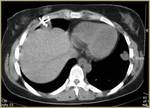
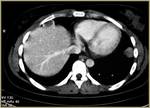
All human studies have been approved by the hospital’s ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
We retrospectively reviewed 20 cases regarding pediatric patients,
who underwent RFA in our department between September 2010 and January 2018. Relative pre- procedural management included laboratory tests (Hemoglobin concentration,
international normalized ratio [INR],
partial thromboplastin time and platelet count),
parents’ consent and fasting 12hours prior to the procedure .
Exclusion criteria were only coagulopathy,
INR >1.5 or a platelet count of less than 60,000/mm3.
Despite the fact that according to literature general anesthesia is preferred over other methods of anesthesia,
the ability of our patients to cooperate and tolerate well the procedure led to our decision to use conscious sedation (administration of midazolam [0.05 mg/kg,
maximum dose 70 0.15 mg/kg] and fentanyl [1 mcg/kg,
maximum dose 3–4 mcg/ 71 kg every 20 min]),
performed by an anesthesiologist.
Regarding this vulnerable patient group the choice of conscious sedation oriented to minimizing any potential risk induced by general anesthesia.
All radiofrequency ablation procedures were performed by trained staff,
experienced in CT-guided percutaneous interventions.
Imaging modality of choice for the percutaneous electrode guidance was spiral CT (Somatom Emotio Duo System; Siemens,
Erlangen,
Germany).
Despite the fact that CT scan protocols may vary for different patient age or size,
we applied neonatal low-dose scan protocols (80 kV,
33 mAs,
slice width 5 mm,
feed/rotation 16,
kernel B40s,
CT dose index 0.9) with the minimum number of scans (n=9–14) in the total of our patients,
in order to avoid the intake of any unecessary radiation in our young patients.
Thus,
we achieved minimizing radiation exposure of our patients with an adequate quality of imaging.
Access sites were defined by the interventional radiologist and different access routes have been chosen to approach the lesion depending on its location.
The shorter route through lung parenchyma was chosen regarding lung lesions,
with the perspective to minimize the potential risk of a serious complication,
such as pneumothorax. Lesions adjacent to the mediastinum and great vessels were not ablated.
Furthermore unproper management during RFA of hepatic lesions may give rise to complications related to the gallbladder,
hilar vessels and large bile ducts.
Therefore,
a thorough evaluation of each patient’s anatomy and potential variations was essential to avoid procedure related complications.
Finally in bone lesions rigorous attention was given to avoid the great vasculature.
Applicator’s and electrode type’s choice was based on tumor size and location.
( RITA Model 1500® electrosurgical generator and a seven-array,
2- to 3-cm multitined electrode -13-G diameter 15-cm length- and MIRAS generator with a single and a triple spiral-shape electrode to cover larger lesions up to 5 cm).
Pulse radiofrequency ablation energy was applied for 7–11 min for each lesion (median 8 min), depending on size,
location and vascularity of the lesion,
at 90–110 W.
The maximum tissue temperature ranged from 90 to 110°C. The ablation was designed to induce coagulation necrosis of the surrounding tissue in a radial of 1 cm beyond its imaging borders,
as well as necrosis of selected tract during electrode retrieval.
The latter technique orients to securing from tumor seeding along the electrode route [4,5]. As a result,
the possibility of disease dissemination has almost been reset.
Technical success is implied by the total eradication of the ablated tumor according to protocol directly post-procedurally.
Therefore,
to assess technical success we performed a dual-phase low-dose contrast-enhanced spiral CT immediately post RFA application with hepatic arterial and portal venous phases.
Absence of enhancement regarding ablated lesions and a surrounding hypervascular halo were indications of completely ablated lesions.
effectiveness was evaluated after a 3 months follow up period and was implied by complete necrosis of the lesion in post-ablation CT scans or other imaging modalities[3,6] .
A 24-hour hospitalization established on monitoring grounds was given to all of our patients,
who were discharged the day after if no complication occurred.
Post-ablation complications are classified as major (pneumothorax,
massive hemorrhage,
pulmonary abscess after lung radiofrequency ablation,
hematoma or liver abscess after liver ablation) or minor (pain,
postablation syndrome,
skin burns)[7-14].
Follow-up regarding pediatric patients may include multimodality imaging (CT U/S or MRI) as well as thorough clinical examination.
We assessed our patients after 1 month,
3 months and 6 months post RFA sessions and every 6 months thereafter.
Necrotized lesions appeared hypoechoic in US,
whereas after the administration of US contrast media no enhancement was present.
Concerning MRI follow- up total necrosis is defined as lesion’s high signal on T2-W images,
low signal in T1-W images and no enhancement after administration of gadolinium (Gd).