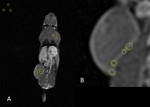
Nanoparticle synthesis
SN132D nanoparticles contains a core prepared from monomer precursor (Fig. 1A – compound 1) which is coated with a dipodal coating precursor (Fig. 1A – compound 2).
Mn ions,
in the form of MnCl2,
was loaded into the coated nanoparticles in a phosphorus to Mn ratio of 7.
The nanoparticles between 70 and 10 kDa were collected by tangential flow filtration.
The diafiltered material was then concentrated using a 10 kDa filter and further formulated with CaCl2 and MnCl2 to be in electrolyte balance with human plasma.
The pH was adjusted to physiological pH with NaOH. SN160 nanoparticles,
were produced as SN132D,
and thereafter depleted for Mn.
Thus,
enabling studies of the particle by itself.
In-vitro studies
In vitro Mn cytotoxicity of SN132D was evaluated in terms of growth,
morphological changes,
viability and apoptosis,
on cultivated cell lines of murine (macrophage Raw264.7 and fibroblast L929) and human (monocytic THP1,
fibroblast HFL-1 and liver HepG2) origin.
SN132D was compared to MnCl2 at equal Mn concentrations and against SN160 at equal silicon concentrations.
For cell growth determinations the CellTiter-Blue® Cell Viability Assay (Promega) was used.
Ames test was performed for SN132D at Charles River Laboratories (CRL) Den Bosch BV,
the Netherlands.
Animal studies
MRI and efficacy studies were performed at Redoxis AB (Sweden),
using Balb/cJRj mice (Janvier Labs,
France) subcutaneously inoculated with the syngenic 4T1-mammary cancer cell line (ATCC® CRL-2539TM).
Efficacy studies were also performed at Adlego AB (Sweden) in Balb/cJBomTac mice (Taconic,
Denmark) and in F344 rats (CRL,
Germany).
Mass balance and bile duct catherization experiments with SN132D,
and an extended single dose toxicity experiments with SN160,
were performed in Wistar Han rats (CRL,
Germany) at Adlego AB,
Sweden.
All animal experiments were approved by the Regional Ethical Committees for Animal Research in Stockholm or Lund,
Sweden.
Animal studies regarding toxicity,
safety pharmacology,
pharmacokinetic and excretion (only minipig) were performed at CRL in Lyon.
The investigated species were Wistar Han IGS rat (CRL,
France) and Göttingen minipig (Ellegaard,
Denmark).
All studies were reviewed and approved by the ethical committee of the test facility.
Elemental analysis (ICP-OES)
Elemental analysis of plasma samples (rat and minipig) and urine (minipig) were analysed for Mn and Si using a Perkin Elmer Optima ICP-OES.
The methods were validated at CRL.
Determination of Mn and Si in urine and bile (rat) and digested samples from tumors and muscles (mouse and rat) were performed with an Agilent ICP-OES 710.
MRI
MRI experiments were performed using a 1.5T MAGNETOM Avantofit MR scanner (Siemens Healthineers,
Erlangen,
Germany).
A 4-channel small flex coil was used for imaging signal collection.
Anesthetized animals were intravenously injected with a SN132D dose corresponding to 106 µmol Mn/kg.
The mice were placed in prone position inside the coil,
with the body at the isocenter of the magnet.
Coronal images through the tumor were acquired using T1-weighted,
fat saturated turbo spin echo (TSE).
Parameters were: echo time = 8.0 ms,
repetition time = 320 ms,
scan time 2 min.
Two-mm slices were imaged with 6 averages and 0.2 mm spacing.
Image analysis
The unaltered raw TSE MR images were analyzed with ImageJ.
The standard deviation,
σ,
of the background signal and the mean signal intensity of the muscle and tumor ROIs (Fig. 2) were determined by the software.
The signal-to-noise ratio (SNR) was calculated by dividing the mean signal intensity by σ,
and the tumor versus muscle contrast-to-noise ratio (CNR) was calculated by subtracting SNRmuscle from SNRtumor.

![Fig. 1: A) Core monomer precursor 1 and coating monomer precursor 2. B) Conceptual 3D-model of SN132D. Core atoms in orange and PEG coating in blue [1].](https://epos.myesr.org/posterimage/esr/ecr2019/146808/media/802990?maxheight=150&maxwidth=150)