Aortic Dissection
Aortic dissection is the most common acute aortic emergency.
If left untreated it is often fatal.
Dissection occurs when there is a tear in the intima,
allowing blood to enter into the vessel wall.
This parietal blood extends longitudinally and circumferentially,
progressively detaching the intima from the media and creating an intima-media flap,
which separates the true lumen from the false intraparietal lumen.
The most common sites of the intima’s lesion are in the ascending aorta,
most often in the right wall,
and in the descending aorta,
between the origin of the left subclavian artery and the insertion of the arterial ligament.
Because they are more fixed,
these regions are subject to greater pressures and greater hydraulic stress,
being damaged more easily.
Aortic dissection may also result from an IMH or a PAU.
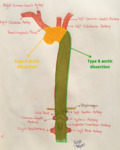
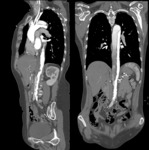
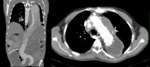
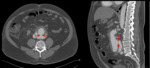
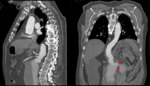
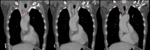
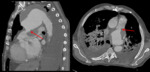
Aortic dissection is divided into two types by the Stanford classification (Fig. 3): in type A (about 60% of dissections),
dissection involves the ascending aorta and / or the crossa of the aorta,
regardless of the location of the lesion of the intima and its distal extent; in type B (about 40% of dissections),
the dissection is distal to the emergence of the left subclavian artery.
This classification is extremely important because it has both therapeutic and prognostic implications.
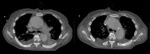
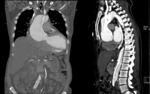
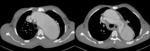
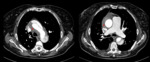
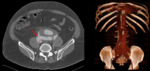
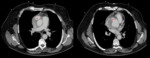
Type A dissections (Fig. 4 and Fig. 5) should be treated with emergent surgery because of the high risk of serious and potentially lethal complications: rupture into the pericardial cavity and development of cardiac tamponade; occlusion of the ostia of the coronary arteries or emerging branches of the aortic arch,
with consequent ischemia of their territories; and disruption of the aortic valve with possible acute aortic valve insufficiency.
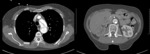
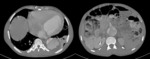
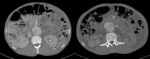
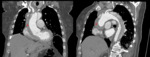
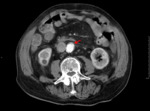
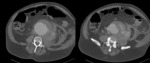
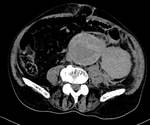
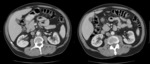
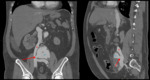
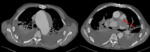
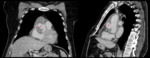
Type B dissections (Fig. 6 and Fig. 7) are usually treated by medical approach through blood pressure control,
along with tight clinical and imaging monitoring.
Surgery may be necessary if there is imaging evidence of parietal rupture,
refractory pain,
or rapid increase of aortic diameter.
For the diagnosis of aortic dissection,
images without intravenous contrast of at least the thoracic aorta should initially be acquired to increase the likelihood of detecting haematomas in the false lumen or intra-mural haemorrhage,
which after contrast injection may be confused with mural thrombi.
Mediastinal haematomas,
hemopericardium and hemothorax are also best characterized in non-contrast studies.
Subsequently,
contrast-enhanced images should be acquired,
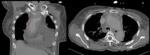
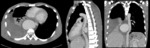
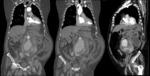
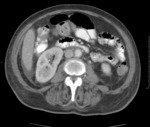
since the definitive diagnostic image of aortic dissection is the demonstration of two contrast-filled lumens,
separated from one another by an intimal flap (Fig. 8,
Fig. 9,
Fig. 10 and Fig. 11).
The true lumen presents an intense precocious opacification,
continues with the lumen of the non-dissected aorta and is often smaller.
The false lumen is located typically in the external curvature of the aorta,
it’s larger and has a later enhancement due to slower flow.
In the false lumen we may have the beak sign (spread of a haematoma in the false lumen) or see cobwebs,
pieces of the middle layer that are plucked into the false lumen as the dissection spreads.
There are other less specific imaging findings which may also be useful.
In the pre-contrast images,
we can see: internal displacement of calcifications of the intima; dimensional enlargement of a segment of the aorta; hyperdense material in the false lumen,
if acute thrombosis ensues; intimal flap visible as a linear structure slightly hyperdense relative to the blood surrounding it,
if there is concomitant anemia; and bleeding secondary to aortic rupture.
In post-contrast images,
is possible to see: also false lumen thrombosis; fenestrations of the intima consistent with the tears,
whose extremities usually point to the false lumen; extension of the dissection to the various vessel branches; ischemia / infarction of the organs vascularized by the vessels branches; and extravasation of contrast,
indicative of rupture of the aortic wall.
Intra-Mural Hematoma (IMH)
IMH is a spontaneous haemorrhage of the middle layer of the aortic wall.
It may occur as a primary event in hypertension,
due to the rupture of the vasa vasorum,
or secondary to a PAU that perforates the internal elastic lamina and reaches the media,
with consequent bleeding.
IMH can undergo longitudinal and radial extension,
weakening the vascular wall and predisposing evolution to dissection,
aortic rupture or aneurysm formation.
Type A IMH (the same anatomical distribution of type A aortic dissection) should also be treated surgically because of the high risk of serious and potentially lethal complications similar to those described above for type A dissections.
Type B IMH are usually treated by a medical approach through blood pressure control,
along with tight clinical and imaging monitoring.
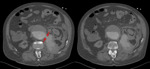
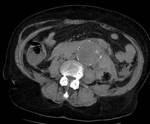
At CT scan,
IMH presents as an area of parietal thickening of the aorta,
circular or in crescent,
which is spontaneously hyperdense compared to the vascular lumen in non-contrast images.
In contrast-enhanced images,
IMH can be confused with atherosclerosis or mural thrombi,
although IMH creates a smooth interface with the contrasting aortic lumen.
The presence of intimal calcifications displaced into the vessel may help in the differential diagnosis between IMH and mural thrombus,
in the contrast studies.
Slight peri-aortic stranding may be observed in probable relation with minimal mediastinal hemorrhage,
not necessarily meaning rupture of the aorta (Fig. 12,
Fig. 13 and Fig. 14).
Further development of new projections of the IMH can be observed,
appearing as globular contrast collections within the mural haematoma and communicating with the lumen.
When present,
they increase the likelihood of progression to dissection and aneurysm formation.
Penetrating Atherosclerotic Ulcer (PAU)
PAU is an atherosclerotic lesion that undergoes ulceration and penetrates the internal elastic lamina,
with consequent formation of a haematoma in the media layer of the aortic wall.
It occurs in patients with advanced/severe atherosclerosis.
The haematoma may undergo longitudinal extent,
such as in IMH,
staying confined to the middle layer,
or a communication between the aortic lumen and the haematoma at the base of the ulcer may be established,
which can progress to dissection.
In addition to these two possible complications,
there is also a parietal weakening of the aorta,
with a greater propensity for aneurysm formation,
pseudoaneurysm and transmural rupture.
PAU are more common in regions most affected by atherosclerosis: the descending thoracic aorta and the abdominal aorta,
where is usual the association with abdominal aortic aneurysms.
PAU are often multiple,
so the approach will depend primarily on the characteristics of the dominant or complicated ulcer.
Surgical treatment is indicated in the PAU located in the ascending aorta or the aortic crossa,
in the PAU with a diameter greater than 20 mm or depth greater than 10 mm,
in hemodynamically unstable patients or patients with persistent symptoms,
if there is aneurysm formation and evolution to dissection or type A IMH.
The remaining patients should be treated conservatively,
by controlling blood pressure.
At CT,
a PAU (Fig. 15 and Fig. 16) may give rise to the following imaging findings: a focal ulceration,
manifested by a eccentric collection of contrast on the wall of the aorta; elongated wide-mouth ulcers,
appearing as a focal dilatation of a segment of the aortic lumen; thickening and/or enhancement of the wall adjacent to a haematoma; an IMH,
easily proving the penetrating component of the ulcer; pseudointimal flap,
thick and irregular,
which corresponds to the margins of the ulcer and separates the aortic lumen from an elongated intra-mural haematoma.
Possible complications are aneurysm or pseudoaneurysm formation,
aortic dissection and transmural rupture.
Aortic Aneurysm (AA)
An AA is a permanent dilatation of the aortic wall: > 5 cm in the ascending thoracic aorta,
> 4 cm in the descending thoracic aorta and > 3 cm in the abdominal aorta.
A true aneurysm includes the 3 layers of the aortic wall and may be fusiform or saccular.
The main cause of AA is atherosclerosis,
usually forming fusiform aneurysms in the infra-renal abdominal aorta and in the proximal descending thoracic aorta.
Mycotic or infectious aneurysms are usually saccular,
being found more frequently in the abdominal aorta above the renal arteries and in the descending thoracic aorta.
AA are often asymptomatic and incidentally diagnosed (Fig. 17) ,
but may become symptomatic when they cause mass effect or when they complicate.
The most frequent and most lethal complication is AA rupture,
a true aortic emergence,
manifested by severe pain,
hypotension and shock.
Treatment is an emergent intervention,
surgical or endovascular.
In addition to aneurysm rupture,
emergent treatment is also indicated in the following situations: asymptomatic aneurysm of the descending thoracic aorta > 6 cm,
asymptomatic ascending aortic or abdominal aortic aneurysm > 5,5 cm,
symptomatic aneurysms and rapidly growing/expanding aneurysms during follow-up.
Other possible complications of AA include the formation of pseudoaneurysms,
formation of fistulas,
distal thromboembolism,
thrombotic occlusion of the emergence of its branches and infection.
At CT,
the aneurysms appear as dilatations of the aortic lumen,
and the walls may be thin or thickened,
by the presence of mural/intraluminal thrombi.
Concomitantly,
the detection of calcified atherosclerotic disease is frequent,
since atherosclerosis is the main risk factor for AA formation.
When there is rupture,
a local haematoma,
blood/contrast in adjacent tissues and peri-aortic fat strending are observed (Fig. 18,
Fig. 19,
Fig. 20 and Fig. 21).
Signs of impending rupture or contained leakage include: bulky aneurysms; thrombus cracking; focal discontinuity of intima calcifications (Fig. 22); acute haematoma in the mural thrombus that insinuates into the wall [high-attenuating crescent sign - (Fig. 23)]; and when the posterior wall of the AA molds itself to the anterior surface of the vertebrae (draped aorta sign).
Aortitis
Aortitis is a term that includes a broad category of infectious and noninfectious pathologies that cause inflammation of the aortic wall.
Aorta vasculitis manifests with fever,
pain and,
when chronic,
weight loss.
The treatment is medical,
with corticosteroids or immunosuppressive drugs.
At CT,
a low density enhancement of the peri-aortic soft tissues is observed (Fig. 24 and Fig. 25).
The affected segments of the aorta may be either dilated or narrowed.
Complications
Besides the clinical presentation and some of the pathophysiology,
the AAS also share some complications among themselves.
Besides that,
AAS can evolve/transform into each other.
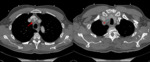
These possible complications are ischemia of the various territories irrigated by the aorta or its branches (Fig. 26 and Fig. 27),
cardiac tamponade,
fistulization to adjacent structures (Fig. 28,
Fig. 29,
Fig. 30 and Fig. 31),
formation of aneurysms and pseudoaneurysms (Fig. 32 and Fig. 33) and rupture.