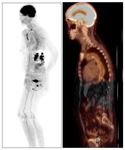
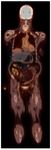
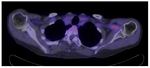
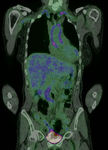
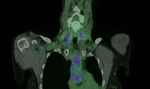
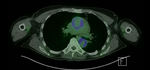
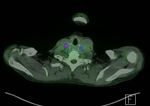
In LVV,
ongoing inflammation leads to locally increased metabolic demands resulting in an increased cellular uptake of a radiolabelled glucose analogue (18F-FDG).
This is reflected by increased radiotracer uptake in the walls of affected vessels.
The affected large vessels include:
· Aorta and its main branches,
especially within the thorax
· Internal and external carotid arteries and their branches
GCA primarily affects extracranial large vessels with or without involvement of the cranial vessels.
TA predominantly effects the aorta and its major branches and pulmonary arteries.
Radiotracer uptake in LVV is contiguous and symmetric in paired vessels [12] although can become patchy in the later stages of TA.
Thoracic vessels are more frequently affected than abdominal vessels in GCA.
Interpretation relies mostly on qualitative methods using a visual grading scale based on vessel-to-liver uptake ratio [13,
14].
Semiquantitative analyses using standardised uptake value (SUV) metrics are based on the evaluation of FDG uptake in different vascular regions (thoracic aorta,
abdominal aorta,
subclavian arteries,
axillary arteries,
carotid arteries,
vertebral arteries,
pulmonary arteries,
iliac arteries,
and femoral arteries) however the clinical utility of this for the initial diagnosis of LVV is currently unknown [13,
14].
The pattern,
localisation and intensity of radiotracer uptake helps differentiate between sterile inflammation,
infection and atheroma formation [9] with focal uptake more likely to represent infection or atheroma formation,
especially if restricted to one vessel.
LVV complications such as vascular stenoses and aneurysm formation can also be identified on contrast-enhanced PET/CT.
Advantages of 18F-FDG PET in LVV
1. Non-invasive procedure
2. Overall good diagnostic accuracy in the diagnosis of LVV [10]
3. Early disease detection
Disadvantages of 18F-FDG PET in LVV
1. Unreliable in diagnosing LVV in patients on steroids [5]
2. Lack of standardisation in image acquisition and interpretation to date [14]
3. Paucity of data supporting its use in follow-up of disease