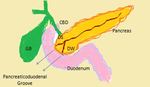
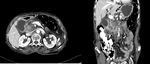
PANCREATİCODUODENAL GROOVE:
- Pancreatic groove is a theoretical space defined by pancreatic head (medially),
2 nd portion of duodenum (laterally),
3 rd portion of duodenum and IVC (posteriorly),
and duodenal bulb (superiorly) also contains distal CBD,
main/accessory pancreatic ducts,
and major/minor papilla (Fig 1)
- Chronic pancreatitis affecting pancreaticoduodenal groove
Pure form (sheet like): affects only pancreaticoduodenal groove
Segmental form (mass like): affects pancreaticoduodenal groove and extends medially into pancreatic head
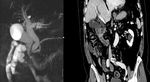
IMAGING FEATURES:
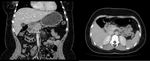
- İll–defined fat stranding and inflamatory change to frank soft tissue in the groove
- Delayed enhancement as a result of fibrotic components
- Thickening of the medial duodenal wall
- Duodenal stenosis
- Small cysts
- Mass like enlargement of the pancreatic head (segmental form)
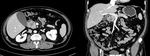
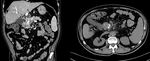
- CBD can appear attenuated and narrowed (This narrowing is relatively smooth,tepered and regular) (Fig 2,
3)
DIFFERENTIAL DIAGNOSİS:
Pancreatic adenocarsinoma:
- Most common pancreatic neoplasm (85-90% of all pancreatic tumors)
- Poor prognosis and high morbidity
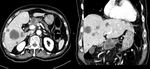
- As pancreatic carcinoma is a hypovascular tumor,
it presents as a hypodens mass on a CT.
- Although predominantly solid,
cyst-like features (cystic degeneration/necrosis) are associated with 8% (Fig 4)
The differentiation of pancreatic adenocarcinoma from groove pancreatitis can be extremely difficult,
and many cases ultimately proceed to surgery because of an inability to reliably make this distinction.
Most pancreatic adenocarcinomas do not show internal cystic change and are much more likely to infiltrate posteriorly into the retroperitoneum and encase the vasculature.
Moreover,
thickening of the medial duodenal wall,
a common finding with groove pancreatitis,
is quite uncommon with pancreatic adenocarcinoma.
Yamada et al (7) found that analysis of time attenuation curves at contrast-enhanced CT allowed differentiation between the two entities: Focal pancreatitis showed a delayed washout pattern,
whereas adenocarcinoma showed a pattern of increasing enhancement over time,
peaking at 150 seconds.
And gadolinium-enhanced magnetic resonance (MR) imaging and magnetic resonance cholangiopancreatography (MRCP) imaging are useful for differentiating chronic pancreatitis from pancreatic adenocarcinoma.
Differentiating features pancreatic adenocarsinoma from paraduodenal pancreatitis
- Mass confined to pancreatic head
- Abrupt cutoff of CBD and pancreatic duct with “double duct” sign
- Atrophy of pancreas
- Signs of vascular invasion or metastases
Duodenal Carcinoma:
• Intraluminal mass with a short segment of bowel wall thickening
• Irregular thickening of duodenal wall
• Concentric narrowing of duodenal lumen
• Local lymphadenopathy and local infiltration (Fig 5)
These tumors can be quite difficult to differentiate from groove pancreatitis,
especially when they present as focal thickening of the medial duodenal wall.
Most small-bowel adenocarcinomas arise from the duodenum or proximal small bowel,
and close attention to the coronal multiplanar reformats may allow the accurate distinction of a mass arising in the duodenal wall from a process truly centered in the pancreaticoduodenal groove.
Ampullary Carsinoma:
- Heterogeneous group of malignant epithelial neoplasms (adenocarcinoma) arising from ampulla of Vater
- Enhancing (not delayed) mass in the region of the ampulla
- Double duct sign with obstruction of both common bile duct (CBD) and pancreatic duct (PD)
- No cystic changes (Fig 6)
The focality of these malignant lesions at the ampulla should be distinguished from the more ill-defined crescentic soft tissue seen with groove pancreatitis.
However,
especially when ampullary carcinomas grow larger,
the distinction may not be so simple.
Focal pancreatitis:
• Caused by acinar cell injury and premature activation of trypsinogen to trypsin
• Focal enlargement of the pancreas or a focal inflammatory mass,
often in the pancreatic head
• Not primarily centered in pancreaticoduodenal groove,
usually involves entire pancreas
• Associated with peripancreatic inflammation,
including retroperitoneal fluid and fat stranding
• Should resolve on follow-up examinations
• Pseudocyst is the most common complication of acute pancreatitis (Fig 7)
Groove pancreatitis typically shows little retroperitoneal inflammation or fluid,
and even in the segmental form,
involvement of the pancreas is usually limited to the pancreatic head.
This should be contrasted with typical acute edematous pancreatitis,
which usually involves a substantial portion of the pancreatic parenchyma,
does not appear centered in the groove,
and is typically characterized by peripancreatic fluid and inflammation tracking into the pararenal spaces.
Moreover,
elevated lipase levels are not characteristic of groove pancreatitis and are an important differentiating feature.
Finally,
typical edematous pancreatitis should usually resolve on follow-up studies,
whereas the imaging findings associated with groove pancreatitis often persist.