Diagnostic approach to thoracic irAEs
The diagnostic approach to irAEs requires multidisciplinary effort by pulmonologists,
medical oncologists,
and radiologists.
Clinical manifestations
While some immune-related adverse events can be defined and characterized by quantifiable laboratory values; for example,
immune thyroid,
pituitary,
or adrenal effects manifest as changes in thyroid-stimulating hormone,
free thyroxine,
adrenocorticotropin,
and cortisol; immune-related pulmonary toxicities clinically manifest with relatively non-specific features.
Clinical manifestations might be quite difficult to distinguish from disease-related complaints or other treatment-related complications such as infection or anaemia.
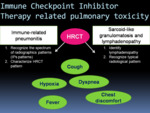
Symptoms might include dyspnoea,
chest discomfort,
cough,
or,
less commonly,
fever.
Hypoxia might develop and,
in some cases,
progress rapidly.
Fig. 3: Pulmonary toxicity of cancer immunotherapy: immune-related pneumonitis, sarcoid-like granulomatosis and lymphadenopathy. Immune-related pulmonary toxicities often present with non-specific clinical manifestations. Radiologists play a mayor role in diagnosing pulmonary complications of immunotherapy.
Pulmonary function testing
Irrespective of the specific radiologic features,
irAEs most commonly show a restrictive pattern,
featuring reduced diffusion capacity,
in pulmonary function tests. Some studies suggest routine pulmonary function testing,
might be helpful in screening for immune-related pulmonary toxicity during treatment with checkpoint inhibitors.
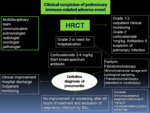
Radiologic findings in immune related-pneumonitis
Pulmonary toxicity of cancer immunotherapies is a new important clinical event that requires prompt identification and management.
Imaging is a key component of evaluating pulmonary irAEs and radiologists play a major role in detecting and monitoring these adverse events.
Once suspected,
HRCT is the preferred diagnostic study,
because chest radiography fails to detect up to one-quarter of these cases.
In this context,
HRCT can be used as a imaging biomarker to assess drug toxicity.
The radiologic manifestations of the irAEs in the lung comprises a broad and overlapping spectrum of pulmonary manifestations.
Radiologic patterns of immune-related pneumonitis have been characterized according to the American Thoracic Society (ATS)/European Respiratory Society (ERS) classification of interstitial pneumonias.
Recognition of the radiologic patterns of penumonitis is the first step in the image-based approach.
Radiologic patterns associated with immune related pneumonitis include:
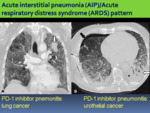
-Acute interstitial pneumonia (AIP)/acute respiratory distress syndrome (ARDS) pattern
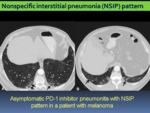
-Nonspecific interstitial pneumonia (NSIP) pattern
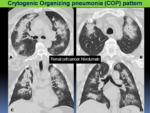
-Cryptogenic Organizing pneumonia (COP) pattern
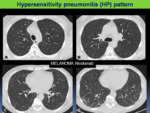
-Hypersensitivity pneumonitis (HP) pattern

Fig. 4: Radiologic patterns and CT features according to the ATS/ERS classification of interstitial pneumonias associated with immune related pneumonitis.
The clinical course and the radiographics patterns are variable among patients.
Some patients have a fulminant clinical course with rapidly worsening respiratory that require admission to the intensive care unit and intubation with AIP/ARDS pattern on chest CT.
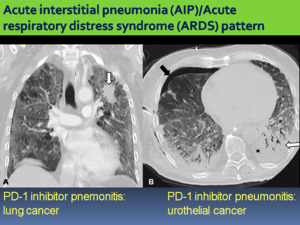
Fig. 5: A. Pneumonitis with AIP/ARDS pattern in a 65-year-old patient with lung cancer in the left upper lobe (white arrow) treated with nivolumab. Coronal chest CT image obtenied shows bilateral and diffuse GGOs. B. Pneumonitis with AIP/ARDS pattern in a 70-year-old patient with urothelial cancer. Onset of acute respiratory failure and fever after 1 week of starting pembrolizumab. Axial chest CT image shows right pneumothorax (black arrow) with thoracic tube, pleural effusion (asterisk), basal consolidations (white arrow), bilateral and diffuse GGOs and lung volume loss.
Other patients have a milder clinical course with CT findings indicative of NSIP,
COP and HP patterns.
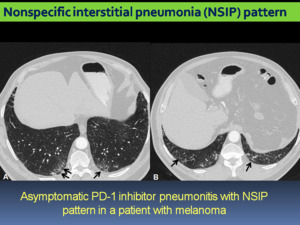
Fig. 6: Pneumonitis with NSIP pattern in an asymptomatic 45 year-old-patient with melanoma treated with nivolumab. A, B: axial chest CT images show subtle reticular and ground-glass opacities in a predominantly peripheral and basilar distribution.
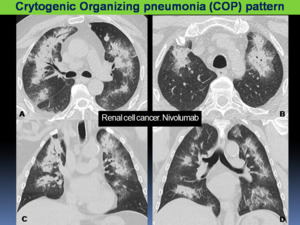
Fig. 7: PD-1 pneumonitis: COP pattern in a 62 year-old-patient with renal cell cancer after 5 cycles of nivolumab. Axial (A, B) and coronal (C, D) chest CT showing multifocal areas of consolidation with peripheral ground-glass opacities, predominantly with peripheral distribution. Bronchial dilatation was noted within the areas of consolidation.
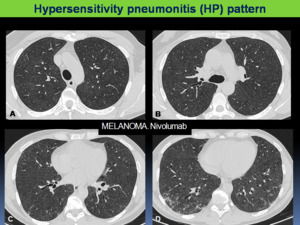
Fig. 8: Pneumonitis with hypersensitivity pneumonitis pattern in a 40-year-old patient with melanoma treated with nivolumab. The patient presented with new dysphnea and cough, without fever. A-D: Axial chest CT images show diffuse centrilobular ground-glass opacities and subtle reticular opacities of basal location.
These patients are often successfully treated with corticosteroid therapy and some of them are able to restart the immunotherapy.
A recent study that investigated a cohort of 20 patients treated in 10 nivolumab trials with pneumonitis noted a spectrum of radiologis patterns of COP,
NSIP,
HP and AIP/ARDS patterns in decreasing order of presentation (3).
Moreover,
these radiologic patterns had a signifficant association with the clinical severity of pneumonitis,
from AIP/ARDS being the highest grade (grade 3),
followed by COP (grade 2),
NSIP (grade 1) and HP (grade 1) patterns,
having the last two lower grades (3).
These results indicate that imaging can have an important role in the initial evaluation of patients with suspected pneumonitis,
guiding management according to disease severity.
Additionally,
thoracic (including hilar and mediastinal) adenopathy with sarcoid-like granulomatosis might develop in the setting of autoimmune pulmonary toxicity,
particularly with CTLA-4 inhibitor ipilimumab.
CT findings include enlarged mediastinal and hilar lymphadenopathy,
which may also be accompanied by parenchymal changes simulating features of sarcoidosis,
as micronodules with a perilymphatic distribution.
The entity can be clinically silent and can resolve spontaneously.

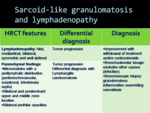
Fig. 9: Sarcoid-like granulomatosis and lymphadenopathy. HRCT typical features, differential diagnosis and diagnosis approach.
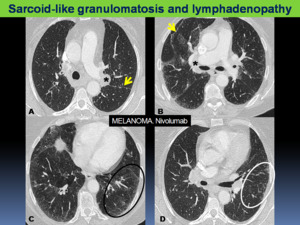
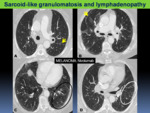
Fig. 10: Sarcoid-like granulomatosis of the lung with hilar lymphadenopathy in a patient with melanoma who was treated with nivolumab. A-D. Axial chest CT images show typical perilymphatic distribution of micronodules with irregular thickening of fissures (yellow arrows), patchy ground-glass opacities (black circle) and micronodules (white circle). Hilar lymphadenopathy are also present (asterisks).
Radiological differential diagnosis
Pulmonary toxicity has a range of differential diagnosis of infectious and neoplastic processes that might make the diagnostic process challenging for radiologists.
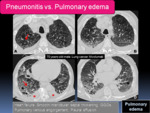
Also pulmonary edema can be confused with pneumonitis.

Fig. 11: Challenging of imaging interpretation of pneumonitis: differential diagnosis.
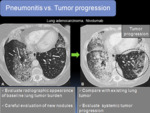
In patients with advanced cancer with lung baseline involvement,
it is difficult to distinguish pneumonitis from tumor progression.
In some cases,
OP pattern might appear as pulmonary nodules,
which presents a diagnostic dilemma because such nodules can be confused with new or worsening metastatic disease.
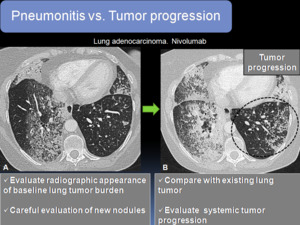
Fig. 12: Tumor progression in a patient with lung adenocarcinoma. A. Axial CT image showing baseline lung tumor burden with patchy areas of consolidation and GGOs and micronodules in left lower lobe (LLL). In the follow-up the patient presented with progressive dyspnea. B. Axial CT image showed increase in baseline radiological findings with increase in the size and number of micronodules in the LLL (circle), indicative of tumor progression.
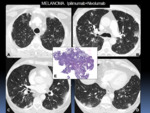
It is also challenging to differentiate sarcoid-like granulomatosis and lymphadenopathy from metastases and tumor progression.
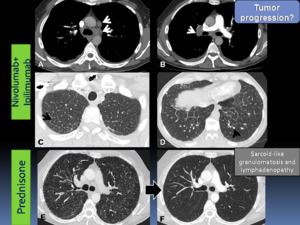
Fig. 13: Sarcoid-like granulomatosis and lymphadenopathy in an asymptomatic 45 year-old-patient with melanoma who was treated with nivolumab+ipilimumab. Axial chest CT images (A-B) mediastinal window showing new mediastinal and right hilar lymphadenopathy (white arrows); bilateral pleural effusions were also present. (C-D) Axial CT images in lung parenchyma window showed diffuse septal thickening (black arrow) and micronodules (arrowhead). Tumoral progression was suspected. Transbronchial biopsies were performed showing sarcoid-like reaction. Patient underwent corticosteroid therapy. E. Early follow-up CT showed radiological worsening with multiple micronodules in a perilymphatic distribution resembling sarcoidosis. F. Axial chest CT obtained 3 months after the initiation of prednisone showed complete resolution of findings.
Accurate evaluation of the CT appearance of baseline lung tumor burden and also assessment of extrathoracic tumor burden are often the clues for distinguish cancer progression from pneumonitis.
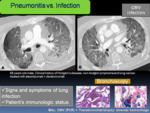
Exclusion of pulmonary infection is an important step in almost all cases of pneumonitis.
It requires clinical correlation with detailed history of symptoms and immunologic status of the patients.
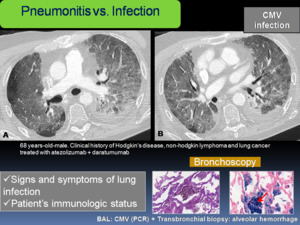
Fig. 14: 68 years-old-male with clinical history of Hodgkin's disease, non-hodgkin lymphoma and lung cancer treated with atezolizumab + daratumumab, presented with increased shortness of breath, cough and fever. A-B: Axial chest CT showed diffuse GGOs, bronchial dilatation and left pleural effusion. A bronchoscopy with transbronchial biopsies was performed. Cytomegalovirus (CMV) was detected by PCR in bronchoalveolar lavage (BAL) and signs of alveolar hemorrhage were noted on biopsies, indicating Cytomegalovirus-Induced Alveolar Hemorrhage. C. Transbronchial biopsy with intraalveolar hemorrhage with abundant hemosiderophages. H & E. D. Transbronchial biopsy stained with Perls, to demonstrate alveolar hemosiderophages (red arrow).
Pulmonary edema is another frequent complication in cancer patients,
sometimes due to fluid overload during treatments.
Radiological signs of edema can be confused with pneumonitis.
Recognizing the radiological findings of heart failure is essential in the differential diagnosis,
particularly new smooth interlobular septal thickening,
pulmonary venous engorgement and the presence of pleural effusion.
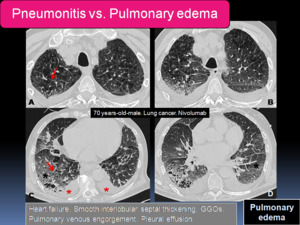
Fig. 15: 70 years-old-male with lung cancer after 3 cycles of nivolumab presented with increased dyspnea. A-D: Axial chest CT images show bilateral GGOs (black asterisks), smooth interlobular septal thickening (red arrows) and bilateral pleural effusions (red asterisks). The patient was admitted to the intensive care unit and eventually died despite heart failure treatment.
Cytological and pathological features
In some cases,
it is necessary to perform a cytological and pathological evaluation by bronchoscopy that might discriminate infection and lymphangitic spread from the inflammatory changes characteristic of pneumonitis.
Bronchoalveolar lavage fluid might show inflammatory and lymphocytic infiltration.
A biopsy (bronchoscopic or criobiopsy) might show histopathological findings that can include cellular interstitial pneumonitis,
organising pneumonia,
and diffuse alveolar damage.
In more than one-quarter of cases,
no pathological abnormalities are identified.
In cases of suspected sarcoid-like granulomatous reactions,
histological sampling is required for diagnosis demonstrating granulomatous inflammation resembling sarcoidosis.
Interstitial fibrosis have also been reported.
A combination of diagnostic clues at imaging and clinical dialogue in a multidisciplinary context can contribute to accurate diagnosis and optimal patient care during immunotherapy.
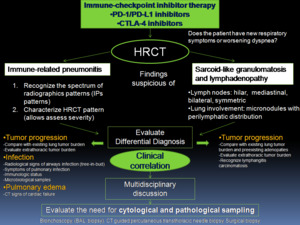
Fig. 16: Diagnostic approach to thoracic immune-related adverse events. Modified from Nishino et al. Radiographics. 2017 Sep-Oct;37(5):1371-1387.
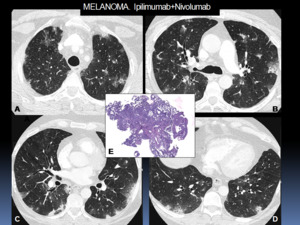
Fig. 17: 50 year-old-patient with melanoma treated with Ipilimumab+Nivolumab combination therapy. A-D: Axial chest CT images show patchy consolidation with sorrounding ground-glass opacities (halo sign) with a predominantly subpleural and peribronchial distribution. Pulmonary infection was excluded by bronchoscopy (negative microbiology tests in bronchoalveolar lavage). A transbronchial biopsy was performed in order to rule out progression disease. E: The histologic sampling showed mild interstitial thickening and early signs of organization, indicative of organizing pneumonia.
Management
In cases of clinical and radiological suspected immune-related pulmonary toxicity,
management should be started immediately,
without waiting for pathological evaluation.
The initial step in management is withholding immune checkpoint inhibitor treatment.
For patients with asymptomatic pneumonitis,
this might be sufficient.
Generally,
patients require also corticosteroid therapy either orally or intravenously,
depending on disease severity.
In cases refractory to steroids,
additional immunosuppressive agents might be considered,
including infliximab,
cyclophosphamide,
or mycophenolate mofetil.
We present the multidisciplinary management algorithm of pulmonary immune-related adverse event proposed in our hospital.
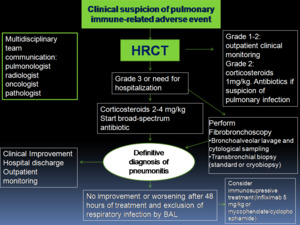
Fig. 18: Multidisciplinary management algorithm of pulmonary immune-related adverse events proposed in our hospital.
BAL: bronchoalveolar lavage.
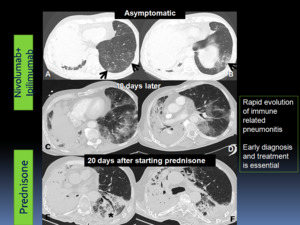
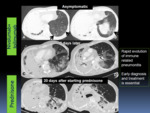
Fig. 19: Rapid evolution of immune related pneumonitis in a patient with pulmonary oat cell carcinoma treated with nivolumab and ipilimumab combination therapy. A-B: Axial chest CT images showed new subtle GGOs in predominantly peripheral and basilar distribution (A-B) in an asymptomatic patient. The patient presented new dyspnea and cough and visited the emergency department 10 days after the first chest CT. C-D: A new chest CT showed increase of GGOs. Infection was excluded and the patient underwent corticosteroid therapy. E-F: 20 days after starting prednisone, a chest CT showed decreased GGOs and the presence of basal and peribronchial consolidations with bronchial dilatation, indicative of organizing pneumonia.
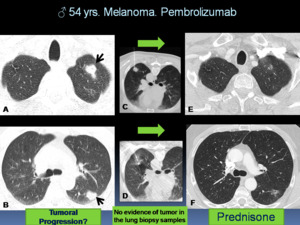
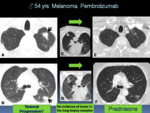
Fig. 20: Pulmonary ir-AEs in a 54 years-old-patient with melanoma who was treated with pembrolizumab. A-B: Axial CT images show new pulmonary nodules (arrows). Tumoral progression was suspected. Lung nodules biopsies were performed. C: fine-needle aspiration biopsy, D: core-biopsy, with no evidence of tumor in the biopsy samples. E-F: Axial CT images obtained 1 month after the starting prednisone showed a decrease in size and density of pulmonary nodules.
As a result of the high doses and prolonged duration of corticosteroid therapy,
opportunistic infections might occur; in this case,
pneumocystis prophylaxis might be administrated.
Patients with recurrence of symptoms while receiving treatment with corticosteroids or immunosuppressive agents,
an infectious cause should be considered.
Repeat endoscopy with bronchoalveolar lavage might be indicated in this context.
Restart immune checkpoint inhibitors therapy after pneumonitis is an area of debate.
One-third of patients are able to start immunotherapy after a successfully treated pneumonitis,
however it has been reported 25-28% of recurrent pneumonitis during retreatment.
Moreover,
pneumonitis may recur after finishing a taper of corticosteroid therapy,
in the absense of restarting immunotherapy.
This phenomenon termed pneumonitis flare may involve underlying autoimmune mechanism.
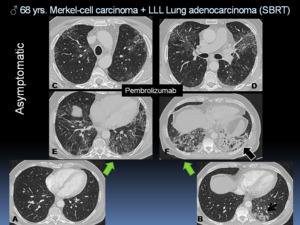
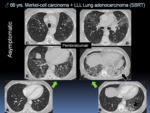
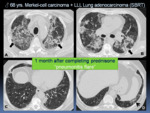
Fig. 21: A-B. Pneumonitis in an asymptomatic 68 year-old-patient with Merkel-cell carcinoma and left lower lobe (LLL) pulmonary adenocarcinoma treated with Stereotactic Body Radiotherapy (SBRT) (short arrow). The patient received immunotherapy (pembrolizumab). C-F. A routine chest CT follow-up during treatment showed new GGOs with a peribronchial distribution and a left lower lobe consolidation (black arrow), suspicious of immune related pneumonitis. The patient underwent corticosteroids treatment.
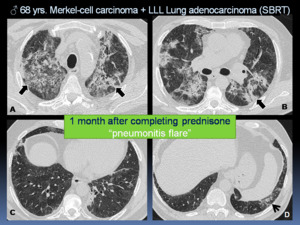
Fig. 22: Follow up of the previous case (Figure 21). After corticosteroid therapy was tapered, the patient presented with a new cough and dyspnea. Chest CT was performed showing increase of peribronchial consolidations, GGOs and traction bronchiectasias in upper lobes (A-B: black arrows) with mild improvement of basal consolidation and GGOs (C-D: short arrow), representing a “pneumonitis flare”. The patient persisted clinical worsening despite prednisone treatment and eventually died.