Approximately one third of all brain tumors are glial in origin,
the astrocytoma being the most common type and the glioblastoma (GBM) the most common subtype.
Globally they are the most frequent primary brain malignancies as they represent up to 70% of these tumors and their prognostic will strongly depend upon the WHO classification,
grading them from I to IV depending on their malignancy (IV being the most malignant).
More recently new advances in the molecular and genomic field of brain tumors have proven to be determinant as prognostic factors,
so mutations in IDH,
p53,
TERT,
ATRX loss,
methylation of MGMT and 1p19q co-deletion could outweigh the tumor’s grading itself (1) (2).
Tumor grading is not only important for prognosis,
but it also plays a major role in new guidelines to decide therapy.
Surgical resection with broad surgical margins is the first step in the treatment of gliomas and depending on the tumor grading and the molecular characteristics of the tumor,
the scheme and timing of chemo/radiotherapy may vary (3).
Magnetic resonance (MR) is the gold standard study for pre-operative and follow-up imaging in brain tumors.
It plays a fundamental role in the initial differential diagnosis,
the anatomic location of the tumor and its relation with adjacent structures.
It also serves as a guide for biopsy aiming for the component with the highest malignancy probability in heterogeneous tumors and for surgical planning (4).
Early follow-up (24-48 hours post-op) is recommended as it can differentiate residual tumor from reactive surgical changes/enhancement (5).
Subsequent follow-up MR studies should evaluate response to treatment based on response assessment criteria (the RANO criteria is the most commonly used) and should aim to identify the presence of progression,
pseudoprogression and pseudoresponse.
In the last decade great advances have been made in neuroradiology with the detection of early/subtle signs of high-grade malignancy.
The development and upgrade of advanced MR sequences currently form part of the brain tumor protocol with sequences such as diffusion weighted imaging (DWI) and the ADC map,
susceptibility weighted imaging (SWI),
spectroscopy and dynamic perfusion imaging (DSC,
DCE).
These sequences address the key aspects of tumor grading such as tumor cellularity and the presence of tumoral necrosis,
hemorrhage and neoangiogenesis.
Susceptibility weighted imaging (SWI) has shown a great potential as it is a cost-effective sequence that complements structural imaging allowing better visualization of blood,
calcium,
iron deposition and microvessels without contrast agents (6)(7).
SWI sequence is generated from gradient-echo (GRE) pulse sequences,
typically acquired in 3D mode,
with flow compensation in all three directions to prevent artifacts and parallel imaging to reduce acquisition time.
A key feature of SWI is that magnitude and phase information are independently processed/displayed as well as combined for diagnostic purposes (8).
A broad spectrum of pathologies will show different patterns in SWI sequence (9).
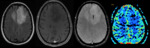
In the study of brain tumors,
SWI findings present as intratumoral signal susceptibility (ITSS) adopting the morphology of hypointense/signal loss areas with dot-like,
lineal,
mixed and gross/chunk-like structures related to the presence of deoxyhemoglobin in venous structures,
intratumoral blood products (hemosiderin and ferritin) and calcium.
The presence of microbleeds will typically present as dot-like hypointense/signal loss areas whereas microvessels will tend to present as hypointense lineal structures than can be followed in consecutive images; usually they both coexist in the same lesion as mixed structures.
Gross areas of hemorrhage will adopt a chunk-like hypointense /signal loss covering big parts of the tumor.
Overall these findings will represent two main characteristics of high-grade tumors: intratumoral hemorrhage and neoangiogenesis.
There are publications that correlate the presence of ITSS with dynamic MR perfusion imaging.
Xiaoguang Li et al.
correlated dynamic contrast-enhancement (DCE) with the degree of ITSS concluding that there was a moderate correlation between Ktrans and ITSS in the same tumor segments (10).
Park et al.
also correlated dynamic susceptibility contrast (DSC) MR perfusion with ITSS concluding that there is a significant correlation between the degree of ITSS with maximum relative cerebral blood volume (rCBVmax) in the same tumor segments,
although densely prominent ITSS did not correspond with the rCBVmax,
supposedly because ITSS did not only show increased vascularity but also areas of hemorrhage (11).
Pinker et al.
also demonstrated a correlation between areas of ITSS post-contrast administration with areas of high blood flow in positron emission tomography (PET-CT) studies and showed a statistically significant correlation between conglomerates of vascular proliferation on histopathology corresponding with areas of high ITSS (12).
Calcium is a diamagnetic compound,
and if present,
it can adopt any type of morphology in SWI (fig 1).
Identifying areas of calcification is important as it may narrow our differential diagnosis (calcified supratentorial intra-axial lesions) and can be easily differentiated from paramagnetic structures (blood products,
microvessels) in the filtered phase images,
as it will have an opposite signal intensity to paramagnetic structures; hyperintense in “right-handed” scanners (GE,
Phillips) or hypointense in “left-handed” scanners (Siemens,
Canon) (13).
In case of doubt,
comparing the signal intensity of the structure suspected to have calcium in phase images to the signal intensity of a prominent vein (i.e.
the torcula) is useful,
as they should have an opposite signal intensity.
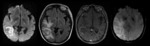
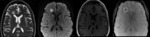
SWI can also be useful in the differential diagnosis between necrotic glioblastomas and pyogenic brain abscesses,
as the evaluation of DWI sometimes is not conclusive depending on the characteristics of the lesion.
A publication by Toh CH et al.
concluded that SWI images processed in minimum intensity projection (mIP) might be helpful in differentiating these two lesions based on the morphologic differences of their margins (14).
A dual rim sign (peripheral fine hypointense rim with an inner hyperintense rim) is the most specific feature distinguishing both lesions and is typically seen in pyogenic abscesses.
We also present two cases of non-pyogenic abscesses that exhibit this sign (fig.
2) (fig.
3).
Other SWI findings in the context of brain tumors are related to the treatment itself,
such as the presence of blood products due to surgical changes and radiation-induced cavernous malformations,
which are most likely to be found in low grade tumors as they have long latency periods after the radiation exposure (15).