Splenic implants have irregular architecture with a poorly formed capsule[3],
are small in size (< 3 cm)[11] and generally asymptomatic[4].
They can be solitary or multiple (up to 400 nodules)[11] and may slowly grow over time (up to 12 cm)[10].
For these characteristics,
the differential diagnosis includes abdominal malignancies,
metastatic disease,
lymphoma and carcinomatosis[2].
A definitive preoperative diagnosis requires a high index of suspicion and should be established cautiously: a detailed medical history,
as well as correlation with physical examination and laboratory tests,
is necessary[3; 6; 10].
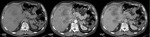
Pancreatic splenosis (Figure 2) could be difficult to recognise.
Splenic nodules in the pancreas can represent intrapancreatic accessory spleen or splenosis.
Both these entities must be differentiated from pancreatic malignancies,
specifically,
pancreatic neuroendocrine (islet) tumour,
or metastatic disease.
On computed tomography (CT),
the diagnosis of intrapancreatic splenosis should be considered when a well-defined nodule with enhancement paralleling that of the spleen is identified in the pancreatic tail region,
particularly along the dorsal surface[12].
Rare cases of splenosis in the head of the pancreas have also been described[13],
therefore,
adenocarcinoma should also be considered.
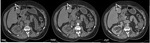
In intrahepatic splenosis,
the imaging of splenosis is almost indistinguishable from hepatic neoplasms like hepatic cell carcinoma (HCC)[9].
Presumably,
for a splenic nodule to implant in the liver there must be a simultaneous rupture of the liver capsule at the time of trauma[4].
Because of these,
intra-hepatic splenic nodules are usually in subcapsular position and multiple [4].
(Figure 3)
Splenosis can also mimic renal masses when found near or adherent to the surface of the kidneys[12].
In a case report]14],
investigators described a large splenic nodule adherent to a kidney that even developed a cystic component,
which is hypothesized to be the result of central necrosis secondary to inadequate blood supply from surrounding tissues[12; 14].
Splenic nodules are often first seen on ultrasound imaging[12].
The sonographic findings are not specific: they appear as solid,
round or oval-shaped soft-tissue masses with homogeneous hypoechoic echotexture[7].
Looking closely at the differential diagnosis with neoplastic diseases,
splenic nodules will have the same CT-characteristics of a parenchymatous structure,
but will not show necrosis or calcifications.
On CT,
in the arterial-dominant phase (HAP),
splenic nodules will show mild enhancement,
but on portal venous phase (PVP),
there will not show washout,
whereas,
on delayed phase (DP),
the attenuation will decrease[10-12].
The usual behaviour of a malignant lesion is either heterogeneous or ring enhancement on HAP and/or washout on later phases,
but malignancies like HCC,
may only show washout.
In these cases,
establishing a definitive diagnosis can be difficult.
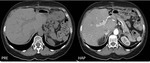
A patient’s previous cross sectional imaging examination can be helpful,
because if an unknown lesion shows size stability over time,
it is more likely to be a benign lesion[3].
(Figure 4)
Magnetic Resonance (MRI) can further characterize splenosis.
Normal adult spleen is hypointense on T1-weighted imaging and hyperintense on T2- weighted imaging[15].
Likewise,
splenic implants demonstrate intermediate to low signal intensity on T1-weighted images,
intermediate to high signal intensity on T2-weighted images and a heterogeneous enhancement on HAP which later becomes homogeneous[3,10].
In-phase (IP) and out-of-phase sequences (OOP) can help rule out the presence of fat in splenic nodules.
Malignancies,
instead,
might contain it.
On Diffusion-weighted images (DWI),
spleen has high signal on high b sequences,
like splenosis and malignancies.
An Apparent diffusion coefficient (ADC) map will confirm the DWI restriction only on malignant lesions.
If a diagnostic dilemma persists,
fine needle aspiration is the gold standard,
but it is an invasive procedure not immune to complications[2],
so,
in these cases,
nuclear scintigraphy is recommended[2;3;9-10] Tc-99m sulphur colloid scintigraphy is classically used.
The tracer is sequestered in the reticuloendothelial system and easily detects heterotopic splenic tissue[10].
Functional imaging of the spleen may also be obtained with denaturated blood cell scintigraphy[6].
Labeled Tc-99m red blood cell studies may be particularly useful if there is overlap of liver and spleen at 99mTc sulfur colloid imaging.
Single positron emission computed tomography allows a direct correlation with other imaging techniques[6;12].