SN132D is a Mn-based contrast agent composed of nanoparticles with high relaxivity
SN132D is a Mn-containing nanoparticle contrast agent for tumor selective MRI.
SN132D contains a polymeric core with Mn(II)ions chelated to phosphonate groups.
A coating layer of covalently attached-PEG-polymers covers the core (Fig.
1B).
The coating layer prevents aggregation,
reduce binding to plasma proteins and makes the nanoparticle immunogenically inert [1].
The phosphonates are efficient chelators with an average of 14 Mn(II)ions bound to each particle,
and a chelating strength of the same order of magnitude as EDTA,
resulting in a free concentration of Mn in the low nanomolar range [1].
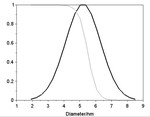
The average hydrodynamic diameter of SN132D is 5.6 nm,
thus promoting renal excretion and tumor contrast,
which enables macromolecules to selectively accumulate in tumors based on the Enhanced Permeability and Retention (EPR) effect (Fig. 3).
The SN132D particle contains magnetically active Mn(II)ions at a concentration of 65.1 ± 0.8 mM,
as determined by ICP-OES,
with a per Mn longitudinal relaxivity (r1) of 24.8 ± 0.24 mM-1 s-1 at 1.5 T and 37 °C in water,
corresponding to a relaxivity for each nanoparticle to 347 ± 3 mM-1 s-1.
The relaxivity in serum is significantly higher than in water,
due to an increase in medium viscosity and chelation of 19 ± 2% of Mn(II) ions to human serum components,
most likely human serum albumin [1].
The per Mn relaxivity is among the highest reported for Mn-containing systems and is considerably higher than the contrast agents currently on the market [4].
Contrast anhancement by MRI up to 4.4 h after administration
MRI scans of 4T1 tumors were performed predose,
and up to 4.4 h after administration of 106 µmol Mn/kg of SN132D (Fig. 4).
In the image taken prior to injection,
the tumor is visible because of its bulkiness,
but no contrast is seen between tumor and nearby tissue.
At 10 minutes post-injection no contrast enhancement is seen.
At 1 h post-injection,
SN132D accumulates in the tumor rim,
which becomes noticeably whiter,
giving a clear border between the tumor and the surrounding tissue.
The contrast between tumor and surrounding tissue is present up to 4.4 h and noticeably improved at 4.4 h compared to 1 h.
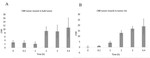
The CNR between tumor and muscle was calculated (Fig. 5).
In the bulk tumor,
a significant increase in CNR (3.1 times) was reached at 2 h and maintained at a constant level until 4.4 h.
At the tumor rim,
a gradual build-up in CNR was observed at 2,
3 and 4.4 h,
with an increase compared to pre-injection CNR of 48.5,
62.6 and 70.5 times,
respectively.
Increase of Mn in tumor vs muscle after 2 h
The accumulation of SN132D in tumor versus non-tumor tissue in different subcutaneous tumor models was assessed (Table 1).
Relative accumulation of SN132D in tumors was 7-51 times higher than in muscle 2 h after administration,
showing a high and selective accumulation of SN132D in tumors,
presumably due to the EPR effect.
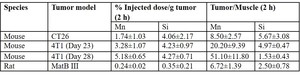
Table 1: Summary of studies assessing tumor accumulation of SN132D (2 h). 4T1 and MatB III are syngeneic tumor models of breast cancer in mouse and rat, respectively. CT26 is a syngeneic tumor model of colon cancer in mouse.
SN132D nanoparticle biologically inert
Mn(II) ions are tightly chelated in the particle formulation,
but will in biofluids distribute fairly quickly.
Human serum albumin is the major contributor to competitive Mn-binding,
and in a static situation in-vitro,
relaxivity experiments have shown that around 20% of the Mn of SN132D is chelated to serum components [1].
The SN132D particle itself does not bind irreversibly to plasma proteins,
and does not induce complement activation in vitro in human plasma or serum,
or cause hemolysis of whole blood,
suggesting that the particle is relatively inert,
biologically.
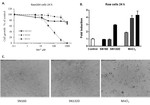
Proliferation experiments showed that SN132D induced less growth inhibition in cell culture compared to freely administrated MnCl2 at the same Mn concentration.
For all cell lines tested and exemplified by Raw264.7 cells in Fig. 6A,
there was a clear difference between cell cultures treated with SN160 and SN132D,
when compared at equal silicon concentrations.
SN160 did not significantly inhibit cell growth in vitro,
and the minor effects seen on growth by SN160 were not reflected by changes in apoptosis and cell morphology at any dose compared to unexposed control cells.
Monocytic Raw264.7 cells were most sensitive to Mn exposure whereas mouse fibroblast L929 cells were most affected by SN132D.
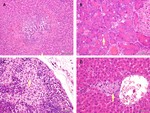
Histologically,
there was a clear difference after 24 h between cell cultures treated with SN132D and MnCl2,
and similarly MnCl2 induced apoptosis and cell death in Raw264.7 cells at 100 µM or above (Fig. 6B and C).
Thus,
the cytotoxicity in all cell lines investigated,
were due to Mn-induced apoptosis and did not originate from the nanoparticles.
The genetic toxicity of SN132D has been investigated in an Ames test and was not found to be mutagenic,
neither in the Salmonella typhimurium reverse mutation assay nor in the Escherichia coli reverse mutation assay.
Mn is the cause of observed toxicity at high doses in-vivo
The toxicity of SN132D has been evaluated in vivo in single dose toxicity studies of up to 2 weeks’ duration in rat and minipig.
Findings are described in Table 2 and in Fig. 7A-D.
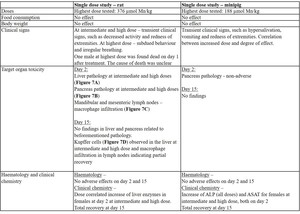
Table 2: Findings in toxicity studies in rat and minipig administered with SN132D. All studies were performed as extended dose toxicity studies. Maingroups were euthanized at day 2 and subgroups after a recovery period of 14 days.
An extended single dose study was performed to assess the toxicity of SN160 in female rats equal to a dose where SN132D gave findings.
Administration of SN160 caused minimal accumulation of foamy macrophages in the mesenteric and mandibular lymph nodes on day 2 and after 14 days recovery,
presumably indicating a clearance mechanism involving phagocytosis of the nanomaterial by phagocytic cells.
The absence of toxicity in this study strongly suggests that the toxicity caused by SN132D relates to the Mn component of the substance rather than the nanoparticle matrix component.
Safety pharmacology studies were performed to assess the pharmacological profile of SN132D on the central nervous system (CNS),
cardiovascular system and respiratory system.
Overall,
no effects were observed at doses in rat and minipig,
well above the anticipated pharmacologically active dose at 10 µmol Mn/kg to be tested in FIH trials.
Two-way excretion
Mn from SN132D will distribute fairly quickly to plasma proteins.
One fraction of nanoparticles will keep Mn tightly chelated and remain in the circulation for a longer time period.
Mn chelated to plasma proteins is actively cleared from the blood in order to maintain a strictly controlled Mn homeostasis,
and will be excreted in faeces via the hepato-biliary excretion pathway [5].
The fraction of tightly chelated Mn to SN132D nanoparticles will be excreted in urine (shown as Mn excreted via urine).
The size distribution profile of the nanoparticles will lead to renal clearance for the particles with sizes below the renal filtration cut-off (shown as Si excreted via urine) (Table 3).
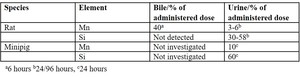
Table 3: Excretion of SN132 measured by Mn and Si in rat and minipig
Pharmacokinetic modelling and proposed clinical dose
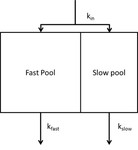
Two pharmacokinetic (PK) models,
a two-pool model for Mn (Fig. 8) and a kidney-clearance model for Si (Fig. 9) were developed.
The main reason for creating these models was to obtain a better predictive power for pharmacokinetics of the nanoparticles and the chelated Mn ions in humans.
The models fit the data well with a small number of adjustable parameters and with good predictive value for allometric scaling to humans.
Mn has been established as the source of infusion-related clinical signs and target organ toxicity in nonclinical studies,
and allometric scaling is therefore based on Mn,
and not on Si.
A correlation between toxicity at specific dose levels and peak plasma concentrations of Mn in investigated animal species has been observed.
No obvious correlation was observed between AUC(Mn) and dose levels.
Exposure for allometric scaling is based on peak plasma concentration of Mn calculated from the two-pool model.
A clinical starting dose of 10 µmol Mn/kg has been proposed for the first-in-human clinical trial,
providing a safety margin of 27x to maximal dose as based on NOAEL obtained in rats and minipigs.

![Fig. 1: A) Core monomer precursor 1 and coating monomer precursor 2. B) Conceptual 3D-model of SN132D. Core atoms in orange and PEG coating in blue [1].](https://epos.myesr.org/posterimage/esr/ecr2019/146808/media/802990?maxheight=150&maxwidth=150)
![Fig. 3: Illustration of Enhanced Permeability and Retention (EPR) effect. Nanoparticles enters the tumor due to rigid and hyper-permeable (“leaky”) blood vessels (arrow), and the nanoparticles remains in tumor tissue due to impaired lymphatic drainage. Immunohistochemistry image of tumor tissue from murine mammary tumor virus-polyoma middle T (MMTVPyMT) mice stained with anti-PEG (brown) and hematoxylin counterstaining (blue) 30 min post i.v. injection [2]. References: Eriksson et al. (2014) Novel Nano-Sized MR Contrast Agent Mediates Strong Tumor Contrast Enhancement in an Oncogene-Driven Breast Cancer Model. PLoS ONE 9(10): e107762. https://doi.org/10.1371/journal.pone.0107762](https://epos.myesr.org/posterimage/esr/ecr2019/146808/media/810244?maxheight=150&maxwidth=150)
![Fig. 4: Representative T1-weighted full-body MR spin echo images selected to show 4T1 mammary tumors (s.c.) [3] pre-injection and at different times post injection of SN132D (10 min, 1 h, 2 h, 3 h, 4.4 h). Arrows point to tumor.](https://epos.myesr.org/posterimage/esr/ecr2019/146808/media/803392?maxheight=150&maxwidth=150)