Patients were 46 males and 10 females (aged 48-84,
mean age 68 years).
All were smokers and Median Pack-year was 48.2 (range 6-120 p/y).
Tumour stage was IIIb in 16/56 (28.6%) and IV in 40/56 (71.4%).
Tumour histology was adenocarcinoma in 27/56 (48.2%) and squamous-cell carcinoma in 29/56 (51.8%).
Using iRECIST criteria,
radiological analysis of CT scans demonstrated:
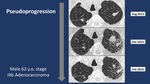
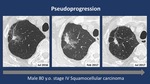
- Pseudoprogression pattern in 3/56 (5.3%)
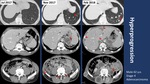
- Hyperprogression pattern in 5/56 (8.9%)
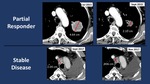
- Other Responders (OR):
- CR: in 0 patients
- PR: in 6/56 (10.7%)
- SD: in 17/56 (30.3%)
- PD: in 25/56 (44.6%)
We did not find any statistically significant difference in patients age (> and < 65 years),
tumor stage (IIIb/IV) and tumor hystology (adenocarcinoma/squamous cell carcinoma) between the three goups of different radiological responders.
Althought we didn't find statistically significant differences between pseudoprogression and Hyperprogression group,
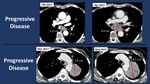
we found that most of Pseudoprogression patients had stage IV adenocarcinoma (Figures 1-2) while most of Hyperesponders were stage IV squamous cell carcinoma.
The mean age of patients with Pseudoprogression was 70.3 years while patients with Hyperprogression had a mean age of 53.5 years with the youngest one aged only 42 (Figure 3).
This result differs from the most recent literature data,
where Hyperprogression is correlate with elder age [10].
Patients with pseudo-progressive disease were re-classified as PR in the 3rd and 4th follow up CT.
This data suggest that clinicians should feel confident that patients with apparent worsening of the disease in the early post-treatment period do not necessarily have a real poor response to Nivolumab.
In these cases,
swithing off Nivolumab should be based not only on CT images but also on clinical data (for examples worsening of performance status or appearance of side effects).
Hyperprogression global incidence in our patients was of 8.9%; this data is in accordance with the work of Champiat et al.
where up to 9% of patients threated with Nivolumab developed hyperprogression [8].
Our study is concordant to literature data regarding the lack of significant difference in tumor stage and histology between the three groups of responders as radiographic pattern of treatment response to Nivolumab therapy in advanced NSCLC is heterogeneous,
especially in the early post-treatment surveillance period [11].
Globally,
OR (Figures 4-5) represented the 85,8% of patterns with a slight prevalence of PD over SD.