I- Development of HCC
Hepatocellular carcinoma (HCC) is the fifth most common cancer in the world with increasing incidence,
especially in Western countries.
The most important clinical risk factor for HCC is cirrhosis,
with about 80% of HCC develop in patients with a cirrhotic liver.
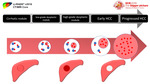
Hepatocarcinogenesis is defined as the gradual transformation of nonmalignant liver cells into HCC.
It is a complex,
multistep process which occurs at two levels;
a) molecular and cellular level,
progressive accumulation of epigenetic and genetic alterations is thought to be triggered by chronic liver inflammation with repeated cycles of cell injury,
death and regeneration.
b) histologic level,
progression of successively more advanced precancerous,
early cancerous,
and overtly malignantly lesions occur in a multi-step process which represents a biologic continuum.
[1] (Figures 2)
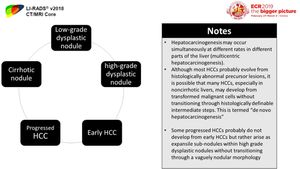
Fig. 2: Different pathological stages of hepatocarcinogenesis
II- Pathologic alterations of HCC and their imaging appearance
Angiogenesis
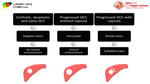
Angiogenesis refers to the progressive increase of the unpaired (nontriadal) arteries while the portal tracts (which contain portal veins and nontumoral hepatic arteries) progressively diminish.
Cirrhotic nodules and low-grade dysplastic nodules usually have similar arterial and portal venous flow.
High-grade dysplastic nodules and early HCCs usually have diminished arterial and portal venous flow.
Moderately differentiated,
progressed HCCs usually have elevated arterial flow with reduced or absent portal venous flow.
This leads to the progression in appearance on the vascular imaging phases from isoenhancement relative to background liver,
to hypoenhancement relative to background liver in the arterial and portal venous phases and finally hyperenhancement in the arterial phase and washout appearance in portal venous and delayed phases.
[1] (Fig.3)
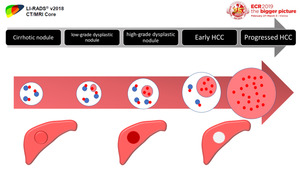
Fig. 3: Diagram shows the progressive increase of unpaired (nontriadal) arteries and decrease of the portal flow with the development of arterial phase hyperenhancement.
Venous drainage
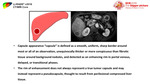
Cirrhotic nodules,
dysplastic nodules,
and early HCCs drain into hepatic veins,
while progressed HCCs without fibrous capsules drain to sinusoids.
Progressed HCCs with fibrous capsules drain into portal veins.
This may explain the phenomenon of corona enhancement which is considered an imaging feature of hypervascular,
progressed HCC.
[1] (Figure 4)
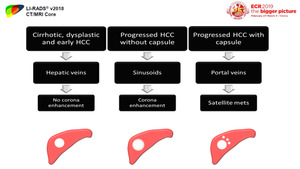
Fig. 4: Diagram shows the difference in venous drainage between cirrhotic (nodules, dysplastic nodules, and early HCCs), progressed HCCs without fibrous capsules and progressed HCCs with fibrous capsules.
Corona enhancement is not specific for HCC and can occur with any hypervascular neoplasm with peritumoral neovascularization.
It is best detected at the late arterial or portal-venous phase.
[2] (Figure 5)
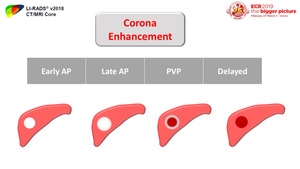
Fig. 5: "Corona enhancement" is the peritumoral parenchymal enhancement that begins a few seconds after enhancement of the tumor itself. It is attributed to passage of contrast material from the tumor through the draining sinusoids and portal venules into the surrounding parenchymal sinusoids, with which the drainage vessels communicate. Corona enhancement is evaluated on late arterial and PV phases, and it should lag behind the tumor enhancement
Tumor capsule and fibrous septa
Capsule formation is a characteristic histopathologic feature of progressed HCCs with expansile growth.
On the other hand,
capsule formation is rarely observed in early,
very well-differentiated HCCs and in infiltrative,
poorly differentiated HCCs,
and it does not occur with intrahepatic cholangiocarcinoma (ICC). [3] (Figure 6)
Fat content
Hepatocytes may accumulate fat early during hepatocarcinogenesis,
thus low-grade dysplastic nodules,
high-grade dysplastic nodules,
and early HCCs may become more steatotic either focally within clonal-like subnodules or diffusely than background liver.
[ 2,4] (Figures 7 & 8)
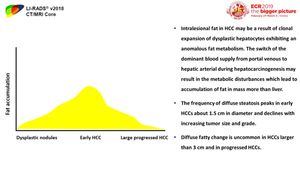
Fig. 7: The frequency of diffuse intranodular steatosis increases from low-grade dysplastic nodule to high-grade dysplastic nodule and then to early HCC where it peaks in early HCCs about 1.5 cm in diameter and declines with increasing tumor size and grade. Thus, diffuse fatty change is uncommon in HCCs larger than 3 cm and in progressed HCCs and usually is not observed in poorly differentiated HCC

Fig. 8: Difference of fat deposition in a mass more than the surrounding liver and fat sparing in a mass in a diffusely fatty liver
References: The schematic drawings are reproduced from LI-RADS v2018 with permission form the American college of radiology
Iron content
In cirrhotic livers without diffuse iron deposition,
iron may accumulate in low-grade dysplastic nodules and some high-grade dysplastic nodules.
These iron-rich nodules commonly are described as “siderotic nodules”.
With further dedifferentiation,
hepatocytes become “resistant” to iron accumulation,
so most high-grade dysplastic nodules,
early HCCs,
and progressed HCCs are iron free. [1,2] (Figure 9)
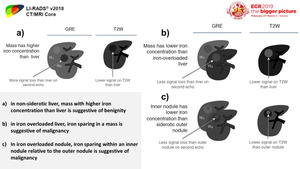
Fig. 9: Iron resistance is observed also in dysplastic foci, dysplastic nodules, and HCCs in livers with diffuse hepatic iron overload due to any cause. So, in diffusely iron-overloaded livers, a solid nodule free of iron is likely to be dysplastic or malignant.
References: The schematic drawings are reproduced from LI-RADS v2018 with permission form the American college of radiology
Reduction in organic anionic transporting polypeptides
(OATP) transporters
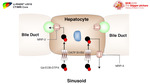
OATP transporters are a family of proteins expressed in hepatocytes along the sinusoidal membrane and involved in transport of bile salts.
[4] (Figure 10)
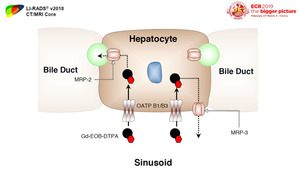
Fig. 10: OATP1B1/B3 is one of these transporters that is suggested to be responsible for uptake of gadoxetate disodium and gadobenate dimeglumine by hepatocytes
References: Reproduced from LI-RADS v2018 manual with permission from the American College of Radiology
The expression of OATP transporters is suggested to diminishes during hepatocarcinogenesis,
showing higher levels in cirrhotic nodules and low-grade dysplastic nodules,
with lower levels in many high-grade dysplastic nodules,
early HCCs,
and progressed HCCs.
[1,
2] (Figure 11 & 12)
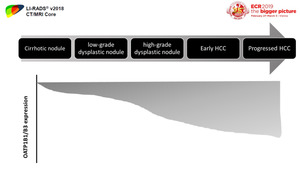
Fig. 11: Diagram shows reduction in OATP expression during hepatocarcinogenesis
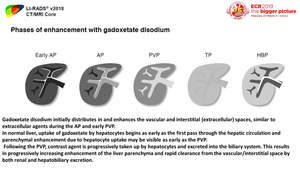
Fig. 12: The degree of gadoxetate uptake by a lesion depends on the expression and activity of OATPB1/B3, which in turn is determined by the underlying cytogenetic profile.
References: The schematic drawings are reproduced from LI-RADS v2018 with permission form the American college of radiology
III- Imaging of HCC
The prognosis of HCC depends on the stage at which it is detected,
thus,
early detection of HCC is critical to improve patients’ survival.
(HCC) is unique among malignancies in having tumor characteristics on cross-sectional multiphasic contrast computed tomography (CT) or magnetic resonance imaging (MRI) that allow for a highly accurate diagnosis of HCC without an invasive biopsy.
[5]
Clinical management guidelines for surveillance of patients at risk of HCC have been founded by many scientific societies.
Despite differences in surveillance strategies,
all current guidelines recommend ultrasonography (US) as the primary imaging test for surveillance,
while computed tomography (CT) and magnetic resonance (MR) imaging are not advocated for surveillance.
When a surveillance test is positive,
a more definitive imaging examination is required.
Currently,
all guidelines endorse multiphasic CT and MR imaging with extracellular agents as first-line modalities for noninvasive diagnosis and staging of HCC.
[1]