Nutcracker syndrome and/or nutcracker phenomenon have a pathophysiology that depends on aetiological factors.
They determine compression of LRV with increased ipsilateral renal venous pressure and,
consequently,
break of thin-wall varices in the collector system.
In an early phase,
collateral veins are developed and increase venous capacity; in this phase the continence of ovarian and lumbar vein valves is maintained.
This anatomic situation results in clinical manifestations: flank pain,
proteinuria,
micro/macrohaematuria (occasionally with anemia requiring blood transfusions).
Abdominal pain in left side and in pelvis may be present and it is the second most common symptom.
Pain is exacerbated by sitting,
standing or riding a moving vehicle.
Left-sided colic pain may also be due to passage of blood clots in left ureter.
Nausea and vomiting may also be present for possible compression of splanchnic veins [1,3].
In late stage chronic hypertension in LRV causes valvular incontinence and massive blood reflux in ovarian vein or in lumbar veins.
In this pathologic stage in women,
pelvic and vulvar varices may occur with the characteristic symptoms of pelvic congestion syndrome Fig. 1 .
Pelvic congestion syndrome is defined as chronic pelvic pain for more than 6 months secondary to pelvic venous insufficiency and with associated pelvic,
vulvar and gluteal varicose veins,
dysmenorrhea,
dyspareunia,
post-coital pain and dysuria.
The presence of left varicocele and varicose veins of lower limbs is common in the late phase of nutcracker phenomenon.
Pathophysiology of varices may be associated with thin walls veins,
lack of valves,
collagen abnormalities,
hormonal and haemodynamic factors in pregnancy.
The smooth muscle layer in ovarian vein undergoes thinning and reduced contractility with age [4].
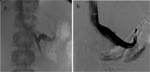
Diagnosis is confirmed by results of imaging using Doppler ultrasonography (DUS),
CT,
MRI,
intravascular ultrasound and retrograde venography [5] Fig. 2 .
However,
diagnosis of NCS can be difficult relying only on imaging; clinical signs and symptoms have a primary importance role in management options. In the diagnosis of NCS,
real-time DUS has a high sensitivity and specificity,
respectively of 69% and 90% and 89% and 100%.
In a study of 23 children with nutcracker syndrome and 26 healthy patients,
Fidoz et al.
demonstrated that orthostatic imaging reveals comparatively narrower angles between aorta and SMA and more pronounced trapping results in patients with nutcracker syndrome [5,6]. Where DUS is not diagnostic,
CT and MRI imaging may be necessary; these methods can demonstrate the compression of LRV between SMA and abdominal aorta,
varicocele and pelvic congestion.
However CT and MRI are not dynamic methods and they don’t allow to measure flow velocity and direction accurately. A characteristic feature in CT or MRI axial images is the "beak sign" and the diameter of LRV (Figure 3) Fig. 3 .
Although CT is not an invasive test,
it exposes to ionizing radiation.
MRI doesn’t use radiation and it is multiparametric.
However results of diagnostic accuracy between MRI and CT are similar.
If diagnosis of NCS remains unclear,
it can be confirmed by venography or intravascular ultrasound by measuring venous pressure gradient between LRV and IVC.
Both diagnostic procedures still remain the "gold standard" in diagnosis of NCS,
despite being invasive methods.
Normally aorto-mesenteric angle is about 90° in healthy individuals,
with angles less than half in patients with nutcracker phenomenon.
This angle varies depending on position of the patient with increased compression of LRV by SMA in an upright position.
Normal pressure gradient between LRV and IVC is = <1 mm Hg.
We speak of hypertension of LRV when gradient is greater than or equal to 3.0 mm Hg.
Diameter of normal left gonadal vein is about 3 mm.
LRV has a length between 6 cm and 10 cm and,
unlike right renal vein,
receives blood from the left adrenal veins,
left gonadal,
ureteral and lumbar veins before they flow in IVC.
These tributary veins have valves that prevent reflux of blood,
but in presence of renal venous hypertension and incontinence of valves can occur varicocele [1].
Treatment in symptomatic patients may be surgical or endovascular by LRV stenting [1,3].
In 1974 Pastershank reported the first case of NCS surgical treatment [7].
Management options range from observation to nephrectomy depend on severity of symptoms.
Up to 18 years,
conservative approach is recommended with observation of at least 2 years,
because spontaneous resolution of haematuria is obtained in 75% of cases [8].
Use of angiotensin inhibitors is recommended in patients with NCS with orthostatic proteinuria [9].
Interventional or surgical treatment should only be considered when symptoms are severe or persistent: pain,
severe haematuria,
renal insufficiency and failure of responce to conservative treatment after 24 months [1].
Most interventions aim to decrease hypertension in LRV,
others to prevent pelvic venous reflux.
Various surgical approaches have been made including medial nephrectomy with renal excision,
LRV bypass,
transposition of LRV between SMA and aorta,
SMA transposition,
renal-IVC shunting,
renal autografts,
gonado-caval bypass and also nephrectomy for persistent haematuria; they are only some possible surgical treatment [10,
11,
12,
13].
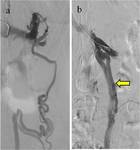
Endovascular stent is an alternative treatment option Fig. 4, Fig. 5 . It may be preferred to surgery because of the long period of renal congestion and the possibility of post-surgical complications,
paralytic ileus,
hematoma or retroperitoneal hemorrhage and persistent pelvic pain after surgery.
Complications after stent placement are rare: thrombosis,
migration,
stent rupture and restenosis.
The procedure is generally performed under local anesthesia; common femoral vein or right internal jugular vein is picked percutaneously and a vascular introducer is positioned.
Then the selective catheterization of LRV is performed with aid of a guide and an angiographic catheter.
In NCS during angiography there is evidence of stagnation of contrast medium in LRV,
reflux in its collaterals,
pelvic varicocele and a renocaval gradient greater than 3 mmHg.
One of main technical points is type of stent to be used and in particular the choice of diameter can be an important factor for risk of migration [14,
15].
Quevedo et al.
have reported more than 150 cases of endovascular stenting performed successfully in patients with anterior nutcracker syndrome with resolution of symptoms.
Stents from 10 mm to 20 mm in diameter and from 40 mm to 60 mm in length were used.
Choice of stent size is highly dependent on accurate measurements of LRV in intravascular ultrasound prior to placement,
and is usually oversized up to 20% to prevent migration.
Ideal stent should have a high radial force with little shrinkage in length to allow accurate positioning [16].
In patients with posterior nutcracker variant,
it is reported that after undergoing transposition of LRV,
patients still had a 20% reoperation rate with endovascular approach due to stenosis or LRV thrombosis.
Therefore,
some authors propose an immediate stenting approach at time of transposition of LRV,
preventing collapse or thrombosis,
to avoid reoperation [17].

![Fig. 2: Diagnostic flow-chart of nutcracker syndrome based on initial symptoms.
[K. Ananthan, S. Onida, A.H. Davies. Nutcracker Syndrome: An Update on Current Diagnostic Criteria and Management Guidelines. Eur J Vasc Endovasc Surg (2017) 53, 886 e 894].
References: [K. Ananthan, S. Onida, A.H. Davies. Nutcracker Syndrome: An Update on Current Diagnostic Criteria and Management Guidelines. Eur J Vasc Endovasc Surg (2017) 53, 886 e 894].](https://epos.myesr.org/posterimage/esr/ecr2019/147684/media/826216?maxheight=150&maxwidth=150)