ANATOMY
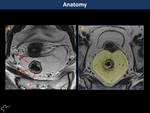
The rectum is approximately 15cm in length and layers can be well depicted by MR (Fig. 1):
-
Inner hyperintense layer,
mucosa and submucosa (differentiation may be difficult)
-
Intermediate hypointense layer,
muscularis propria
-
Outer hyperintense layer,
perirectal fat tissue.
Mesorectum (Fig. 1) contains lymph nodes,
vessels,
and several fibrous septa,
and is surrounded by the mesorectal fascia,
that represents the circumferential resection margin (CRM) when total mesorectal excision is used as the surgical approach.
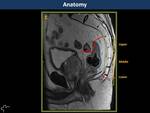
The rectum is divided into thirds since outcomes and surgical management are affected by the location of pathology(1) (Fig. 2):
-
Upper: the lowest edge of pathology is more than 10cm from the anal verge.
The anterior wall is covered by the peritoneal reflection.
The point of peritoneal reflection attachment occurs at a variable height,
particularly in women.
Careful assessment of the peritoneal reflection in upper rectal tumors.
-
Lower: the lowest edge of pathology is less than 5cm from the anal verge.
At this level,
the mesorectum tapers sharply; anteriorly the mesorectal fascia fuses with the remnant of the urogenital septum (rectoprostatic fascia in the male; rectovaginal septum in the female).
The anorectal junction is held forward by the puborectal sling.
Perirectal space is the pelvic continuation of the retroperitoneum.
It is bordered superiorly by the peritoneal reflection and laterally by the endopelvic fascia and lateral ligaments of the rectum.
It is subdivided in the mesorectal and retrorectal spaces by the mesorectal fascia.
Presacral and rectosacral fascias separate retrorectal space from the presacral space (Fig. 2).
RECTAL
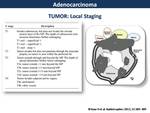
Rectal tumours can be classified accordingly to the wall layer where they arise or their most frequent form of presentation (Table 1, Table 2).
1.
Mucosa
Adenocarcinoma
Adenocarcinoma is the most common type of rectal cancer (98%),
presented in patients older than 50 years-old.
They often present as an irregular thickening of the rectal wall with ulcerating mucosa.
Polypoid pattern is also possible,
especially in low grade adenocarcinomas.
They show intermediate signal intensity on T2WI and restriction on diffusion.
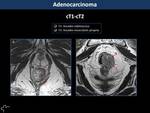
MR has lower accuracy than eco-endoscopy in the staging of tumours limited to the mucosa (T1) or to the muscularis propria (T2).
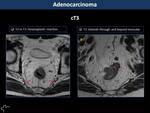
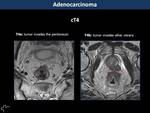
Infiltration of mesorectal fat and draining veins (T3) and peritoneal reflection and adjacent organs (T4) are well depicted by MR (Fig. 3,
Fig. 4,
Fig. 5,
Fig. 6)
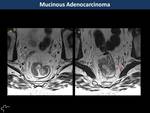
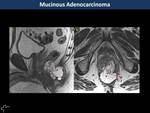
Mucinous is a less frequent (10-20%) subtype of rectal adenocarcinoma,
with poorer prognosis.
They present large mucin pools hyperintense on T2WI that represent >50% of the tumour (Fig. 7, Fig. 8).
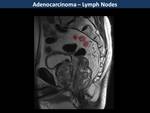
Lymph nodes with spiculated or indistinct margins or heterogeneous signal intensity are considered pathologic (Fig. 9).
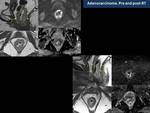
Patients with locally advanced rectal cancer received presurgical chemo-radiotherapy (2).
Secondary changes to chemo-radiotherapy before total mesorectal excision such as proctitis and ulceration,
fibrosis and desmoplastic reaction and changes in mesorectal lymph nodes (Fig. 10).
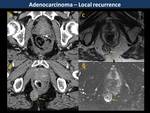
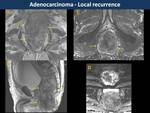
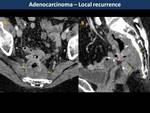
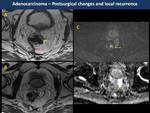
Most local recurrences occur within the first 3 years after surgery and they are usually associated with distant metastases.
CT has a great sensitivity to detect local recurrence but lacks specificity(2) in differentiating recurrence from postsurgical changes.
MR offers better specificity,
although it also has its challenges.
Low signal intensity on T2WI may be secondary to fibrosis,
but it cannot rule out cluster of tumor cells within it.
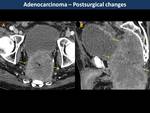
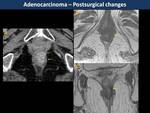
Persistence of soft-tissue signal intensity on treated lesions and restriction on DWI suggests residual tumor (local recurrences in Fig. 11, Fig. 12, Fig. 13 and postsurgical changes in Fig. 14, Fig. 15, Fig. 16).
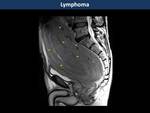
Lymphoma (Fig. 17)
Colorectal location only represents 6-20% of gastrointestinal lymphomas.
They usually occur in male older than 60 years-old with widespread large B-cell lymphoma,
although they may also present in uncontrolled longstanding celiac patients as a primary rectal lymphoma (MALT).
They more often present as polypoid masses than concentric wall thickening with ulcerating mucosa,
with heterogeneous hyperintensity on T2WI,
marked restriction on diffusion and mild-to-moderate contrast-enhancement.
Important clues in the differential diagnosis with adenocarcinomas are the lack of obstruction with aneurysmal dilatation of the rectum and preservation of the mesorectal fat (1).
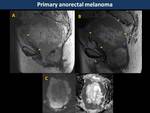
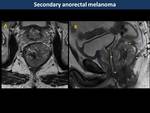
Anorectal melanoma (Fig. 18, Fig. 19)
It accounts for <2% of all melanoma cases and it presents as a part of metastatic melanoma rather than primary rectal melanoma.
It appears as an intraluminal polypoid mass without colonic obstruction,
showing hyperintensity on T1WI because melanin content,
although 10-29% of them are amelanotic.
They also show high signal intensity on T2WI,
avid contrast-enhancement but minimal perirectal infiltration or lymphadenopathies (3)
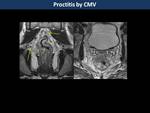
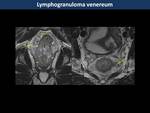
Proctitis (Fig. 20, Fig. 21, Fig. 22) (4)
Inflammation of the rectum and anus may be related to different etiologies that share similar clinical and imaging characteristics: moreover,
these features could also mimic malignancy.
On CT,
they appear as concentric wall thickening,
mucosal hyperemia and mural stratification,
associated with mesorectal fat stranding and lymphadenopathy.
On MR,
active inflammation is depicted by high signal on T2WI and restriction on diffusion,
whereas chronic processes could present with submucosal fat deposition or low signal on T2WI with delayed contrast-enhancement due to fibrosis.
Proliferation of mesorectal fat and ingurgitation of vasa recta may also be present.
Proctitis in ulcerative colitis usually extends proximally to the sigma,
whereas skip lesions are often en in Crohn's colitis.
This last one often presents more severe wall thickening and it is often associated with fistula-in-ano,
perirectal abscesses.
Infectious proctitis may be secondary to sexually transmitted infections such as citomegalovirus,
N.
gonorrhoeae or C.
trachomatis.
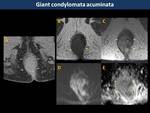
Common anal lesions found in syphilis include ulcers,
fissures and condylomata lata.
Other etiologic types of proctitis include stercoral in elderly or neurologically impaired patients with impacted fecal material; radiation in over 50% of patients with prior radiotherapy; and ischemic,
very rare due to a rich collateral vascular supply.
2.
Submucosal
Metastases
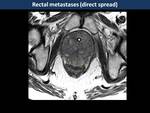
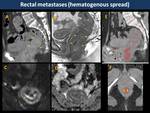
Secondary neoplastic involvement of rectum is uncommon and it is usually by direct invasion rather hematogenous spread (1)
Direct spread appears as a target sign on CT and concentric ring pattern on T2WI due to exaggeration of normal zonal anatomy by infiltrative tumour in the submucosa and around the muscle layer (Fig. 23).
Metastases from hematogenous spread appear as a submucosal mass or as linitis plastica characterized by an atypical wall thickening with mucosal preservation and non-distensible rectum that may be overlooked by endoscopy (Fig. 24).
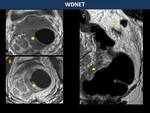
Well-differentiated neuroendocrine tumour(WDNET) (Fig. 25)
They arise in the deep portions of the submucosal glands of rectal wall.
They are found incidentally in middle-aged patients,
since carcinoid syndrome is rare unless liver metastases are present.
They present as solitary submucosal nodules above the dentate line,
hyperintense on T2WI with avid contrast-enhancement. Areas of cystic and necrotic degeneration may be present in the large ones.
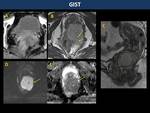
Gastrointestinal stromal tumour (GIST) (Fig. 26)
Rectum represents the third most common location of GIST (<5%).
On MR,
they frequently present as large,
eccentric and well-circumscribed masses with solid component with intermediate-to-high signal on T2WI,
heterogeneous contrast-enhancement and necrotic degeneration.
Unlike adenocarcinomas,
GISTs don't usually present perirectal or lymph node invasion neither bowel obstruction.
Leiomioma and leiomiosarcoma
Extremely rare mesenchymal tumours frequently presented in female with prior radiotherapy.
They present as polypoid masses isointense-to-mildly hyperintense on T2WI,
with variable contrast-enhancement.
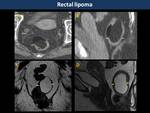
Lipoma and liposarcoma (Fig. 27)
They are broad-based intraluminal tumours,
sometimes pedunculated,
that follow the signal intensity of fat in all sequences.
Contrast-enhancement is variable according to the aggressiveness of the tumour.
Hemangioma
Benign vascular tumours presented in young men.
On MR,
there is a diffuse hyperintensity of the thickened wall on T2WI with serpentine flow voids.
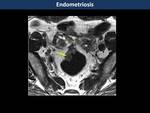
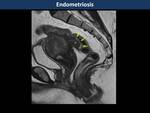
Endometriosis (Fig. 28, Fig. 29)
Deep infiltrating endometriosis affects women presented with abdominal pain during the menstrual period,
infertility and intestinal obstruction.
When endometriomas or deep infiltrating endometriosis signs are detected during pelvic MR,
a careful look of the rectosigmoid should be made.
Most cases are limited to the serosa and less frequent infiltrating deeply into the intestinal wall.
MR signs included:
-
Nodular or masslike bowel wall thickening in favour of hypertrophy of muscular propria.
-
“Mushroom cap” sign: T2WI show affection of anterior rectosigmoid wall with low signal of a thickened muscular propria layer and high signal of mucosa and submucosal layers (displaced into the bowel lumen)
-
Internal cystic areas or hemorrhagic components may be present.
Colitis cystica profunda
Benign tumour-like condition incidentally found in middle-aged patients associated with solitary ulcer and rectal prolapse.
They present as mucin-filled cysts without contrast-enhancement.
PERIRECTAL
To simplify the classification of pathology,
perirectal masses can be initially categorized into two groups according to presence of intralesional fat (Table 2)(5)
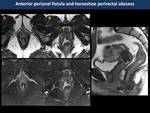
Anorectal abscess and fistulae (Fig. 30)
They most commonly arise from obstruction and infection of anal glands (90%),
especially in patients with Crohn's disease and immunosuppression.
Patients usually present with pain,
fever and a fluctuated collection.
Fistulae most commonly arise from an abscess,
but also from inflammatory bowel disease or malignancy.
On CT,
they present as well-circumscribed collections with thickened and contrast-enhanced walls and with debris and air bubbles inside (50%).
On MR,
T2WI with fat-suppression and STIR sequences have greater accuracy than CT for depicting the extent of the abscess and fistulous tracts and their relationship with the anal sphincter complex and elevator plate muscles.
Rectovaginal and rectovesical fistulas may also be depicted.
Fluid,
pus and granulation tissue of the fistulous tracts appear hyperintense on T2WI with avid contrast-enhancement and restriction on diffusion.
Longstanding fistulas with fibrosis appear hypointense with delayed contrast-enhancement.
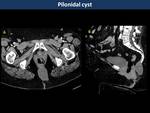
Several conditions can mimic perirectal and perianal abscess,
such as necrotic or mucinous anorectal tumours,
thrombosed hemorrhoids,
ruptured Bartholin gland cysts,
furuncles and pilonidal cysts.
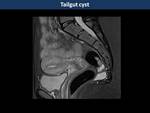
Developmental cysts (Fig. 31, Fig. 32) (5)
They are the most common retrorectal cystic tumours (60%).
They are found incidentally in middle-aged women.
-
Epidermoid and dermoid cysts are lined with stratified squamous epithelium.
On MR,
they are well-circumscribed thin-walled cysts,
hyperintense on T2WI,
without contrast-enhancement.
Restriction on diffusion is characteristic of epidermoid cysts,
whereas dermoid cysts may present fat nodules (skin appendages).
-
Enteric and neurenteric cysts are lined either with intestinal mucosa or mature mucosa of endodermal origin with lamina propria,
respectively.
Tailgut cysts are enteric cysts with multilocular cysts with variable signal intensity depending on mucin content.
Rectal duplication cysts are unilocular cysts with low signal bands representing the two layers of smooth muscle.
If they become infected,
they present thickened and contrast-enhanced walls,
pericystic fat stranding and heterogeneous signal may be present.
Malignant degeneration may be present,
especially in duplication cysts more than tailgut cysts (20 vs.
13%) They present contrast-enhanced nodules,
internal septa and irregular wall thickening.
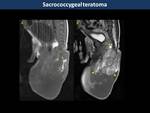
Sacrococcygeal teratoma (Fig. 33)
Most common congenital tumour in fetus and neonates with female predilection (4:1)
They often developed outside the pelvis (47%) but they can also arise in the presacral space.
They are solid-cystic lesions with avid contrast-enhancement.
Fat nodules,
fat-fluid levels (80%) and calcifications (50%) may be present (6).
Neurogenic tumours
Schwannomas and neurofibromas are well-circumscribed tumours with indistinguishably radiological features,
but schwannomas mostly show heterogeneous signal intensity and contrast-enhancement,
whereas neurofibromas show a target-like pattern with peripheral myxoid stroma hyperintense and central collagenous tissue hypointense on T2WI.
They are frequently associated with thickened nerve roots and widened neural foramina.
Malignant degeneration should be suspected in large and irregular tumours with peripheral contrast-enhancement,
perilesional edema and intratumoral lobulations.
Osseous tumours
Giant cell tumour is benign and appears as eccentric and lytic tumour with a thin sclerotic rim,
associated with a soft-tissue mass. On MR,
they demonstrate moderate hyperintensity both on T1 and T2WI.
Aneurysmal bone cyst may be present.
Chordoma is malignant and appears in older man at midline. Unlike GCT,
amorphous and peripheral calcifications are very common (90%) and it presents avid contrast-enhancement (5,6)
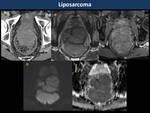
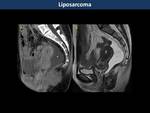
Lipoma and liposarcoma (Fig. 34)
Unlike in the rectal wall,
retrorectal liposarcomas are more frequent than lipomas and they are solid tumours with heterogeneous hyperintensity on T2WI with contrast-enhancement and scattered fat foci.
Benign cyst mesothelioma (Fig. 35)
They are intermediate-grade tumour of the mesothelial cells of the peritoneum presented in childbearing age women,
especially with prior surgery or pelvic inflammatory disease.
They present as thin-walled multicystic mass without lymphadenopathies.
They rarely extend beyond pelvis,
but in this cases scalloping of the liver and spleen may be present without peritoneal implants,
unlike peritoneal carcinomatosis (6)
Other perirectal conditions
- Mielolipoma, well-circumscribed and lobulated solid lesion.
- Myxoma, cyst-like lesion with leak of myxomatous tissue.
- Extramedullary hematopoiesis, well-circumscribed fat-containing mass adhered to a thickened presacral fascia.
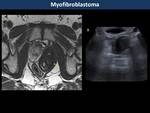
- Myofibroblastoma, solid mass heterogeneously hypointense on T2WI (Fig. 36)