The three “key questions” the radiologist have to answer…
- Is it hemorrhage or ischemic stroke? -> Non-enhanced CT
- Is there a large vessel occlusion? -> CT angiography
- Is there salvageable tissue? -> clinical picture,
CT perfusion
1. NONENHANCED CT
1.1 PROTOCOL:
-5mm section thickness parallel to the inferior orbitomeatal line,
from skull base to vertex.
-120 kVp,
400 mAs.
1.2 PURPOSE:
1.
Detection of hemorrhage = the key role of nonenhanced CT
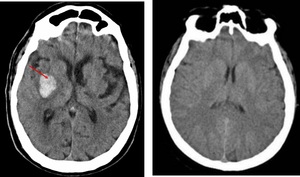
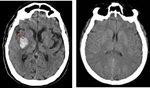
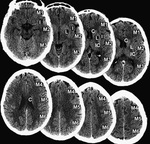
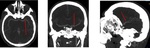
Fig. 4: Non-contrast CT. Hemorrhage stroke (hypertensive basal ganglia bleed).
Ischemic stroke (1h of onset of PACI symptoms). Normal CT.
2.
Detection of other possible mimics of stroke (Neoplasia,
infection,
malformation,
subdural collection..)
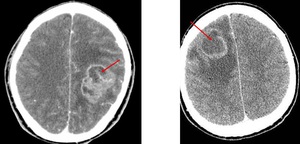
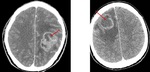
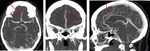
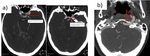
Fig. 5: Contrast-enhancement CT. Left frontoparietal glioblastoma multiforme.
Contrast-enhancement CT. Left frontal brain abscess.
3.
Detection of ischemic signs of established infarction. (low sensitivity: 25% in the first 3 hours,
50-60% in the first 6 hours).
- Cortical-subcortical hypoattenuating area within a vascular territory
- Cortical sulcal effacement
- Loss of the insular ribbon
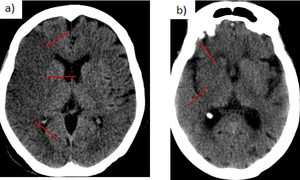
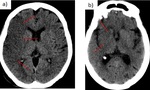
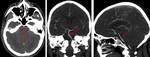
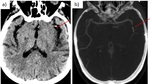
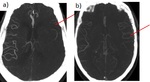
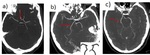
Fig. 6: a) Loss of gray white mater differentiation with diffuse hypoattenuation involving right MCA territory. Cortical sulcal effacement. b) Obscuration of right caudate and lentiform nucleus. Insular ribbon sign.
- Hyperdensity of a large vessel (“hyper-attenuating MCA sign” or “dot sign” in an M2 branch). False positive: increased hematocrit,
wall calcifications,
polycythemia,
or arterial dolichoectasia.
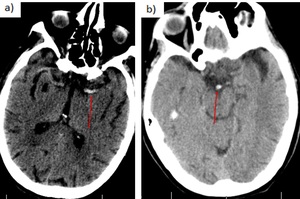
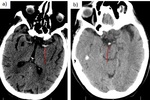
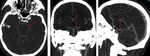
Fig. 7: Hyperdensity of a large vessel: a) Dense MCA sign (segment M1 of left MCA). b) Dense basilar sign.
4.
To assess the ASPECTS (Alberta Stroke Program Early CT Score).
It is a 10-point quantitative topographic CT scan score used in patients with MCA stroke.
It has also been adapted for the posterior circulation.
A normal CT receives an ASPECTS of 10 points.
An ASPECTS ≤ 7 predicts a worse functional outcome at 3 months as well as symptomatic hemorrhage.
CAUTION = ASPECTS is less useful if there are chronic brain infarct or microangiopathic changes.
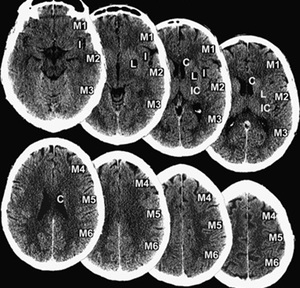
Fig. 8: ASPECTS. 1 point is deducted from the initial score of 10 for every region involved. Subcortical structures are allotted 3 points: C (caudate), L (lentiform nucleus), and IC (internal capsule); MCA cortex is allotted 7 points: insular cortex, M1, M2, M3, M4, M5 and M6) .
References: Al-Ajlan et al.(2017). Multimodality CT based imaging to determine clot characteristics and recanalization with intravenous tPA in patients with acute ischemic stroke. Neurovascular Imaging
1.3 STRUCTURED REPORT:
1- With a standard window (70 WW,
35 WL) we assess:
-Intracranial hemorrhage: yes/no.
-Midline shift: yes/no
-Basal cisterns well visualized: yes/no
-Ventricular system normal for age: yes/no
-Other finding (chronic brain infarct,
microangiopathic changes,
space occupying intracranial lesions...).
2- With a second narrower setting (35 WW - 35 WL; or 40 WW - 40 WL) we try to demonstrate subtle abnormalities that suggest ischemia: Cortical-subcortical hypoattenuating area within a vascular territory,
cortical sulcal effacement,
loss of the insular ribbon,
and hyperdensity of a large vessel.
3- With a soft tissue window (360 WW,
60 WL):
-orbits
-paranasal sinuses and mastoid air cells
-soft tissues
4- With a bone window (2000 WW,
800 WL) we evaluate osseus structures.
2.
CT ANGIOGRAPHY (CTA)
2.1 PROTOCOL:
-From the aortic arch up to the vertex.
-120 kVp,
250 mAs.
80 mL of contrast material intravenously at a flow rate of 4 mL/sec with a bolus tracking method.
Slice thickness of 0.8 mm.
2.2 ATLAS OF NORMAL VASCULAR ANATOMY
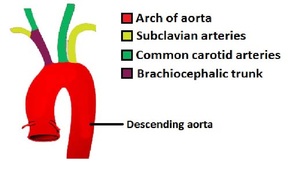
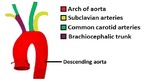
Fig. 9: Schematic of the Aortic arch and major branches.
References: https://teachmeanatomy.info/abdomen/vasculature/arteries/aorta/
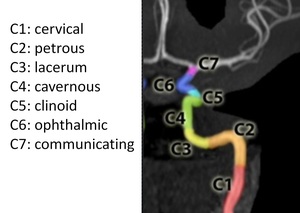
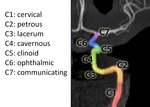
Fig. 10: Internal carotid artery. Segments.
References: Dr Behrang Amini, From the case: Carotid artery segments (diagram). Radiopaedia, 2009.
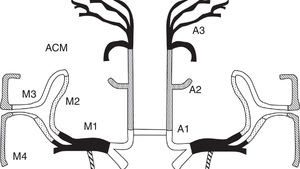
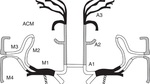
Fig. 11: Segments of MCA and ACA
References: M. Mendigaña Ramos et al. (2015). Evaluación vascular en el código ictus: papel de la angio-tomografía computarizada. Radiología.
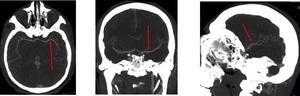
Fig. 12: Medial cerebral artery (MCA). Axial, coronal, sagittal.
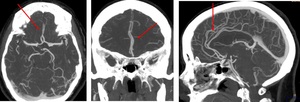
Fig. 13: Anterior cerebral artery (ACA). Axial, coronal, sagittal.
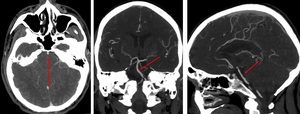
Fig. 14: Basilar artery. Axial, coronal, sagittal.
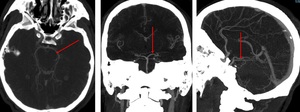
Fig. 15: Posterior cerebral artery (PCA). Axial, coronal, sagittal.
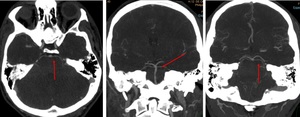
Fig. 16: Superior cerebellar artery. Axial, coronal, sagittal.
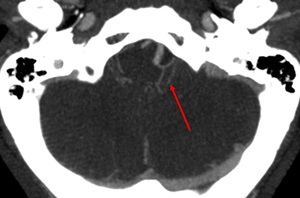
Fig. 17: Posteroinferior cerebellar artery (PICA). Axial.
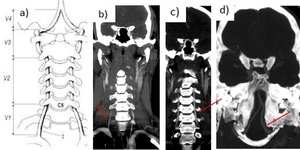
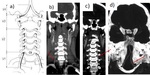
Fig. 18: Vertebral arteries. a) Segments. V1 (preforaminal, from its origin in the subclavia artery to C6 vertebra). V2 (foraminal, from C6 to C2). V3 (atlantic. C2-C1). V4 (intracranial).
b) V1 segment. c) V2 segment. d) V4 segment.
2.2 MAIN ROLE OF CTA:
The main role is to define the occlusion site and grade of collateral blood flow.
It is useful to characterize carotid atherosclerotic disease or to depict arterial dissection too.
1. Evaluation of intracranial and extracranial circulation
PRIMARY GOAL = To identify and locate vascular occlusion to assess the potential benefit of a fibrinolytic or interventional endovascular treatment.
It is more precise for the detection of proximal thrombus.
1.1.
OCCLUSION: It is the most common cause of ischemic stroke.
a) M1 segment MCA occlusion:
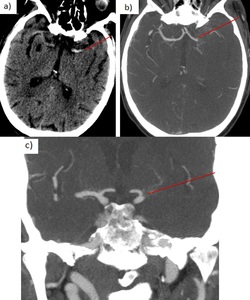
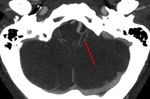
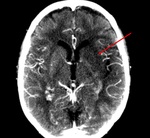
Fig. 19: 78 yo man with right hemiparesis. Ischemic stroke. a) Non-contrast CT: hyperdense left MCA sign.
b) Axial CTA: occlusion left M1 MCA. Grade 1 leptomeningeal collaterals (<50%). c) Coronal CTA: occlusion left M1 MCA.
b) M2 segment MCA occlusion:
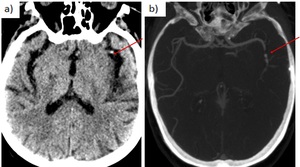
Fig. 20: a) 72 yo man with right hemiparesis. Ischemic stroke. Axial non-contrast CT showing MCA dot sign (left M2 segment in the Sylvian fissure is hyperdense).
b) 79 yo woman with Broca’s aphasia. Axial CTA: short occlusion left M2 MCA.
c) ACA,
MCA and Anterior communicating artery occlusion:
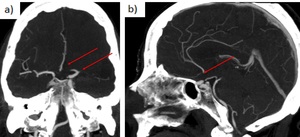
Fig. 21: 67 yo woman with right hemiparesis and global aphasia. Occlusion ACA, MCA and anterior communicating artery. a) coronal CTA. B) sagittal CTA.
d) Tandem occlusion:
Simultaneous extracranial cervical ICA severe stenosis/complete occlusion + ipsilateral large vessel intracranial occlusion (most common ICA+MCA).
High recanalization rates are possible using endovascular therapy.
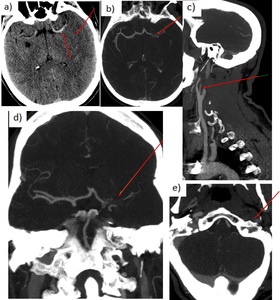
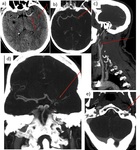
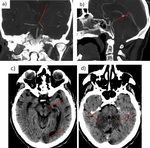
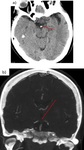
Fig. 22: 45 yo man with tandem occlusion (left ICA+MCA). a) Dense MCA sign (segment M1 of left MCA), diffuse hypoattenuation involving left MCA territory and cortical sulcal effacement. b) Axial CTA: occlusion left M1. c) Sagittal CTA: complete occlusion of the left ICA 2 cm from its origin. d) Coronal CTA: occlusion left M1. e) Axial CTA: lack of contrast enhancement in petrous segment left ICA.
e) Carotid T- occlusion:
A T-type ICA occlusion is defined when the terminal segment (C7) of the ICA is not visible,
and there is occlusion of the M1 segment in the middle cerebral artery and the ipsilateral A1 segment in the anterior cerebral artery.
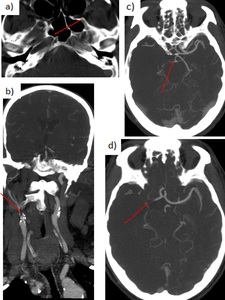
Fig. 23: 58 yo man with acute left-sided hemiplegia. Right carotid T occlusion. a-b) axial and coronal CTA: complete occlusion right ICA 1 cm from its origin. c-d) axial CTA: occlusion of the right A1 segment, M1 segment, and PCA (fetal origin). Poor collateral flow.
f) Top of the basilar (rostral brainstem infarction):
Thromboembolic occlusion of the tip of the basilar artery.
This results in bilateral thalamic ischemia due to occlusion of perforator vessels.
TRICK: patients with normal non-contrast CT and loss of consciousness/coma=look for Top of the basilar.
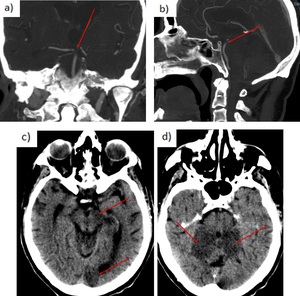
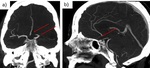
Fig. 24: a - b) Coronal and sagittal CTA: filling defect at the top of the basilar artery and in the left P1 segment. Right PCA and both superior cerebellar arteries are well opacified with contrast.
c - d) Non-contrast CT 1 month later: low density (infarction) involving the left midbrain, the inferior portion of the temporal lobe and occipital lobe, and cerebellum.
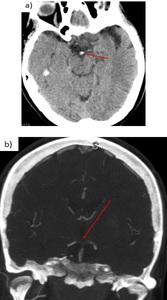
Fig. 25: 66 yo woman. She falls down and enters a coma. a) The top of the basilar artery is markedly hyperdense. b) CTA confirms the presence of a filling defect at the top of the basilar artery and both P1 segments of the PCA.
1.2 CRANIOCERVICAL ARTERY DISSECTION:
Common cause of stroke in young and middle-aged patients.
Anticoagulation is the most frequent therapy.
a)Carotid artery dissection (68%):
-Location: cervical part of the artery distal to the carotid bulb and tends not to extend beyond its entry into the petrous portion.
-Findings: FLAME-LIKE SHAPE OCCLUSION (typically 2-3 cm above the carotid bifurcation),
narrow eccentric lumen with increase of the external diameter of the artery (50%),
arterial occlusion (30%),
intimal flap (10%,
pathognomonic)...
b)Vertebral artery dissection (27%):
-Location: is predominantly located in the pars transversaria (V2) or the atlas loop (V3).
-Findings: increased external diameter and crescent-shaped mural thickening and a narrow eccentric lumen,
irregularity/abnormal vessel contour...
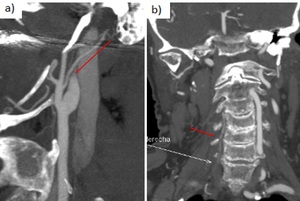
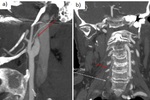
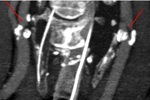
Fig. 26: a) Dissection of the left cervical ICA in a 48 yo woman. Sagittal CTA shows the typical flame-like shape occlusion 3 cm above the carotid bifurcation.
b) Dissection of the right vertebral artery in a 63 yo man. Coronal CTA shows an irregularity of the right vertebral artery from its origin.
2. Assessment of leptomeningeal collaterals
It is related to the clinical outcome,
the infarcted tissue volume and the response to endovascular treatment.
A favorable pattern of leptomeningeal collaterals as visualized on CTA correlates with improved functional outcomes in acute ischemic stroke patients.
Grades
-Grade 0: no collaterals visible to ischemic site
-Grade 1: <50% collaterals
-Grade 2: >50 and <100%
-Grade 3: 100% collaterals
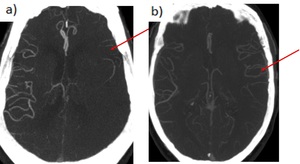
Fig. 27: Leptomeningeal collaterals. Grade 1 (< 50%). b) Grade 2 (> 50%)
3. CTA-SI (computed tomography angiography source image):
HOW IS IT DONE?: We increase the slice-thickness without MIP,
and narrow the window. In this case the hypodensity is due to a decrease in blood cerebral perfusion (whereas hypodensity on non-contrast CT is due to cytotoxic edema).
USEFULNESS = Performing ASPECTS on CTA-SI increases the sensitivity and accuracy for the detection of early ischemic changes,
improves predictions for final infarct size,
and may indicate the hemorrhage transformation risk with recanalizing therapies. CTA-SI hypoattenuation volumes is correlated with concurrent DWI hyperintense volumes.
CAUTION = There is a tendency with the current fasters scanners to overestimate the final infarct size on CTASI,
whereas non-contrast CT tends to underestimate it (the CTASI parenchymal abnormality includes ischemic penumbra and thus overestimates final infarct size).
This could result in inappropriate exclusion of patients from thrombolysis or thrombectomy.
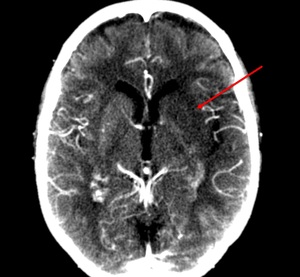
Fig. 28: CTASI shows hypoattenuating left MCA territory (caudate and lentiform nucleus, internal capsule, insular cortex).
4. Assessment of atherosclerosis.
4.1 TYPES OF ATHEROMATOUS PLAQUES: noncalcified,
mixed and calcified plaques.
4.2 PERCENT DEGREE OF STENOSIS: no significant stenosis (<50%),
significant stenosis (>50%),
severe stenosis (>70%),
critical/near occlusion (>90%).
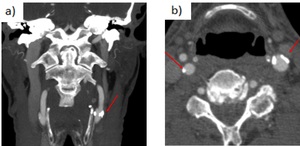
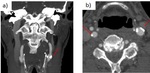
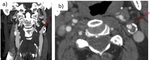
Fig. 29: Carotid bulbs calcium plaques with no significant stenosis (<50%). a) Coronal CTA. b) Axial CTA.
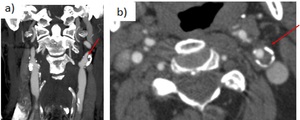
Fig. 30: Mixed atheromatous plaques with left ICA severe stenosis (>70%). a) Coronal CTA. b) Axial CTA.
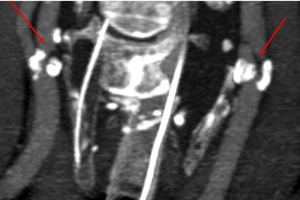
Fig. 31: Near-occlusion left ICA stenosis. Severe right ICA stenosis. Coronal CTA.
5. Other findings: Vascular anatomical variants.
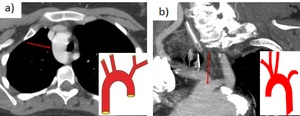
Fig. 32: a) Right aortic arch.
b) Bovine aortic arch, most common variant of the aortic arch = Inominate artery shares a common origin with the left common carotid artery.
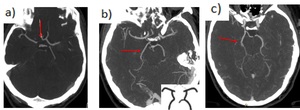
Fig. 33: a) Absent A1 segment of ACA.
b) Fetal posterior cerebral artery (estimated to occur in 20%, most common unilateral). P1 is absent. The posterior communicating artery supplies the bulk of the blood to the PCA. If bilateral, the basilar artery is significantly smaller than normal.
c) Partial fetal PCA. Posterior communicating artery is larger than P1.
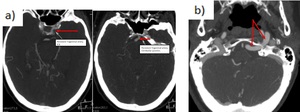
Fig. 34: a) Persistent primitive trigeminal artery. It is one of the persistent carotid-vertebrobasilar anastomoses (it is a junction between cavernous ICA and basilar artery). It is present in 0,1-0,5%. b) Persistent hypoglossal artery. It is a persistent carotid-vertebrobasilar anastomoses, the second in frequency (0,02-0,1%). It arises from the distal cervical ICA (between C1-C3). After passing through an enlarged hypoglossal canal, it joins the basilar artery inferiorly.
2.3 A SYSTEMATIC APPROACH TO ANGIO- CT READING
PRIMARY INTERPRETATION:
- WHERE = We inform the neurologist verbally in the CT room.
- OBJECTIVE= To identify large vessel occlusion susceptible to mechanical thrombectomy (M1,
proximal M2,
basilar,
carotid).
Assessment of leptomeningeal collaterals.
SECONDARY INTERPRETATION:
- WHERE = at our work station.
- OBJECTIVE = to make an accurately report
① We analyze the source images in the axial plane (not processed).
② We perform MIP reconstructions,
increasing the section thickness to 4 mm,
what helps to detect the occlusion.
③ We perform coronal and sagittal multiplanar (MPR) reconstructions.
④ Volumetric 3D reconstruction may be helpful.
STRUCTURED REPORT:
① MAIN FINDING:
-Is there a large vessel obstruction? yes/no
-Location:_
-Is it susceptible to mechanical thrombectomy?
-Thrombus length/Clot size: _mm
-Leptomeningeal collaterals: grade 0,
1,
2,
3.
-CTA-SI
②Origin of the supra-aortic trunks: normal,
or with anatomical variants (bovine aortic arch,
right arch ...)
③ Common and internal carotid artery: occlusion,
dissection,
and stenosis / atheromatosis.
④ Circle of Willis: occlusion,
dissection,
aneurysms.
⑤ Vertebrobasilar system: occlusion,
dissection,
stenosis / atheromatosis.
⑥ Vascular anatomical variants.
⑦ Other findings: pulmonary apex,
neck,
bone ...
3.
CT PERFUSION (CTP):
3.1 PROTOCOL:
-Contrast material 40 mL at a rate of 4 mL/sec,
prescan delay 6 seg.
-Cine mode,
centered in basal ganglia.
Slice thickness 5 mm,
kV 80,
mA 150,
angle parallel to orbital roof,
DFOV 25 cm.
-It could be performed before or after the CTA according to Clinical Guidelines.
We recommend performing it AFTER CTA (on CTA we are able to see if there is a delay on contrast distribution due to a cardiac failure,
and if so,
we increase the CTP-prescan delay 5s more than usual).
3.2 MAIN GOAL = TO ASSESS THE EXTENT OF SALVAGEABLE TISSUE.
It enables differentiation of salvageable ischemic brain tissue (PENUMBRA) from irrevocably damaged infarcted brain (INFARCT CORE).
-MAIN INDICATIONS = wake up stroke (unknown onset),
and time-limit stroke (4-5 h) with favorable non-contrast CT.
3.3 HOW IS IT DONE? = The parameters typically used in determining these two areas are:
-MEAN TRANSIT TIME (MTT) is the time between the arterial inflow and the venous outflow.
The most sensitive in early stages.
-Time to peak (TTP) is the delay between the first arrival of contrast material intracranially and the time at which the contrast reaches its peak concentration in a given region of parenchyma.
-CEREBRAL BLOOD FLOW (CBF).
Normal = 50-60 ml/min/100 g.
-CEREBRAL BLOOD VOLUME (CBV) is the volume of blood available per unit of brain tissue.
Normal = 4-6 ml/100g.
-PHISIOPATHOLOGY: Cerebral blood flow is equipped with an autoregulatory mechanism.
When the cerebral blood flow decreases, it tries to compensate by increasing the blood pressure and inducing vasodilatation (PENUMBRA).
However,
if the blood flow decreases so much that it falls below a critical level,
infarction results (INFARCT CORE).
3.4 INTERPRETATION
Postprocessing of the data is done by using specialized software that generates color perfusion maps.
-1º MTT values are evaluated =if the MTT is raised as compared to the normal side,
ischemia/infarction is present.
-2º to differentiate ischemic and infarcted tissues,
CBF and CBV values are compared.
If CBF is reduced and CBV is normal or slightly reduced,
the tissue ischemia is likely to be reversible.
If CBF and CBV are markedly reduced or if TTP is not measurable,
the tissue may be infarcted.
Core: increased MTT/Tmax,
markedly decreased CBF,
markedly decreased CBV
Penumbra: increased MTT/Tmax,
moderately reduced CBF,
near normal or increased CBV
3.5 USEFULNESS = there are discrepancies between different studies on the usefulness of CTP.
-CTP may be useful when assessing a patient for treatment (thrombolysis or clot retrieval) = patient with a small core and a large penumbra is most likely to benefit from reperfusion therapies.
-Cortical rCBV was found to be significantly lower in patients with poor clinical outcomes.
-Cine mode allows us to assess the collateral grade.
3.6 PAY ATTENTION/POTENTIAL PITFALLS
-In patients with decrease blood flow (congestive heart failure,
atrial fibrillation,
severe extracranial arterial stenosis),
there is a delay in passage of the contrast that can lead to overestimated MTT and underestimated CBF= core infarct may be overestimated.
-Perfusion protocols are centered upon the basal ganglia/supraganglionic level.
This excludes a large volume of brain.
-Small infarcts such as lacunar infarcts,
could be not visualized on perfusion maps due to their low resolution = false negative result.
-In cases of seizures,
the ictal region shows hyperperfusion,
which may lead to an interpretation of hypoperfusion in the contralateral hemisphere mimicking infarct.
- Appropriate selection of arterial input and venous output functions from the source images is critical to accurate perfusion representation and to avoid errors.
We typically obtain the arterial input from the A2 segment of the ACA and the venous output routinely from a dural venous sinus.
-MRI (DWI sequence) is more sensitive to the early parenchymal changes than CTP,
but it is less accesible.
3.7 OUR EXPERIENCE = CTP is another valuable imaging tool that helps neurologist and neurointerventionists for identifying patients amenable for intervention. There must not be considered as the only tool for determine the treatment,
and radiologist must analyze it with cautions.
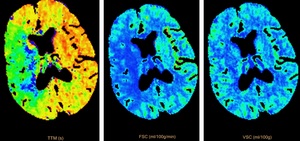
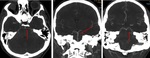
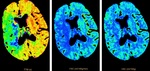
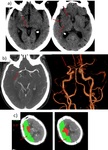
Fig. 35: CTP imaging study showing an acute stroke of the right MCA territory with salvageable penumbra. The first and second images indicate increased TTP and decreased CBF respectively. The third image indicates no significant CBV loss. This patient underwent thrombectomy on the basis of CTP imaging findings and NIHSS score.
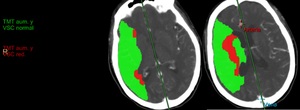
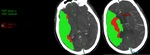
Fig. 36: Same patient. Larger region of right MCA territory penumbra (represented by the green zone).
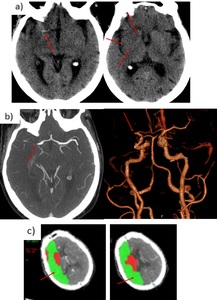
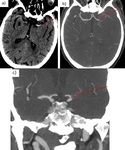
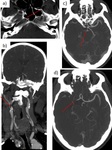
Fig. 37: 72 yo man. Left hemiparesis. Right M1 occlusion.
a)Non-contrast CT. Hyperdense M1 segment of right MCA. Hypoattenuation of the ipsilateral insular cortex and basal ganglia.
b) Axial CTA and 3D reconstruction. Complete occlusion of the right M1 segment just distal to the bifurcation.
c) CTP. Established infarct core in the right MCA territory, with a larger surrounding ischemic penumbra.