Several lesions and pseudolesions can simulate pancreatic primary neoplasms.
In this presentation they are divided in four main groups:
1- Pseudolesions related to pancreas developmental anomalies and normal variants;
2- Lesions arising from adjacent pancreas structures;
3- Inflammatory pancreatic and peripancreatic lesions;
4- Pancreatic metastasis.
1- Pseudolesions related to pancreas developmental anomalies and normal variants.
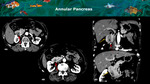
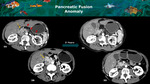
Pancreatic fusion or migration anomalies (Figures 3,
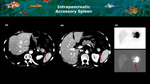
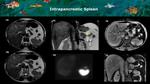
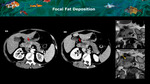
4 and 5) and normal variants such as accessory splenic tissue (Figures 6 and 7) or fat deposition (Figure 8) may give rise to imaging appearances that mimic primary pancreatic neoplasia,
including pancreatic adenocarcinoma and neuroendocrine tumors.
Also,
the patients with pancreatic developmental anomalies and normal variants may have predisposal for specific pancreatic and peripancreatic disease,
which makes the correct diagnosis important in these cases.
Knowledge of pancreatic embryology (Figure 3) and of normal anatomic variants is essential to identify these entities and to help differentiate them from pathologic conditions,
thus preventing potential unnecessary imaging investigation or more invasive procedures such as biopsy or surgery.
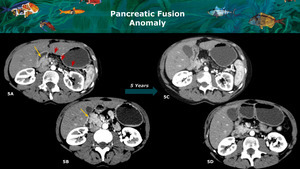
Fig. 5: 5A and 5B: CT axial images on arterial phase show normal appearance of the pancreatic head and uncinate process (yellow arrows), and diffuse fat replacement of the body and tail (red arrows) with an abrupt change, findings that should raise suspicion for a pancreatic neoplasia. Note there is no main pancreatic duct dilatation. In a follow-up scan 5 years later, no significant changes were seen (CT axial views on arterial phase images - 5C and 5D). Those findings suggest a pancreatic fusion anomaly with fat replacement of the dorsal pancreas and preservation of the ventral parenchyma.
2- Lesions arising from adjacent pancreas structures.
A variety of lesions from surrounding structures,
especially large or infiltrative lesions,
can be mistaken for primary pancreatic lesions.
They may originate from [8]:
-
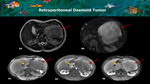
Retroperitoneum (Figure 9);
-
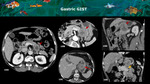
Gastrointestinal tract (Stomach - Figure 10,
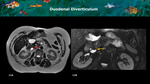
Duodenum - Figure 11,
Small Bowel,
Colon);
-
Liver (Figure 12);
-
kidney;
-
Adrenal gland;
-
Nervous plexus.
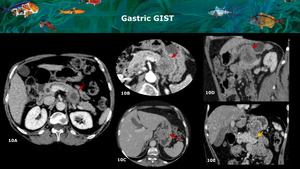
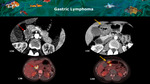
Fig. 10: A and B: CT axial show a partially necrotic solid mass within the pancreatic body / tail (red arrows) simulating an adenocarcinoma. C, D and E: CT axial, sagittal and coronal reconstructions show that the lesion was arising from gastric small curvature wall (red arrows) and compressing pancreatic parenchyma (yellow arrow), that remains with normal enhancement and size. This demonstrates the importance of using reconstructions in CT to analyze pancreas relations and identify the real origin of the lesions.
Many tips and tricks can help radiologists to identify the correct origin of a peripancreatic lesion.
Foremost,
we must be aware of the normal aspect of pancreatic parenchyma in ultrasound,
CT and MRI images.
Also,
CT multiplanar reconstructions can provide more accurate views of the anatomy and adjacent organs.
Certain nodal chains when enlarged and involved in neoplastic,
inflammatory,
or infectious disorders may mimic lesions of the pancreas (Figure 13).
Most common locations for lymph nodes to be misdiagnosed as a pancreatic lesion are portocaval,
portal,
peripancreatic,
and celiac chains [8].
3- Inflammatory pancreatic and peripancreatic lesions
Pancreatic and peripancreatic fluid collections are common complications of acute pancreatitis however they might simulate cystic pancreatic lesions (Figure 14).
Facing these findings without a consistent history of a previous pancreatitis is challenging and the possibility of a neoplastic lesion cannot be ruled out.
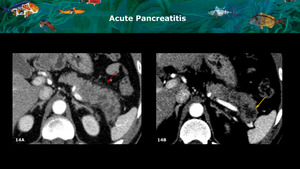
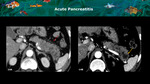
Fig. 14: A and B: CT arterial phase axial images show peripancreatic fat stranding (red arrow), and enlargement of the pancreatic tail, with intrapancreatic hypovascular or necrotic areas (yellow arrow). This patient had a history of abdominal pain for over 4 weeks and elevated serum amylase and lipase, confirming the hypothesis of an acute pancreatitis. It is important to highlight that without those informations the differential diagnosis with a cystic neoplasm is not possible.
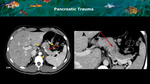
Traumatic pancreatitis and its complications like fistula,
pseudocysts and abscesses may also present as a pseudotumor [5] (Figure 15).
Thus,
in the diagnosis work-up of cystic lesions within or nearby the pancreas,
the clinical history of pancreatitis or blunt trauma and follow-up scans are fundamental for the correct diagnosis.
The main findings of chronic pancreatitis in imaging are dilation of the main pancreatic duct,
atrophy of the parenchyma and presence of ductal calculi [4].
The parenchyma involvement often present asymmetrically or focally,
making differential diagnosis with a solid pancreatic neoplasm very difficult.
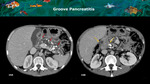
The classic example is paraduodenal groove pancreatitis (Figure 16),
a common entity in patients with chronic pancreatitis that consists of the presence of an inflammatory tissue occupying the duodenal groove adjacent to the pancreas head.
In these cases the differential diagnosis by imaging is practically impossible and it becomes necessary to proceed with an endoscopy ultrasound with cytology samples or with imaging follow-up.
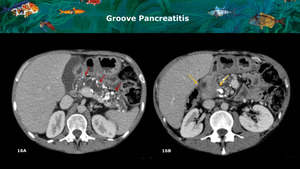
Fig. 16: A: CT axial portal phase images show chronic pancreatitis characterized by main and side-branch duct dilation with multiple calculi and parenchymal atrophy (red arrows). B: An hypovascular solid mass is identified occupying the paraduodenal sulc (yellow arrows), raising the suspicion of pancreas adenocarcinoma. This patient was submitted to surgical resection due to abdominal pain and no neoplastic cells were found.
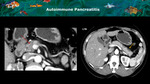
- IgG4-related autoimmune pancreatitis
Pancreas is the most commonly abdominal organ affected on the IgG4-related disease.
This type of autoimmune pancreatitis consists of a pancreatic and peripancreatic fat lymphoplasmacytic infiltrate containing IgG4 and may be present diffusely or focally,
in this case simulating pancreatic neoplastic tissues (Figure 17) [4].
Some classic imaging features favoring this diagnosis are diffuse pancreatic enlargement with loss of contours,
irregularities of the main pancreatic duct and presence of peripancreatic inflammatory tissue (typically seen as an hypovascular halo) and might help in the differential diagnosis with neoplasia.
The finding of multiple organ involvement (being the biliary tree the second most common in the abdomen) is very helpful for the diagnosis,
as well as the response to corticosteroids therapy [4].
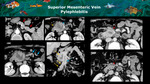
This condition usually occurs secondary to any abdominal inflammation or infection,
leading to a septic thrombosis in the porto-mesenteric system.
Common causes are acute diverticulitis,
appendicitis or cholangitis
On CT and MRI images,
a solid and heterogeneous hypovascular mass along the portal system is seen,
simulating a neoplastic lesion (Figure 18)
Clinical informations of previous acute abdominal conditions and systematic evaluation of the abdomen are essential for the correct diagnosis.
4- Pancreatic metastasis
Unlike primary pancreatic carcinoma,
metastatic lesions to the pancreas are uncommon and account for approximately 2% of all pancreatic malignancies [6].
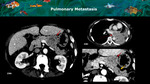
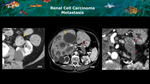
The most common primary tumours with pancreatic metastases are lung cancer (Figure 19),
breast cancer,
renal cell carcinoma (Figure 20),
malignant melanoma,
and carcinoma of the GI tract.
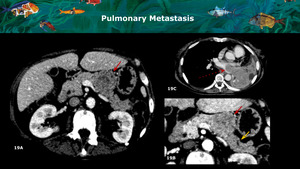
Fig. 19: A and B: Axial CT images on portal venous and arterial phase demonstrate a pancreatic body hypovascular mass (red arrows) proven to be a solitary pulmonary metastasis. Note the abrupt change in the parenchymal attenuation distal to the lesion (yellow arrow) and the normal parenchyma in the head, findings that can also be found in pancreatic adenocarcinoma.
C: Pulmonary infiltrative mass corresponding to the primary tumor (red dotted arrow).
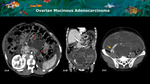
Other primary malignancies less common that have been reported are thyroid,
prostate,
ovarian (Figure 21),
hepatocellular carcinomas,
and many types of sarcomas [6].
In most cases,
the definition of a secondary pancreatic tumour instead of a primary adenocarcinoma is very difficult,
due to the variety of imaging features they may present.
However,
some features could help distinguishing metastatic from primary cancer in the pancreas.
Whereas adenocarcinoma of the pancreas typically appears as a ill-defined hypovascular mass at contrast-enhanced CT,
a large proportion of pancreatic metastases present as heterogeneous or hypervascular lesions [6].
Also,
the presence of multiple lesions is not characteristic of primary pancreatic carcinoma.
Presence of metastases at sites not typically involved in adenocarcinoma of pancreatic origin (such as skeleton or adrenal glands) should also suggest the possibility of metastatic disease of the pancreas [7].