Immunotherapy and irAEs
Immunotherapy has emerged as a promising treatment option for advanced cancer and is now considered standard of care for multiple cancer.
Immune checkpoint inhibitors have shown remarkable beneficial effects in several cancer types,
including lung cancer,
kidney cancer,
melanoma,
head-and-neck cancer,
bladder cancer,
lymphoma and Merkel cell cancer.
Currently approved drugs in this class include:
- CTLA-4 inhibitor: Ipilimumab
- PD-1 inhibitors: pembrolizumab and nivolumab
- PD-L1 inhibitors: durvalumab,
atezolizumab,
and avelumab.
The anticancer activity of immune checkpoint inhibitors therapy is due to the blockade of immune inhibition by tumors,
that activates T-cell-mediated immune responses against tumor.
Immune checkpoint inhibitors are monoclonal antibodies that target natural immune checkpoints such as cytotoxic T-lymphocyte antigen 4 (CTLA-4),
programmed death 1 (PD1),
and PD ligand 1 (PDL1).
Such checkpoints prevent the development of antihost immune effects,
which would otherwise result in clinical autoimmune disease,
thus checkpoint inhibitors might lead to enhanced antitumour immunity.
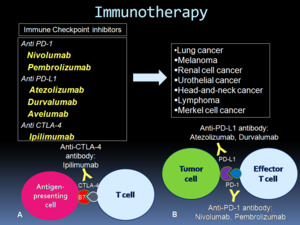
Fig. 1: Immune checkpoint inhibitor therapy. A: CTLA-4 inhibitors by binding to CTLA-4 on T cells and blocking T-cell immune inhibition, activate an immune response against tumor cells. B: PD-1 or PD-L1 inhibitors block the binding and prevent tumoral immune inhibition, inducing an antitumor immune response. Diagram modified from Nishino et al. Radiographics. 2017 Sep-Oct;37(5):1371-1387.
However,
these drugs might also heighten immune activity against self-antigens,
resulting in immune-related adverse events (irAEs) in a substantial proportion of patients.
In terms for drug safety assessment,
adverse events (AEs) mean an unexpected medical problem that happens during treatment with a drug or other therapy.
Adverse events might be mild,
moderate or severe and may be caused by something other than the drug or therapy being given.
The irAEs include a group of complications related to immune checkpoints inhibitors that involve different organs throughout the body,
including skin,
brain, pituitary,
eye,
thyroid,
liver,
adrenal,
kidney,
pancreas,
colon,
and lung.
Examples of irAEs include hypophysitis,
thyroiditis,
pneumonitis,
hepatitis,
pancreatitis,
and colitis.
Among these,
pneumonitis is a relatively rare but clinically serious and potentially life-threatening toxicity.
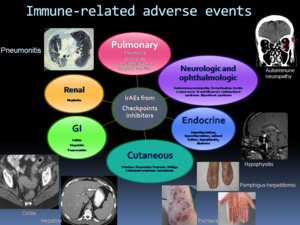
Fig. 2: The spectrum of immune-related adverse events reported with checkpoint inhibitor therapy. Modified from Suozzi KC et al. JAAD Case Rep. 2016 Jul 14;2(3):264-8.
Immune-related pneumonitis during immunotherapy
Pneumonitis related to immune checkpoints inhibitors is an emerging adverse event with special clinical significance.
Large meta-analyses of randomised trials and retrospective studies using anti-PD1 and PDL1 antibodies across melanoma,
lung cancer,
and renal cancer found an overall incidence of pulmonary toxicity of 2,7–3,5%.
Risk of immune-related pulmonary toxicity might vary according to tumor type,
with highest incidence among patients with lung cancer (NSCLC) and incidence particularly low among individuals with melanoma.
NSCLC are also at higher risk of developing high-grade (grade 3 or above) pneumonitis and even fatal toxicities and renal cell carcinoma have a higher incidence rate for all-grade pneumonitis when compared with melanoma.
The median time to onset of pulmonary toxicity after initiation of immunotherapy is 2,3 months,
and tends to occur earlier in lung cancer (2,1 months) than melanoma (5,2 months).
However,
it is now apparent that irAEs,
including pneumonitis,
might occur at any point during treatment.
irAEs are unpredictable.
Although,
it is unclear who will develop pulmonary toxicity or how long they will last,
there are some characteristics that have been associated with higher risk of pulmonary irAEs.
To date,
proposed risk factors include combination immunotherapy,
coadministration of radiation therapy,
impaired baseline pulmonary function,
previous heavy smoking or continued smoking,
and previous high-dose chemotherapy.
However,
these clinical factors are unlikely to sufficiently discriminate among risk groups.
The possibility to predict risk of pneumonitis would allow clinicians to incorporate these factors into treatment decisions.