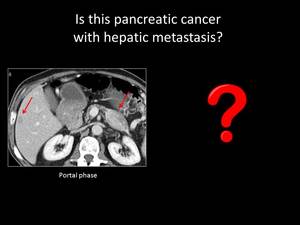
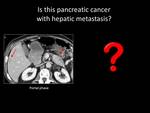
Fig. 20: QUIZ no10
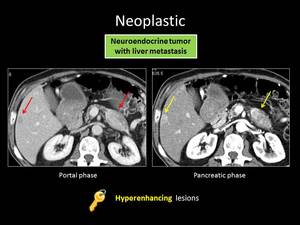
Fig. 21: ANSWER QUIZ no10: Pancreatic neuroendocrine tumor with liver metastasis
PANCREATIC NEUROENDOCRINE TUMORS
Pancreatic neuroendocrine tumors (PNETs) originate from well-differentiated endocrine pancreatic cells and compose a variety of benign to malignant entities with a prevalence of 5-10% of the pancreatic tumors.
Although PNETs can be associated with von Hippel-Lindau disease,
neurofibromatosis type 1,
multiple endocrine neoplasia (MEN) type 1,
and tuberous sclerosis,
their occurrence is mostly sporadic.
Their survival rate (81%) is remarkably higher than PA’s.
PNETs typically present as solid masses that avidly enhance in the arterial phase.
Unlike PA they are well demarcated and do not cause pancreatic duct dilatation,
except for small serotonin secreting tumors which tend to cause pancreatic duct obstruction due to fibrous tissue formation.
In challenging cases with heterogenous morphology or pancreatic duct dilatation octreotide scintigraphy can help us make the correct diagnosis.
PNET liver metastases are not infrequent and are usually hypervascular when they are small and necrotic or hemorrhagic when they are larger.
Although liver metastasis from PA constitutes an absolute contraindication for surgery,
when it comes to PNET metastatic liver disease,
the management is by far different.
Patients with PNET liver metastasis seem to benefit from primary resection and multimodal therapy of hepatic metastasis.
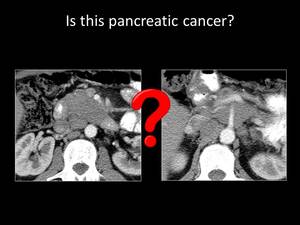
Fig. 22: QUIZ no11
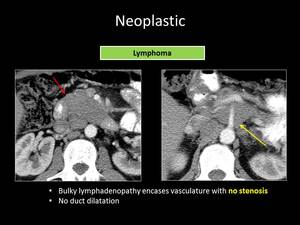
Fig. 23: ANSWER QUIZ no11: Lymphoma
LYMPHOMA
Secondary pancreatic involvement from lymphoma (mainly non-Hodgkin) is not rare and is often part of extranodal involvement in 5-30% of the patients.
It can occur either by infiltration from adjacent peripancreatic lymph nodes or by hematogenous dissemination.
Primary lymphoma of the pancreas is quite uncommon (less than 0.5%).
The focal form of pancreatic lymphoma usually presents as a solitary,
bulky,
homogenous lesion with lower attenuation than muscles and shows mild homogenous enhancement in contrast-enhanced CT.
Knowledge of patient’s history of lymphoma when secondary pancreatic lesions are revealed and the existence of a bulky mass that does not cause significant pancreatic duct dilatation makes the diagnosis of lymphoma more favorable than PA.
Additionally,
lymphomatous involvement of the pancreas is likely to cause vessel encasement without invasion.
Finally,
presence of enlarged lymph nodes below the level of the renal veins is another important imaging clue in favor of lymphoma.