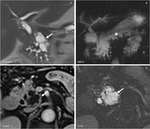
We revised cases of PCL explored in our department in the last 5 years with tomography (CT) and/or magnetic resonance imaging (MRI).
We correlated the imaging findings with the histopathological report.
Our focus was mainly on potentially malignant and malignant lesions.
Imaging techniques
- multi-slice,
depicts most pancreatic lesions
- native and post-i.v.
contrast multi-phase acquisition
- best method to assess vascular involvement
- MRI with cholangiopancreatography (MRCP):
- detection and staging of pancreatic lesions,
more diagnostic than CT
- evaluation of the pancreatic duct and bile ducts
- superior characterization of the cystic nature of a pancreatic lesion
The imaging CT and MRI protocols that we performed on our patient consisted in:
CT examination protocol:
- native phase
- three post i.v contrast injection phases (300 or 370 mg I/ml; 100-150 ml - depending on the patient's weight) using a power injector at a rate of 2.5-3 ml/sec;
- arterial phase starting at 20-30 seconds after the onset of the injection,
with cranio-caudal movement (from the diaphragm muscles to the iliac bones);
- portal phase starting at 70 seconds post i.v.
contrast with cranio-caudal scan as well.
- late phase starts at 160 seconds post i.v.
contrast with cranio-caudal scan as well.
- after the examination images were processed,
by multiplanar reconstruction (MPR),
maximum intensity projection reconstruction (MIP) or volumetric rendering technique (VRT).
MRI examination protocol
In case of an MRI examination,
we performed on 1.5-T MR scanner,
using torso phase array coil (TORSOPA) and administration of i.v.
gadolinium contrast agent,
Gd-BOPTA (0.1ml/kgcorp) using a power injector at a rate of 2.5-3 ml/sec.
The MRI protocol included:
- breath-hold sequences,
axial T2 fast-spin echo (FSE) with fat saturation,
- T1 in-phase/out-of-phase,
- single shot T2 FSE with long time of echo (TE) and short TE
- diffusion weighted imaging with ADC mapping
- coronal FSE single-shot heavily T2 weighted MR cholangiography with fat saturation (3DMRCP),
with the possibility of mapping the biliary tree and the Wirsung duct
- three phase 3D-T1-weighted with fat saturation after dynamic i.v Gd-BOPTA injection: arterial phase (after 20-30 sec),
portal phase (after 70 sec),
equilibrium/ late phase (180 seconds post-administration).
The differential diagnosis between a benign and a malignant PCL was based on imaging criteria: walls aspect,
attenuation coefficient/ signal intensity,
presence of thin/thick septa and calcifications,
restriction of diffusion,
enhancing pattern.
Serous cystadenoma:
| CT features (Fig. 2) |
MRI features (Fig. 3) |
- cystic areas with fluid densities and honeycomb-like setting;
- contrast enhancement of the peripheral capsule and internal septa;
- a characteristic central fibrous scar with associated calcification in 33% of the cases.
[14,15]
|
- multicystic,
lobulated mass with individual cysts of 1-20 mm - „Bunch of grapes” ;
- cystic lesions with fluid signal,
T1 low signal,
T2 high signal,
separated by small septa and limited by a well defined wall;
- intratumoral heterogenous signal determined by: calcifications or intracystic haemorrhage with T1 high signal in the subacute phase;
- on delayed contrast enhanced images,
central scar may enhance.
[14,15]
|
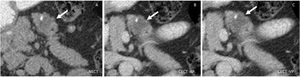
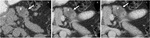
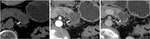
Fig. 2: Serous cystadenoma: Unenhanced CT (A), and CT after CM injection (B,C): well defined lobulated cystic mass in the body of the pancreas (arrow), with small peripheral calcification
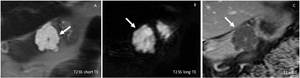
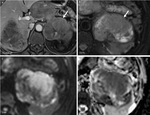
Fig. 3: Serous cystadenoma: MRI T2 single shot with long and short TE (A,B), T1 after CM injection: Multicystic polilobulated lesion (arrow) with central late enhancing scar.
Mucinous cystadenoma:
| CT features (Fig. 4) |
MRI features (Fig. 5) |
- near-water density cystic lesion with internal septations;
- hypovascular,
wall and septum contrast enhancing;
- calcifications may be seen;
- internal septa and small pancreatic ducts dilation may be present.
[14,15]
|
- uni/multilocular macrocystic mass;
- when homogenous,
it is low signal on T1 and high signal on T2;
- it can have heterogenous structure because of the secreted mucin,
haemorrhagic components and calcifications seen only when big enough.
[14,15]
|
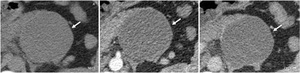
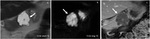
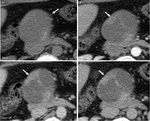
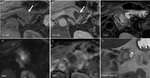
Fig. 4: Mucinous cystadenoma: Unenhanced CT (A), and CT after CM injection (B,C): large well defined uniloculate cystic mass in the tail of the pancreas (arrow), with non enhancing thin wall
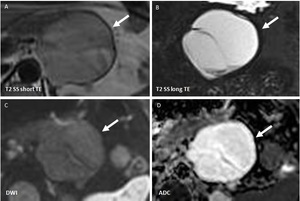
Fig. 5: Mucinous cyst adenoma: MRI T2 single shot with long and short TE (A,B), Diffusion weighted imaging (C,D): Large uniloculated lesion (arrow) with thin internal septa, showing slight drop of signal in T2 sequences and no restriction of diffusion
Mucinous cystadenocarcinoma: malignant variant of mucinous cystadenoma
| CT features (Fig. 6) |
MRI features |
- Larger cysts;
- contrast enhancing solid excrescences;
- cystic-like,
round hepatic metastases with regular thick wall,
if present.
[14,15]
|
- Gadolinum contrast enhanced images can emphasize septa and parietal excrescences;
- Mucin components show high T1 signal while calcified components are low signal in both T1 and T2.
[14,15]
|
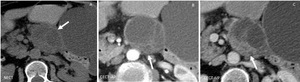
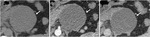
Fig. 6: Mucinous cystadenocarcinoma: Unenhanced CT (A), and CT after CM injection (B,C): Large heterogenous cystic lesion in the body of the pancreas (arrow) with internal septa and solid parietal enhancing excrescences
Intraductal Papillary Mucinous Neoplasm:
- Main Duct IPMN (MD-IPMN)
- Branch Duct IPMN (BD-IPMN
| CT features (Fig. 7,
Fig. 8) |
MRI features (Fig. 9,
Fig. 10) |
- dilated Wirsung duct,
segmental or entire;
- overlaying pancreatic parenchyma may be thinned;
- solid mural nodules may suggest malignant transformation.
[20,22]
|
- low signal T2,
intermediate signal T1,
gadolinium enhancing;
- polycystic mass with internal septa and parietal protruding papilla ;
- Wirsung dilation; pancreatic tissue atrophy; with/without MBD obstruction;
- Diffuse MD ectasy type- diffusely dilated Wirsung with no identifiable cause.
[20,22]
|
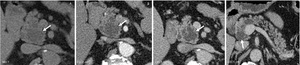
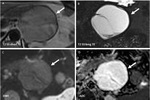
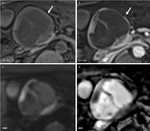
Fig. 7: Intraductal Papillary Mucinous Neoplasm (main duct) : Unenhanced CT (A), and CT after CM injection (B,C,D): multiloculated cystic lesion in the head of the pancreas (arrow) asociating pancreatic atrophy and wirsung duct dilation
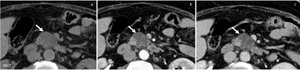
Fig. 8: Intraductal Papillary Mucinous Neoplasm (branch duct) : Unenhanced CT (A), and CT after CM injection (B,C): multiloculated cystic lesion in the uncinate process of the pancreas (arrow).
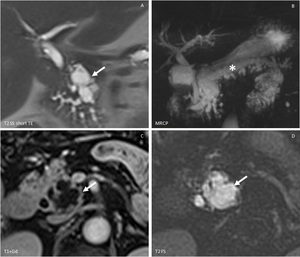
Fig. 9: Intraductal Papillary Mucinous Neoplasm (main duct): MRI with T2 single shot with long Te and FS (A,D), MRCP (B) and T1 with CM injection(C): polcicystic cephalo-pancreatic mass (arrow) with small parietal enhancing nodules associating entire Wirsung duct dilation (asterix) and also small pancreatic canclicules

Fig. 10: Intraductal Papillary Mucinous Neoplasm (branch duct): MRI with MRCP (A) T2 single shot with long TE and FS (B,C) and T1 with CM injection (D): polcicystic mass in the uncinate process of the pancreas (arrow) with small parietal enhancing nodules with visible communication with a side branch of the Wirsung duct dilation (arrow head).
Cystic pancreatic endocrine neoplasm:
| CT features (Fig. 11) |
MRI features (Fig. 12) |
- Cystic lesion that rarely obstructs the pancreatic duct;
- Smooth margins;
- Peripheral enhancement usually on both arterial and portal phases because of their rich blood supply.
[19]
|
- Superior fluid and soft-tissue contrast;
- Low signal in T1 weighted;
- High signal in T2 weighted.
[19]
|
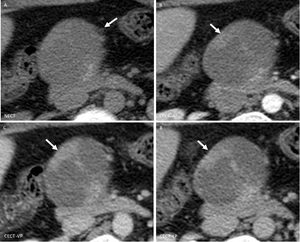
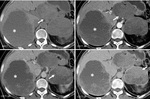
Fig. 11: Cystic Pancreatic Endocrine Neoplasm: Unenhanced CT (A), and CT after CM injection (B,C): large well defined cystic lesion in the neck of the pancreas (arrow) with irregular internal wall and fast persistent enhancement
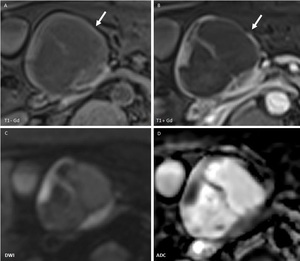
Fig. 12: Cystic Pancreatic Endocrine Neoplasm: MRI T1 before and after CM injection (A,B), Diffusion weighted imaging (C,D): large cystic lesion in the neck of the pancreas (arrow) without Wirsung duct dilation, with solid enhancing components and septa associating restriction of diffusion
Solid pseudopapillary neoplasm:
| CT features (Fig. 13) |
MRI features (Fig. 14) |
- Well encapsulated lesion with varying solid and cystic component;
- Enhancing solid areas are typically noted peripherally;
- Cystic spaces present usually more central.
[14,15]
- Well encapsulated lesion with varying solid and cystic component;Enhancing solid areas are typically noted peripherally;
- Cystic spaces present usually more central.
[14,15]
|
- Typically well defined lesion with low heterogeneous T1 signal;
- Heterogeneous T2 signal;
- Heterogeneous slow progressive gadolinium enhancement.
[14,15]
|
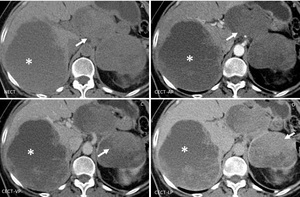
Fig. 13: Solid pseudopapillary neoplasm: Unenhanced CT (A), and CT after CM injection (B,C,D): large well defined heterogenous lesion in the body and tail of the pancreas (arrow) with progressive late enhancement of the peripheral component; large right liver lobe metastasis (asterix) with the same CT pattern
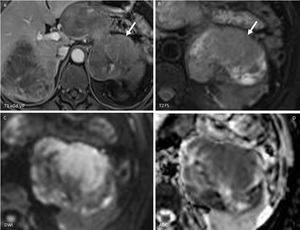
Fig. 14: Solid pseudopapillary neoplasm: MRI T1 after CM injection (A), T2 with fat saturation (B), Diffusion weighted imaging (C,D): central cystic area with peripheral late enhancing solid component (arrow) showing restriction of diffusion.
Cystic degeneration of pancreatic ductal adenocarcinoma:
| CT features (Fig. 15) |
MRI features (Fig. 16) |
- Hypo dense nodules with poor contrast enhancement,
associating pancreatic atrophy;
- Cystic irregular mass with interior septa after i.v.
contrast;
- Double duct sign may be seen.
[23]
|
- Hypo intense T1 diffuse lesion;
- Heterogeneous signal with hypo intense T2 inclusions (cystic component/ necrosis);
- Restriction of diffusion;
- MRCP shows double duct sign.
[23]
|
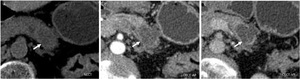
Fig. 15: Cystic degeneration of pancreatic ductal adenocarcinoma: Unenhanced CT (A), and CT after CM injection (B,C): hypo dense, hypo enhancing, ill-defined lesion in the body of the pancreas (arrow) with a small cystic component included
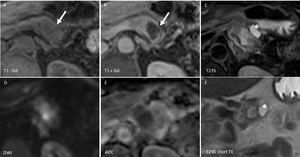
Fig. 16: Cystic degeneration of pancreatic ductal adenocarcinoma: MRI T1 before and after CM injection (A,B), Diffusion weighted imaging (D,E), T2 single shot with long Te and FS (C,F): hyper intense T2, hypo intense T1, hypo enhancing ill-defined lesion in the body of the pancreas (arrow) with restriction of diffusion and a small cystic lesion included (asterix)
In cases of depiction of pancreatic cystic masses the radiologist’s structured report should contain the following:
- Patient medical history (pancreatitis,
Von Hippel Lindau disease,
etc.) gender and age
- Number (single,
multiple)
- Localization (head,
neck,
body,
tail,
uncinated process)
- Shape (round,
oval)
- Dimensions (micro nodular,
macro nodular)
- Contour (regular,
irregular,
well defined,
ill defined)
- Wall (thin,
thick,
enhancing,
non-enhancing)
- Structure: homogenous/inhomogeneous (calcifications,
mucin content,
septa,
debris)
- Enhancing pattern
- Communication with the Wirsung duct
- Relationship with the adjacent structures
- Vascular or biliary invasion
- Associated pathologies (lymphadenopathies,
ascites,
liver metastases etc.)