Keywords:
Outcomes, Outcomes analysis, Ultrasound, Musculoskeletal joint, Interventional non-vascular
Authors:
C. Gianneramo1, F. Bruno1, I. Capretti2, M. V. M. Micelli1, S. Mariani1, F. Arrigoni1, L. Zugaro1, A. Barile1, C. Masciocchi1; 1L'Aquila/IT, 2L' Aquila/IT
DOI:
10.26044/ecr2019/C-1326
Methods and materials
The study population consists of participants (male or female aged ≥40 years) with moderate-severe pain attributable to hip OA (mild- high grade of OA) and supported by Radiographic or MRI evidence of hip OA,
with symptom duration at least 6 weeks not responding to oral therapy [Fig.1].
Exclusion criteria were: hip pain due to other disorders (e.g.
rheumatoid arthritis); current or previous infection of the affected hip; anticoagulant therapy; any other contraindications to the use of our drugs.
Different clinical outcomes,
pain,
functional status were evaluated by using VAS scale (VAS) (subjective measure for acute and chronic pain from =0=no pain to 10=worst pain) and Harris Hip Score (HHS) clinician-based outcome measure consisting of a sum of four subscales (range from 0-100 with higher scores representing less dysfunction and better outcomes)[Fig.2].
The instrumental evaluation was performed using the Kellgren and Lawrence system.
We also evaluated the pain-free time-period by the assessment of the time period between 2 injections protocol in the same patient.
Each patient underwent 3 US-guided joint injections: the first injection of corticosteroids followed,
after 7 days,
by 2 injections of hyaluronic acid (HA) distanced of 15 days from each other.
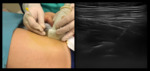
The intra-articular injections were performed in a sterile condition.
A linear transducer sterile covered was positioned to identify the femoral axis [Fig 3]; a 22-gauge spinal gauge was positioned laterally from femoral vessels in the capsular recess [Fig.4].