Type:
Educational Exhibit
Keywords:
Neoplasia, Cancer, Radiation therapy / Oncology, MR, CT, Neuroradiology brain, CNS
Authors:
A. Chattopadhyay1, J. Coates2, I. Craven2, M. S. Igra2, S. Currie2; 1Leeds, West Yorkshire/UK, 2Leeds/UK
DOI:
10.1594/ecr2018/C-3236
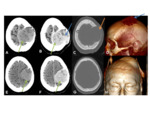
Findings and procedure details
The key imaging features of malignant meningeal lesions are described,
across a wide selection of cases from a large tertiary referral centre.
MENINGIOMA
Background:
- Most common tumour of the meninges- originating from the meningocytes or arachnoid cap cells of the meninges
- WHO classification: Grade I- benign,
Grade II- atypical,
Grade III- anaplastic/malignant.
- Treatment is surgical excision
Imaging:
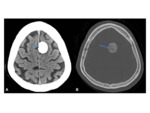
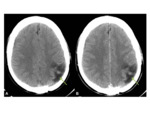
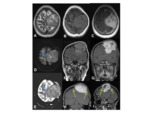
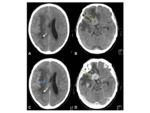
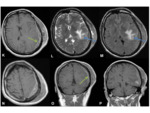
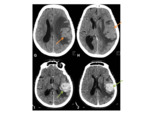
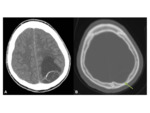
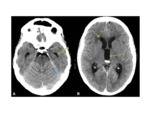
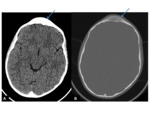
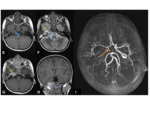
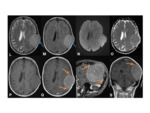
- Extra-axial tumours
- Commonly parasagittal Fig. 1 ,
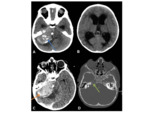
located at the sphenoid ridge Fig. 3 and cerebral convexity.
- Characteristic appearances:
- Well-circumscribed
- Can either be uniformly calcified Fig. 1,
or have foci of internal calcification Fig. 3
- Wide dural base.
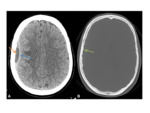
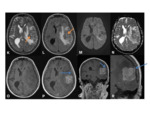
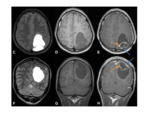
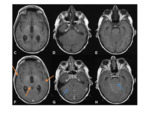
Fig. 2
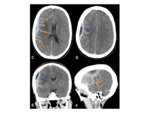
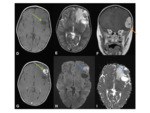
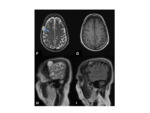
- Dural tail- though not specific for meningioma,
is seen in the majority of cases Fig. 4
- CSF cleft sign- CSF demonstrated between the convex border of the mass and the adjacent brain parenchyma confirms the extra-axial location.
Fig. 2
- Spoke-wheel or sun-burst pattern of vessels and subsequent enhancement Fig. 4
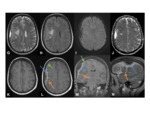
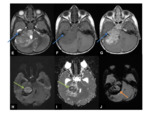
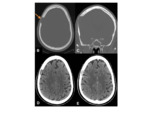
- Often has surrounding vasogenic oedema in the adjacent brain parenchyma Fig. 5
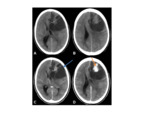
•CT
- Iso to hyperdense mass
- Well-circumscribed extra-axial lesion
- Avid enhancement following contrast
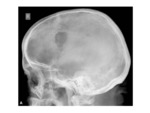
- Causes bony hyperostosis of the adjacent calvarium and skull base.
Fig. 5
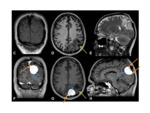
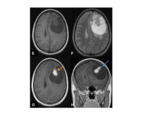
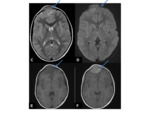
•MRI
- Investigation of choice for confirming the diagnosis and allowing further characterisation Fig. 4
- T1- isointense to grey matter
- T2- isointense to grey matter
- T1+Gd- avid enhancement
- Can show diffusion restriction in the atypical and malignant subtypes
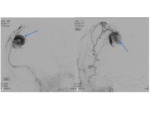
•Angiography
- Can be done pre-operatively
- Vascular tumours
- Characteristic sunburst or spoke-wheel pattern of vascularity.
“Mother in law sign” refers to the contrast blush on angiography- “comes early,
stays late,
and is very dense” !
HAEMANGIOPERICYTOMA
Background:
- Rare neoplasm of the pericytes that originate in the meninges.
- Tends to affect young patients
- The tumours have recently been reclassified: haemangiopericytomas and solitary fibrous tumours of the dura are now considered different manifestations of the same disease- haemangiopericytomas at the aggressive end of the spectrum.
- High risk of disease recurrence post resection.
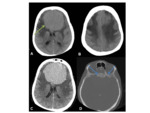
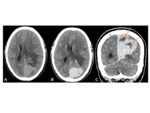
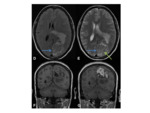
Fig. 9 Fig. 10 Fig. 11
- Treatment is with surgical resection.
Pre-operative catheter embolisation is used to minimise operative blood loss.
- The clinical presentation,
radiological appearances and intraoperative findings may be indistinguishable from those of meningiomas.
Imaging:
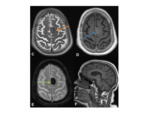
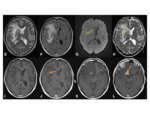
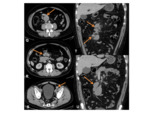
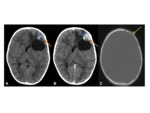
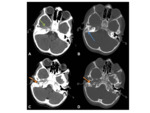
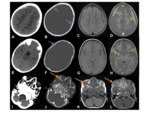
- Can be difficult to distinguish from meningiomas Fig. 6 Fig. 7 Fig. 8
- Large,
locally aggressive dural mass,
with bony erosion and osseous extension through the skull vault Fig. 12
- Almost always solitary
- Often supratentorial
- The borders can be lobulated
- Highly vascular,
demonstrating vivid enhancement
•CT:
- The lesions erode through bone,
rather than causing hyperostosis (unlike meningiomas) Fig. 12 Fig. 13
- Calcification is rare (again,
unlike meningiomas)
- There is marked contrast enhancement.
•MRI:
- T1: Isointense to grey matter
- T2: Isointense to grey matter.
Multiple flow voids will present as linear areas of signal dropout
- T1+Gd: Avid enhancement.
This can also highlight the dural attachment and the dural tail.
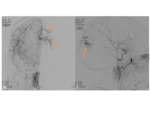
•Angiography:
- Highly vascular lesion,
- Common findings include “corkscrew arteries” and an ill-defined tumour stain.
LYMPHOMA
Background:
- Primary dural lymphoma is a rare subtype of primary central nervous system lymphoma (PCNSL).
It is usually a low-grade marginal zone lymphoma,
whereas the other types of PCNSL are usually high grade,
diffuse large B-cell lymphomas.
- Secondary infiltration from surrounding lymphomatous deposits,
or from disease elsewhere in the body is much more common than primary lymphoma Fig. 22
- Systemic lymphoma can invade the meninges without evidence of direct spread
Imaging:
- Characteristics depend upon whether the patient is immunocompromised or immunocompetent,
and whether the disease is primary or secondary.
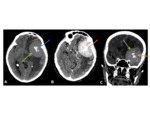
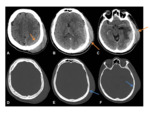
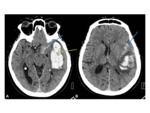
- Variety of radiological manifestations- can appear quite unusual Fig. 14 Fig. 17 Fig. 19
- Can show widespread meningeal calcifications,
discrete masses and single or multiple areas of involvement.
- Tends to show marked meningeal enhancement.
Fig. 15 Fig. 17
•CT:
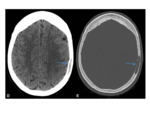
- Lymphomatous deposits tend to be hyperdense on unenhanced studies.
Fig. 17
- Superior to MRI at assessing bony involvement- which typically presents as permeative disease,
occasionally with periosteal reaction.
Fig. 14 Fig. 19 Fig. 20
- Sulcal effacement and mass effect may be suggestive of meningeal disease on non-contrast studies.
- Avid enhancement of both parenchymal and meningeal disease.
- Meningeal disease can result in obstructive hydrocephalus.
•MRI:
- T1 and T2- isodense lesions
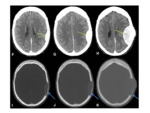
- T2-weighted images clearly demonstrates surrounding vasogenic oedema in the brain parenchyma Fig. 16 Fig. 18
- T1+ gadolinium can show thickening and marked enhancement of the meninges Fig. 16 Fig. 18 Fig. 21
- Typically,
lesions show hypercellularity and diffusion restriction
GLIOSARCOMA
Background:
- Rare,
WHO grade IV tumour, described as a morphological variant of glioblastoma.
- The tumour contains both glial and mesenchymal (sarcomatous) components.
- It usually presents in the adult population,
the peak incidence being around the 6th decade
- The tumour can occur de novo,
be related to a previous glioblastoma or at the site of previous cranial irradiation.
- Known to metastasize to extracranial sites.
- Similar to glioblastoma,
gliosarcoma carries a dismal prognosis.
Fig. 23
- Treatment is surgical with adjuvant radiotherapy,
though mean survival following this is less than a year.
Imaging:
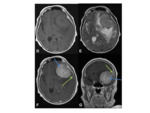
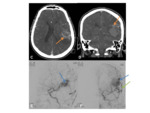
- Radiologically,
gliosarcomas appear similar to glioblastomas,
but can also be well circumscribed,
particularly if there is a high sarcomatous component,
and can then resemble a meningoma.
Fig. 25
- They have a broad attachment to the underlying dura,
with possible direct invasion,
or associated dural thickening
- There is often surrounding vasogenic oedema Fig. 26
•CT:
- Sharply defined due to the sarcomatous component,
round or lobulated hyperdense solid mass
- Homogeneous enhancement
- Surrounding peritumoural oedema
•MRI:
- T1: hypointense,
heterogeneous
- T2: internal heterogeneity due to internal necrosis and haemorrhage
- T1+Gd- thick irregular rim enhancement
•Angiography
- Mixed dural and pial vascular supply.
- Other findings include irregular angioneogenesis,
prominent vascular stain with well-defined tumour margins and early cortical venous drainage.
Fig. 24
PLEOMORPHIC XANTHOASTROCYTOMA (PXA)
Background:
- Rare,
low-grade astrocytoma (WHO grade II)
- Typically found in young patients
- Clinically can present as temporal lobe epilepsy
Imaging:
- Majority of tumours are based supratentorially,
centred upon the cortex Fig. 27 Fig. 28
- Due to its peripheral location,
the lesions abut,
and occasionally involve the overlying meninges.
A reactive dural tail can occasionally be seen.
- PXAs are low-growing tumours,
with scalloping of the adjacent bone,
and minimal surrounding vasogenic oedema for the size of the tumour.
Fig. 29 Fig. 30
- The lesions are predominantly cystic,
and there is avid contrast enhancement of any soft-tissue component
- Calcifications are rare.
•CT
- PXAs are typically well-delineated,
hypodense-cystic,
or isodense lesions.
- Bone window review can demonstrate scalloping of the overlying bone.
•MRI
- T1- the cystic component is low signal,
and any solid areas tend to be either isointense or hypointense compared to grey matter
- T1+Gd- solid areas will demonstrate avid enhancement
- T2- the cystic component is high signal.
Solid areas are isointense or hypointense to grey matter.
•Angiography
- Despite solid components demonstrating vivid enhancement,
PXAs are avascular on angiography.
DESMOPLASTIC INFANTILE GANGLIOGLIOMA
Background:
- Typically presents in young children
- WHO grade I tumours
- Can appear aggressive,
yet is relatively benign,
and has a good prognosis.
•Imaging
- Large solid-cystic tumours Fig. 31
- The peripheral solid component abuts and is attached to the meninges Fig. 32
- Dural involvement prominent
- Can be multiple
•CT
- Solid component tends to be slightly hyperdense,
and is located peripherally along the cortical margin of the mass
- Following contrst,
there is intense enhancement of the solid component
- A dural tail may occasionally be seen.
- May demonstrate calcifications
•MRI
- T1- isointense to brain parenchyma
- T2- isointense to brain parenchyma
- T1+Gd- enhancement of the solid component
METASTATIC DISEASE
Background:
- Direct extension from skull metastases or by haematogeneous spread
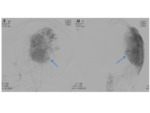
- Most common from primary tumours of the breast Fig. 33 or prostate,
as well as adenocarcinoma of the lung and renal cell carcinoma.
Other primaries include melanoma,
lymphoma and leukaemia.
In the paediatric cohort,
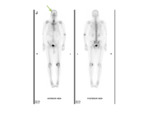
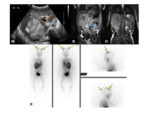
neuroblastoma metastases Fig. 51 ,
as well as haematological involvement are common findings Fig. 53 .
- There can also be seeding from a brain primary such as glioblastoma,
medulloblastoma,
germinoma and choroid plexus tumours.
- If the patient’s primary disease is controlled,
and there is a single,
discrete dural metastasis,
resection is often a consideration.
Imaging:
- Can mimic meningioma with a dural tail if the lesion is discrete
- Leptomeningeal carcinomatosis Fig. 34 is best demonstrated using contrast enhanced modalities,
where leptomeningeal disease is shown as marked enhancement scattered over the cerebral or cerebellar surface in a “sugar-coated” manner.
T1+gadolinium is the modality of choice,
though contrast-enhanced CT can also show similar appearances.
- Leptomeningeal carcinomatosis can cause obstructive hydrocephalus
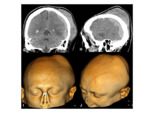
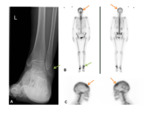
- Bone lesions that involve the meninges can either be primary- such as an Ewing's sarcoma Fig. 35 Fig. 36 Fig. 37 Fig. 38 Fig. 39 or a rhabdomyosarcoma Fig. 40 Fig. 41.
Metastases to the calvarium can have an extra-axial component and can abut/involve the dura.
Cases presented include plasmacytoma Fig. 42 Fig. 43 Fig. 44 Fig. 45,
thyroid metastasis,
Fig. 46 Fig. 47 Fig. 48 Fig. 49 Fig. 50,
neuroblastoma metastases Fig. 51 Fig. 52 and bony infiltration from acute lymphoblastic leukaemia Fig. 53.