Patient selection
We enrolled 42 patients between February and June 2017.
Eligibility criteria were: symptomatic (Fontaine stage II or III) peripheral arterial disease (PAD) and glomerular filtration rate over 60 mL/min /1.73 m2.
The number of the patients was determined by increasing the minimal recommendation of the U.S.
Food & Drug Administration by 33% [20].
Image acquisition
We performed lower limb angiography according to our institutional protocol,
using a Siemens Artis Zee (30x40 cm detector,
Siemens Healthcare GmbH) and a Syngo workstation (Siemens Syngo XWP VD11B,
Service Pack 2,
Munich,
Germany).
CM volumes were judged by the investigator for each individual patient.
The femoral region was recorded using 10 mL,
the knee region 10-14 mL,
the ankle region 14-22 mL CM.
The unsubtracted image series were saved for later evaluation with the kinetic technology.
Post processed DSA images were calculated on the Syngo workstation and used for diagnosis.
Post-processing involved the "pixel shift" motion correction and noise-filtering.
Post-processed DSA image series were also saved and used to create post-processed kinetic images.
Four images representing the same location were generated: kinetic image without post-processing,
sumDSA without post-processing,
post-processed sumDSA and post-processed kinetic image.
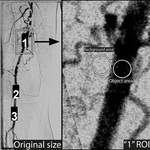
Signal-to-Noise Analysis
SNR measurements were carried out in 1902 ROIs of 110 images.
SNR was determined by the ratio of the average signal value to the image’s random intensity variations,
as introduced by Rose [19].
The signal value was calculated as C = Istructure – Ibackground (C = absolute contrast,
I = average intensity).
Random intensity variations were estimated by measuring the standard deviation of the measured background intensities. (Fig1.)
Images of the inferior parts of the lower limbs were affected by motion artefacts that could be removed with "pixel shift" registration.
However,
image registration involves non-linear interpolations that alter noise distribution and distort its measurement.
For this reason,
SNR calculations were only performed in the abdominal,
pelvic and femoral regions.
The following software were used: Kinepict software (version: 0.2),
for generating kinetic images; Fiji (version: 1.51n) for generating sumDSA images,
selecting ROIs,
and measuring signal and noise; and Microsoft Excel 2016 (Microsoft,
Redmond,
WA,
USA),
for calculating SNRs,
and statistical analysis.
Visual Performance and Quality Comparison
A blinded evaluation of image pairs was done by three vascular surgeons and three interventional radiologists.
Two comparisons were made.
First,
the basic kinetic image and the postprocessed kinetic image were compared (n = 232 image pairs from all regions combined).
Second,
the post-processed kinetic image was compared with the postprocessed sumDSA (n = 238 image pairs).
Image pairs were evaluated in random order.
Each image pair was evaluated once by every participant.
Evaluators had to select the better image based on three questions that focused on different aspects of image quality and usability:
Q1: Which image is more detailed?
Q2: In which image are anatomic structures more discernible?
Q3: Which image is more useful diagnostically?
Statistical analysis
Statistical analysis of the visual comparison was done in several steps.
Agreement between clinicians was determined.
The level of agreement for each region was calculated as the mean of the percentage of the raters choosing kinetic imaging oved DSA for every individual image.
Fleiss’ kappa tests were performed to obtain p values corresponding to the levels of agreement [21,22].
The 95% confidence intervals were also determined.
Calculations were made using Stata 15.0 statistical data analysis software (StataCorp,
College Station,
TX,
USA).