1.
CLASSIFICATION
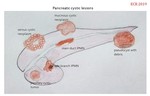
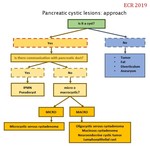
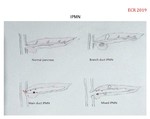
Pancreatic cystic neoplasms can be classified either according to histological type or to morphological appearance.
(Fig.
1)
By histology:
- FREQUENT ETIOLOGIES.
- Benign lesions may have either non-tumor / inflammatory-postraumatic origin (pseudocyst) or neoplastic (SCN).
- Among premalignant lesions we have IPMN intraductal papillary mucinous neoplasm (IPMN) and mucinous cystic neoplasm (MCN).
- Categorized as malignant tumors are those with cystic degeneration,
being adenocarcinoma the most frequent.
- UNCOMMON ETIOLOGIES.
· Epithelial pseudopapillary solid neoplasm (PSN),
with both solid and cystic component.
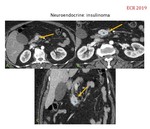
· Others infrequent malignant tumors with cystic degeneration (neuroendocrine tumors like insulinoma - Fig.
25- and some metastases from renal cell carcinoma,
melanoma,
lung,
breast,
hepatocellular or ovarian tumors).
By morphological appearance:
According to the radiological characteristics,
four types of cystic lesions can be classified:
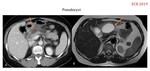
UNILOCULAR (SIMPLE CYSTS):
Normally they are acquired lesions with inflammatory or infectious origin.
Pseudocyst is the most common cystic lesion.
This lesion occurs in the setting of acute or chronic pancreatitis and less frequently after trauma or surgery.
Pseudocysts are round or oval collection,
with a simple thin wall,
without septa or solid nodules.
(Fig.
2).
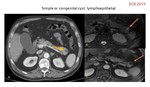
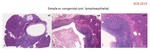
This section incorporates other acquired cysts,
such as retention or exudatives (also related to acute pancreatitis),
lymphoepithelial (Fig.
3-4),
or those secondary to bacterial or parasitic infections (echinococcus multilocularis,
amebiasis).
Rare congenital etiologies include solitary true cyst.
Besides,
pancreatic cystic lesions may also be found in some systemic diseases with cysts in other locations (predominantly in the liver),
such as polycystic kidney disease,
Von-Hippel-Lindau or cystic fibrosis.
MICROCYSTIC:
Microcystic lesions are composed of multiple small cysts surrounded by thick fibrous stroma,
jointly delimiting a larger lesion with lobulated edges,
and very often a central scar image.
This is the characteristic appearance of SCN.
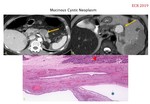
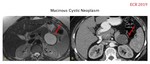
MACROCYSTIC:
As well as microcystic,
macrocystic lesions are formed by multiple little cysts.
The difference is that macrocystic consists of a smaller number of cysts,
but larger (usually > 2 cm).
Most frequent lesions in this group are MCN,
side-branch IPMN (frequently located in uncinate process or body-tail) or oligocystic serous cystadenomas.
In MCN,
a multilocular cystic pattern with several cysts (less than 6) of medium size is characteristic.
The main difference between both will be the communication or lack thereof with ductal pancreatic system.
CYSTIC & SOLID COMPONENT:
Those lesions with a visible solid nodule that generally enhances after administration of intravenous contrast are included in this category.
We must be alert,
as they may become malignant,
or intermediate with high potential for malignancy.
· Neoplasm with cystic degeneration.
It is the case of PSN,
infrequently diagnosed,
and rare entities like islet cell tumor or cystic neuroendocrine tumors.
More often pancreatic adenocarcinoma and metastasis are confirmed.
· True cystic neoplasms.
Some MCN and some IPMN will also have this appearance,
being these the most malignancy risky.
2.
DIFFERENTIAL DIAGNOSIS
As referred,
largely due to the increase in diagnostic sensibility,
the majority of pancreatic cystic lesions are diagnosed casually in asymptomatic patients (found in approximately 2.3% of CT studies).
Normally,
these are benign or low malignancy lesions.
Approach (Fig.
5, Table 1)
First,
we must always determine if it is a non-tumoral lesion or a cystic neoplasm.
- A history of pancreatitis,
alcohol abuse or stone disease or previous abdominal trauma,
added to particular imaging findings (unilocular cyst without or with debris inside),
will guide towards the diagnosis of pseudocyst.
- With no history of the described background,
we should think about a cystic neoplasm.
- If the lesion has unilocular macrocystic appearance and the patient is female,
it may be a MCN.
Sometimes calcification of the wall can be identified.
- In microcystic appearance lesions,
it will be essential to determine connection with ductal system.
- If serous content and images of septa and central scar (without or with calcifications),
we would suspect SCN (the only pancreatic cystic neoplasm without malignant potential).
- If no scar or calcifications are observed,
and/or there is evidence of connection with pancreatic duct (highly specific,
more visible in MRCP or T2),
we would consider a branch-duct IPMN.
(In very small lesions it is sometimes not possible to specify a diagnosis,
complementary methods will be necessary).
MR vs CT
MR is the most sensitive technique for detection of cystic component.
As previously mentioned,
heavily T2-weighted or cholangiographic MR will have greater sensitivity detecting ductal connection (IPMN),
as well as presenting great ability to characterize the internal structure.
On the other hand,
CT will be better at detecting central calcifications in SCN or peripheral murals in MCN.
3.
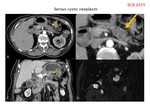
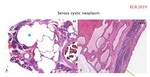
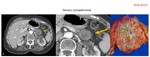
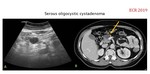
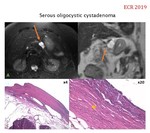
SEROUS CYSTADENOMA (Fig.
6-8).
Epidemiological data: incidence of approximately 10-20 % of cystic pancreatic neoplasms,
typically seen in > 60 year women (also called "grandmother's tumor".
Clinical expression: usually asymptomatic.
If larger (>4cm) they may cause symptoms of abdominal pain or rarely jaundice from mass-effect and duct obstruction.
Location: anywhere,
although slightly higher accuranceincidence in head and neck.
Imaging: cystic lesion of variable size (average 5 cm),
lobulated morphology,
formed by multiple small cysts <2 cm (“honeycomb” or “brunch of grapes”) which contain proteinaceous fluid and separate from each other by connective tissue septa.
The existence of a central stellate scar that may associate amorphous "sunburst" calcifications (18%) is very characteristic.
Pancreatic duct may be displaced,
or less frequently encased or obstructed.
There will be oligo-cystic forms.
(Fig.
9-11).
- US: predominantly solid mass where hypoechoic and echoic areas coexist.
- CT: hypodense,
enhancement of septa after contrast administration.
- Angio: hypervascular with visible alimentary arteries and prominent drainage veins,
with or without neovascularization and arterio-venous shunts; no vascular encasement is visible.
- MR: cluster of small hyperintense cyst on T2; delayed enhancement of thin fibrous septa and central scar.
Prognosis: no potential for malignancy (according to symptoms,
it will be monitored or resected).
4.
MUCINOUS CYSTIC NEOPLASM (Fig.
12-13).
Epidemiological data: 10-40% of pancreatic cystic tumors,
mainly in women,
especially middle-aged (“the mother’s tumor”).
Clinical expression: asymptomatic,
if symptomatic usually abdominal pain or anorexia.
Location: 90% in the tail and body of pancreas (95%).
Imaging: well-defined thick-walled encapsulated mass,
variable size (described up to 2-36 cm),
rounded or lobed edges,
uni or multilocular,
conformed by cystic spaces (>2 cm) separated by thin septa (<2mm) with mucin content.
Size > 4 cm,
mural nodules,
thick wall,
calcifications,
papillary excrescences,
septations are features of malignancy (mucinous cystadenocarcinoma).
Metastases (uncommon) are seen as rounded thick-walled cystic lesions.
- US: echoes may be contained into cysts.
- CT: cystic spaces may have different levels of attenuation.
After contrast administration,
enhancement of septa pattern and wall.
- MR: hypointense in T1,
hyperintense in T2,
enhancement of thick-fibrous wall in late adquisitions.
- Angio: hipo-avascular mass which displaces adjacent vascular structures,
although small areas of neovascularization and vascular blush may be observed.
Prognosis: depends on whether or not has progressed to cystadenocarcinoma at the time of diagnosis; 5-year survival rate of 90% in complete surgical resections.
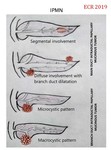
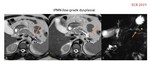
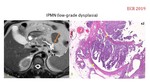
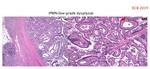
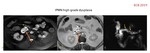
5.
IPMN (Fig.
14-16).
(Fig.
17-23).
Epidemiological data: Average age of onset of 65 years,
slightly more frequent in men.
Clinical expression: usually asymptomatic,
when clinical: recurrent episodes of abdominal pain and / or acute pancreatitis secondary to the interruption of the flow of pancreatic secretions.
Location: frequently multifocal,
up to 10% affect the entire gland.
If branch-duct IPNM,
in order of frequency: uncinate,
tail and body.
Imaging: Intraductal tumor originating in the papillary lining epithelium,
mucin secretor.
Cystic lesions with macro or micro-cystic pattern and a thin fibrous capsule,
surrounded by normal pancreatic parenchyma is the most common appareance.
100% specificity if high viscosity intracystic fluid is found.
Findings suggestive of malignancy are the presence of mural thickening,
irregular septations or solid nodules.
As mentioned previously,
there may be partial or total atrophy of the gland,
and also protrusion of the papilla towards the duodenum (fish mouth papilla, Fig.
21).
- US: hyperechogenic
- CT: hyperdense
- MR: filling defect within a dilated duct,
or appearance of a papillary mural nodule that enhances after contrast administration.
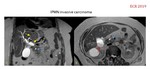
MAIN DUCT IPMN
Depending on its position and size,
whole or segmental dilation of main duct can be associated.
The malignant forms are found in approximately 70% of the cases that affect the main conduct and in approximately 25% of the cases that affect only the lateral branches.
- Diffuse dilation: proximal tumors,
secondarily the parenchyma of the rest of the gland may atrophy.
Also,
obstruction of the common bile duct (either by tumor or by impacted mucin) can be seen,
as well as dilation of adjacent ductal branches.
Although very infrequent,
biliary or duodenal pancreatic fistulas could develop,
or even a peritoneal pseudomyxoma.
- Segmental dilation of main duct: from the location of the lesion,
usually associating dilation of adjacent ducts and without the atrophy of the entire gland.
Normally,
diagnosis will be confirmed with the performance of an ERCP (identifying ampullary bulge,
secretion of mucin or communication with pancreatic duct).
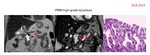
BRANCH DUCT IPMN
Normally,
the small intraductal solid lesion cannot be visualized,
suspecting the diagnosis when identifying a cluster of multiple dilated secondary branches.
The main duct does not usually dilate until the tumor acquires a moderate size.
6.
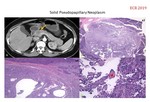
SOLID PSEUDOPAPILLARY TUMOR (Fig.
24)
Epidemiological data: prevalence of 0.17 to 2.7% of all non-endocrine pancreatic tumors,
observed at any age,
more frequent in female and in black and Asian race.
Clinical expression: usually asymptomatic,
symptoms usually referred as poorly defined upper abdominal discomfort,
palpable mass or jaundice (infrequent).
Location: >1/3 tail,
1/3 head; some are exophytic with undetermined dependence.
Most typical appearance is a large solitary mass (6-10 cm most frequent),
encapsulated with round or lobed contours,
heterogeneous with necrotic-hemorrhagic component and cystic degeneration.
When small tumors,
they will predominantly be solid.
They can also be completely cystic,
or have debris inside.
Stippled or amorphous dystrophic calcifications in capsule,
or chunky inside tumor,
may be observed.
We can find compression of adjacent structures without invasion.
It is a hypovascular lesion,
with no enhancement of contrast,
or very slight in solid tissue towards the center of the mass.
- US: echogenic mass with necrotic center surrounded by an echogenic ring (capsule),
or completely cystic with a subcapsular ring of solid tumor.
-CT: hypoattenuating capsule,
cystic components of low attenuation,
isoattenuating solid component.
- MR:
- fibrous capsule: hypointense in T1 and T2.
- solid component: slightly hyperintense in T2 and iso-hypointense in T1.
Heterogeneous peripheral ring enhancement with progressive filling of the lesion.
Hypointense in late acquisitions.
- High signal foci in T1 in a high percentage of cases,
in relation to internal haemorrhage (hallmark).
- Angio: hypo (mild blush) or avascular,
with displacement of vessels.
Prognosis: low-grade and slow-growing malignant tumor with cure ratio of 95% in complete surgical resection; being difficulties in diagnosis the main prognosis factor (PSP is sometimes poorly classified).
In very rare cases,
omental,
liver or locoregional node metastases may be identified.