The role of diagnostic imaging
- to differentiate a group of patients with extensive carcinomatosis or with unfavorable lesion sites,
who are not amenable to CRS from patients,
in whose case surgical procedures are feasible,
- to guide sampling of suspicious sites
- to assess the response to chemotherapy
- to detect a relapse [5,
11]
Imaging modalities
Ultrasound
- an auxiliary modality
- poor sensitivity in the visualization of peritoneal,
nodal and parenchymal metastases (69%,
32%,
and 57%,
respectively) [4]
- a guiding modality for biopsies [4,
6,
8]
Computed tomography
- a modality of choice for primary staging and follow-up → widely available,
high spatial resolution,
speed,
cost-effectiveness,
multiplanar reformations
- an overall sensitivity of 85-93%; for lesions smaller than 10 mm – 7-28%[4]
- metastases located in the root of the mesentery,
lesser omentum,
left hemidiaphragm and in the small bowel serosa often escape detection
- positive oral contrast media increase the conspicuity of serosal deposits (especially cystic) at the cost of the ability to detect calcified metastases,
[2]
- the detection of the peritoneal lesions further compromised by the artifacts from bowel motion,
suboptimal distention of the lumen of the digestive tract and of the urinary bladder
- the sensitivity of contrast-enhanced CT in detecting a relapse is inferior compared to the primary staging ( 43-89%) → distortion of normal anatomy following surgery,
small implants [5]
MRI
- a second choice modality → high cost,
long duration
- the sensitivity of contrast-enhanced MRI in visualizing peritoneal metastases at least equal to CT,
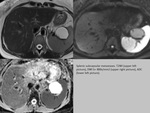
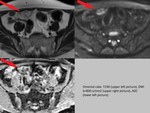
- DWI MRI is used for lesion characterization,
staging and assessment of response to therapy; may detect metastases even as small as 5 mm (Fig.
1)
- false-positive findings on DW-MR might be caused by restricted diffusion in normal spleen and lymph nodes,
or by hyperintensity of the normal bowel wall and ovaries
- false-negative findings may be due to less restricted diffusion in cystic,
necrotic and mucinous lesions (Fig.
2)
- abnormally enhancing peritoneum after 5 min following the administration of gadolinium contrast agent on MRI aids detection of peritoneal involvement [18]
Nuclear medicine
1.
Radioimmunoscintigraphy – recently less popular.
[5]
2.
F18-FDG PET/CT
• peritoneal implants → localized foci of increased F18-FDG uptake or a diffuse increase of tracer concentration in bowel serosa and peritoneum
• the sensitivity of 58-100%
• false-negative results – small implants,
low tracer uptake by mucinous tumors,
no uptake in small pulmonary metastases
• detects metastases in unenlarged lymph nodes [5]
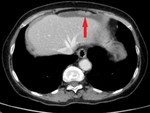
Findings indicating peritoneal carcinomatosis:
1.
Ascites. [3] (Fig.
3)
2.
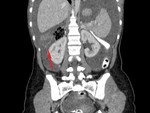
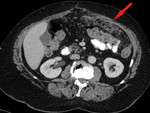
Invasion of the omentum – increased attenuation and small nodules in the omental fat,
later a solid tumor separating the bowels from the anterior abdominal wall – a classical finding of the "omental cake".
[3] (Fig.
4,
5,
6)
3.
Involvement of the mesentery – an increased density of the mesenteric fat and mesenteric nodules,
or a solid mass. [3]
4.
Implants in the peritoneal layers – nodular or diffuse thickening of the peritoneum with contrast-enhancement,
or the fixation of the small intestine to the anterior abdominal wall.
(Fig.
7) [3,
5]
Where to look for peritoneal metastases?
1.
Around the liver – in the ligamentum teres,
ligamentum venosum,
falciform ligament and the liver hilum.
2.
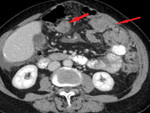
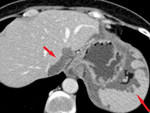
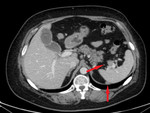
On the surface of the liver and spleen – subcapsular deposits may manifest as the scalloping of the surface of spleen or liver.
It is vital to properly differentiate the superficial subcapsular implants (stage III) from the parenchymal metastases (stage IV) in patients with ovarian cancer.
(Fig.
8,
9)
3.
In the Morrison's pouch and in the gallbladder fossa
4.
In the gastrohepatic ligament – which is a pathway of spread to the periportal space and liver.
5.
In the hepatoduodenal ligament,
around the portal vein,
the common bile duct and the hepatic artery
6.
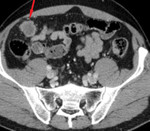
In the transverse and the sigmoid mesocolon
7.
In the paracolic gutters (Fig.
10)
8.
In the small bowel mesentery
9.
In the greater omentum
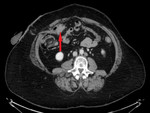
10.
In the lesser omentum,
in the epiploic formanen and in the omental bursa (Fig.
11)
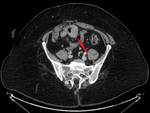
11. In the bowel serosa (Fig.
12,
13)
12. In the pelvic peritoneal recesses [2]
Contraindications against surgery that should be covered by a radiology report include:
• high PCI-score (PCI above 20 in ovarian and colorectal cancer and above 10 in gastric cancer),
• extra-abdominal metastases,
• infiltration of the hepatoduodenal ligament,
• extensive invasion of the mesentery,
• bladder trigone involvement,
• massive involvement of small intestine
• diaphragmatic infiltration,
• involvement of both stomach and colon (ovarian and colorectal carcinomatosis),
• liver parenchymal metastases – except for selected cases of colorectal cancer [10].
Critical findings that need to be reported in cases of ovarian lesions:
• pelvic sidewall involvement – the primary tumor is located closer than 3 mm from the pelvic sidewall or encompasses more than 90% of the external iliac vessels circumference
• involvement of the greater omentum – the involvement of the transverse colon alters the surgical approach
• the involvement of periaortic lymph nodes above the level of renal vessels – an indication for neoadjuvant chemotherapy
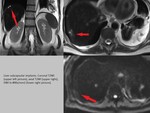
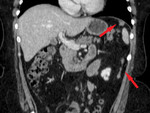
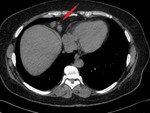
• enlarged (exceeding 5mm in the short axis) cardiophrenic lymph nodes – a negative prognostic factor (Fig.
14)
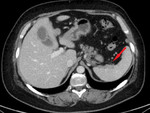
• subcapsular liver lesions extending from the Morrison's pouch to the inferior vena cava at the level of the right hepatic vein – a high risk of extensive bleeding during surgery
• splenic subcapsular and intraparenchymal lesions – may influence the surgeon's decision on performing a splenectomy (Fig.
15,
16)
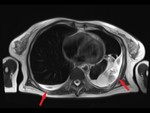
• enhancement of the diaphragmatic surface adjacent to the liver on MRI – an early indicator of diaphragmatic involvement,
• involvement of the pleural cavity – (unless cytologically proven,
does not preclude surgery) (Fig.
17)
• involvement of the lesser omentum,
the hepatoduodenal and the gastrohepatic ligaments – CRS more technically demanding;
• distention of lesser sac and the splenorenal,
gastrosplenic and splenopancreatic ligament infiltration – may preclude the surgery
• involvement of the gallbladder fossa,
the falciform ligament and the periportal space [18]
CC-score - Table 1
CC (Completeness of cytoreduction score) is estimated by the surgeon following debulking surgery and refers to visually determined removal of all visible peritoneal deposits.
[6]
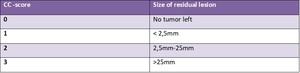
Table 1: CC - score - created basing on Jacquet P, Sugarbaker PH. "Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis."
PCI (Peritoneal Cancer Index) - Table 2
• a well-established scoring system to determine the extent of the malignancy in the peritoneum
• initially calculated based on diagnostic imaging
• finally established following surgery
• divides the abdomen into nine segments – by two transverse planes: the upper,
connecting lowest points of both costal arches,
and the lower,
connecting both anterior superior iliac spines,
and by two sagittal planes that divide the abdomen into thirds [Fig.] (Fig.
18)
• the small intestine is subdivided into four segments [6,
13]
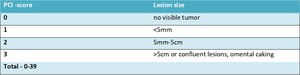
Table 2: PCI score - created basing on Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis.
Simplified Peritoneal Cancer Index (SPCI) - Table 3
• developed to assess tumor burden of carcinomatosis of colorectal and appendiceal origin
• the peritoneal cavity is divided into seven regions: pelvis,
right lower abdomen,
small bowel and its mesentery,
omentum and the transverse colon,
right subphrenic space,
left subphrenic space and subhepatic space-stomach.
[12]
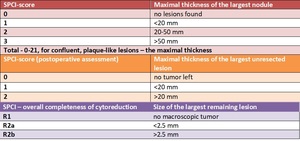
Table 3: SPCI-score- created basing on Verwaal VJ, van Tinteren H, van Ruth S, et al. "Predicting the survival of patients with peritoneal carcinomatosis of colorectal origin treated by aggressive cytoreduction and hyperthermic intraperitoneal chemotherapy."
Fagotti Score - Table 4
• based on a laparoscopic exploration of the peritoneal cavity
• used to describe surgically critical carcinomatosis implants of ovarian origin
• important for a radiologist – some centers decide on eligibility for CRS on the bases of integrated data from diagnostic imaging studies and from diagnostic laparoscopy
• gives an idea of the areas to cover when reporting a CT [15]
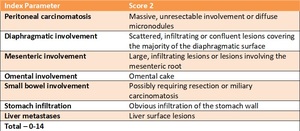
Table 4: Fagotti score - created basing on Fagotti A, Ferrandina G, Fanfani F, et al. "Prospective validation of a laparoscopic predictive model for optimal cytoreduction in advanced ovarian carcinoma."
Modified Fagotti score - Table 5
• a laparoscopic-based score,
used for ovarian cancer
[14]
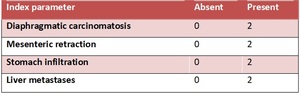
Table 5: Modified Fagotti score - created basing on Brun JL, Rouzier R, Uzan S, Daraï E. "External validation of a laparoscopic-based score to evaluate resectability of advanced ovarian cancers: clues for a simplified score."
Gilly's peritoneal carcinomatosis staging - Table 6
Five stages of peritoneal carcinomatosis of colorectal origin [16]:
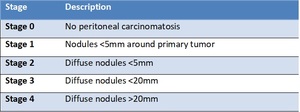
Table 6: Gilly's peritoneal carcinomatosis staging - created basing on Gilly FN, Carry PY, Sayag AC, et al. "Regional chemotherapy (with mitomycin C) and intra-operative hyperthermia for digestive cancers with peritoneal carcinomatosis."
PAUSE – a standardized way to report key findings -Table 7
PAUSE is an acronym proposed by Chandramohan et all.
to standardize radiology reporting of peritoneal metastases to provide proper radiological staging.
[17]
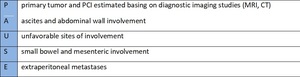
Table 7: PAUSE - adapted from Chandramohan A1, Thrower A2, Smith SA2, Shah N2, Moran B3."PAUSE": a method for communicating the radiological extent of peritoneal malignancy."
The U feature is divided into three categories: [17] - Table 8

Table 8: PAUSE - adapted from Chandramohan A1, Thrower A2, Smith SA2, Shah N2, Moran B3."PAUSE": a method for communicating the radiological extent of peritoneal malignancy."

![Fig. 18: PCI-score – created basing on Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. [13]](https://epos.myesr.org/posterimage/esr/ecr2019/148448/media/815226?maxheight=150&maxwidth=150)
















