Aims and objectives
The purpose of this study was to measure susceptibility artifacts in pediatric stainless steel crowns according to standardized test methods of the American Society for Testing and Materials (ASTM) at 1.5 T and 3 T MRI to clarify if the crown needs to be removed for the examination.
Methods and materials
For this study different pre-fabricated pediatric stainless steel crowns (type 304,
316,
316L,
see Fig. 1) for the maxillary left first and second deciduous molars from three manufacturers (3M Espe,
Acero XT,
Denovo Dental) were compared on a 1.5 T and 3 T MRI (Magnetom Avanto and Prisma,
Siemens,
Erlangen,
Germany).
The signal loss was measured according to ASTM F2119-07 [1] for a TSE (TR 500 ms,
TE 20 ms) and GRE (TR 100 ms,
TE 15 ms) sequence.
The evaluation was performed with the...
Results
There are significant differences in susceptibility artifacts dependent on material composition of the crowns (see Fig. 3).
The total signal loss was 27-to-35 ± 4 mm for TSE sequences and 35-to-46 ± 5 mm for GRE sequences.
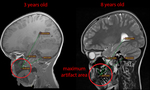
The distances from the maxillary left second deciduous molar to anatomic structures (see Fig. 4) were 36-to-44 ± 5 mm (orbital rim),
39-to-53 ± 6 mm (gyrus temporalis inferior),
70-to-81 ± 5 mm (lower margin of cerebellum) and 109-to-126 ± 7 mm (lateral ventricle).
Conclusion
In general,
pediatric stainless steel crowns do not have to be removed before a MRI examination.
However,
materials with lower artifact size should be preferred.
Mechanical modifications of the crown margins do not result in significantly larger artifacts and can as a rule thus be ignored.
Personal information
Andreas Heinrich,
medical physicists
Department of Radiology,
University Hospital Jena,
Friedrich Schiller University,
Germany
Am Klinikum 1
07747 Jena,
Germany
Fon: +49 3641 9 - 324 898
Fax: +49 3641 9 - 324 832
e-mail:
[email protected]
References
ASTM F2119-07,
ASTM F2119-07,
Standard Test Method for Evaluation of MR Image Artifacts from Passive Implants.
2013,
ASTM International: West Conshohocken,
PA.
Heinrich,
A.,
U.K.
Teichgräber,
and F.V.
Güttler,
Measurement of susceptibility artifacts with histogram-based reference value on magnetic resonance images according to standard ASTM F2119. Biomedical Engineering/Biomedizinische Technik,
2015.
60(6): p.
541-549.
Heinrich,
A.,
et al.,
In vitro stent assessment by MRI: visibility of lumen and artifacts for 27 modern stents. Biomedical Engineering/Biomedizinische Technik,
2017.
62(6): p.
565-573.


![Fig. 2: The maximum artifact size was measured with the software MR-Susceptibility Artifact Measurement (SAM) using histogram-based reference value. The graphical representation of all artifact sizes automatically measured in vertical (red) and horizontal (blue) orientation for a rectangular measurement field. See references [1-3] for more details. References: Andreas Heinrich, Department of Radiology, University Hospital Jena - Jena/DE](https://epos.myesr.org/posterimage/esr/ecr2019/147437/media/782833?maxheight=150&maxwidth=150)