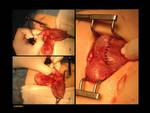
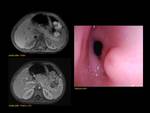
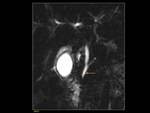
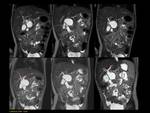
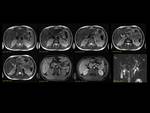
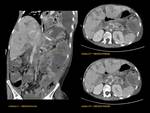
1)Normal pancreas and embriology Fig. 1
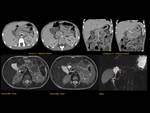
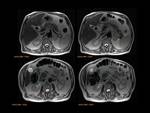
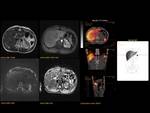
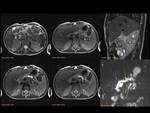
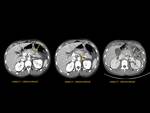
Pancreas grows exponentially the first year of life,
with a pancreatic head more prominent than the body and pancreatic duct (1,5-2mm) in children.
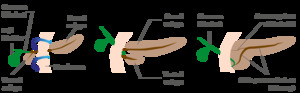
Fig. 1: Pancreas develops from two anlagens that arise from the endodermal lining of the duodenum. The dorsal anlagen develops at the 4th weeks of gestation from the dorsal side of the duodenum and the ventral anlagen develops at 5wg from the ventral side at the same level as the bile duct. As the GI rotates at the 7th week of gestation, so does the ventral anlagen, and the two pancreatic anlagens join into a single gland and their ductal systems fuse. The ventral anlage forms the uncinate process and posterior part of the pancreatic head, and the dorsal anlage forms the anterior pancreatic head, body and tail.
2)Imaging techniques
Sonography (US) is a primary screening tool to evaluate the paediatric pancreas,
due to its multiple advantages as absence of ionizing radiation,
the ability to image without sedation,
and real-time evaluation.
MRI in combination with MRCP is a powerful tool for imaging of the pancreatic duct,
and biliary system in the paediatric population.
Low-dose computed-tomography is a vital and accurate tool for rapid diagnostic evaluation of serious illness or injury in children,
A major disadvantage of CT is the inevitable radiation exposure.
3)Developmental Anomalies
Pancreatic developmental anomalies may result in anatomic variants that predispose to specific pancreatic or peripancreatic diseases.
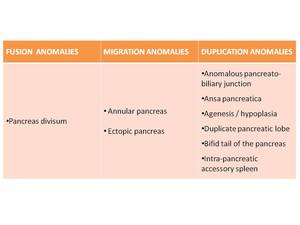
Fig. 2: Anatomic anomalies of the pancreas are classified as either fusion (pancreas divisum), migration (annular pancreas, ectopic pancreas) or a duplication anomalies (number or morphology variation).
3.1)Fusion anomalies
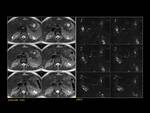
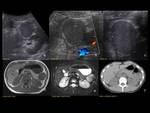
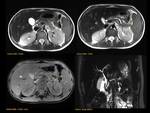
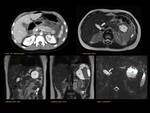
PANCREAS DIVISUM Fig. 3 Fig. 4 Fig. 5
The most common anatomic variant (5%-10%).
Absent or incomplete fusion between the ventral and the dorsal buds of the pancreas. As a result the dorsal duct of Santorini drains the majority of pancreatic secretions into the minor papilla with only the pancreatic head and the uncinate process draining through the duct of Wirsung into the major papilla.
Although may be clinically asymptomatic,
pancreas divisum is associated with acute and recurrent pancreatitis (functional stenosis at the minor papilla).
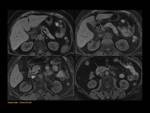
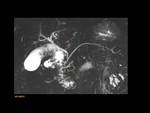
MRCP allows the visualization of the two separate ducts of the pancreas,
shows the crossing of the common bile duct over the main duct of Santorini to join the duct of Wirsung at the major papilla (ampulla de vater).
Variants: Fig. 6
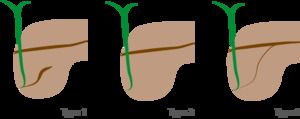
Fig. 6: Type-I (classic): absent fusion between de ventral duct of Wirsung and the dorsal duct of Santorini
Type-II (Absent ventral duct): minor papilla drains all the pancreas
Type -III (functional): rudimentary connection between dorsal and ventral ducts
3.2)Migration anomalies
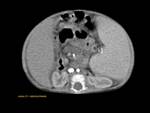
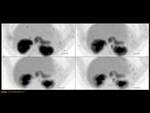
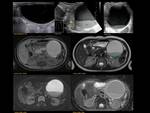
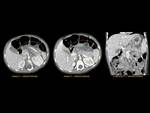
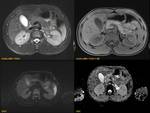
ANNULAR PANCREAS Fig. 7
It is the second most common anatomic variant.
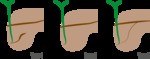
A ring of pancreatic tissue encircles either completely (25%) or incompletely (75%) the second portion of the duodenum.
Classification Fig. 10
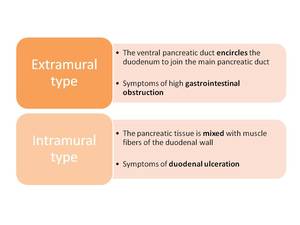
Fig. 10: Types of annular pancreas
It is associated with other congenital anomalies in up to 70% of children,
such as Down Syndrome,
Hirschprung disease,
polysplenia or tracheoesophageal fistula.
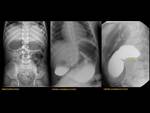
Paediatric patients may present with high-grade duodenal obstruction/subocclusion but most patients may remain asymptomatic for life. Abdominal plain film may show the classic “double bubble” sign seen in duodenal atresia but with distal intestinal air. Fig. 8 Fig. 9
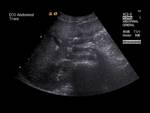
Ultrasound can show abnormal,
concentric course of the pancreatic duct around a dilated fluid-filled duodenum.
MRCP is the imaging technique of choice,
depicts a complete or partial ring of pancreatic tissue around the duodenum and reveal the encircling pancreatic ducts.
Partial annular pancreas is characterized by incomplete encasement of the duodenum "crocodile jaw sign".
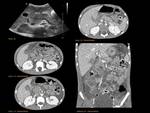
ECTOPIC PANCREAS Fig. 11
Presence of pancreatic tissue outside the normal pancreas location without any anatomic or vascular continuity with the proper gland.
The most common locations: gastric antrum,
proximal duodenum,
jejunum,
Meckel diverticulum or ileum.
Embryological origin theories:
- Residual cells of the primitive ventral or dorsal bud within the bowel,
which displace with the longitudinal growth of the intestinal wall.
- Differentiation of totipotent endodermal cells of intestinal tract into pancreatic tissue
It is usually located in the submucosa (73%) > muscularis mucosa > serosal surface.
Patients are usually asymptomatic,
but if the pancreatic tissue is functional it can develop the same inflammatory or malignant transformation that the normal pancreas.
Obstruction,
intussusception,
ulceration or bleeding may take place when the location is the small bowel.
Imaging techniques:
- Barium examination of the gastrointestinal tract: smooth submucosal lesion with central umbilication
- MRCP: Ectopic tissue with similar intensity as the normal pancreas.
Rudimentary pancreatic duct.
3.3)Duplication anomalies
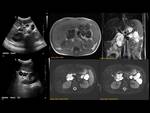
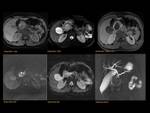
ANOMALOUS PANCREATO-BILIARY JUNCTION (APBJ) Fig. 12
Anomalous union of the pancreatic and bile ducts outside the duodenal wall,
resulting in a long common canal (>15mm).
This anomaly causes pancreaticobiliary reflux,
possibly resulting in an increase of choledocal cyst Fig. 13 ,
pancreatitis and biliary tract malignancy.
ANSA PANCREATICA Fig. 14 Fig. 15
It is a rare congenital variant of the pancreatic ductal system.
There is an obliteration of the dorsal pancreatic duct at the junction with ventral duct and is replaced with an additional reversed S-shaped curved communicating duct between the ventral and dorsal ducts at the pancreatic head,
that drains into the minor papilla.
It can predispose to recurrent pancreatitis due to the suboptimal drainage of pancreatic secretions.
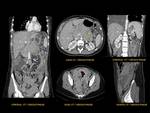
PANCREAS AGENESIS/ HIPOPLASIA Fig. 16
Congenital short pancreas occurs when there is a failure of development of the ventral or dorsal pancreatic buds.
The agenesis of dorsal pancreas is more common and is often associated with polysplenia syndrome.
Patients present with abdominal pain,
pancreatitis due to Sphincter of Oddi dysfunction or diabetes due to the relative lack of islet cells in the pancreatic head.
Differentiation between lipomatosis and pancreatic agenesis is important and is made on the basis of whether the ductal system is present (lipomatosis) or absent (agenesis).
Cross-sectional imaging of dorsal pancreas agenesis show a short truncated pancreas with a globular pancreatic head and uncinate process of variable size (a compensatory enlargement could be seen),
and an absence of the pancreatic neck,
body and tail.
DUPLICATE PANCREATIC LOBE Fig. 17
It is a very rare congenital anomaly defined as an accessory lobe of pancreatic tissue connected with the main pancreatic gland and containing an aberrant duct.
BIFID PANCREATIC TAIL Fig. 14
Fish tale pancreas is a rare congenital branching anomaly of the main pancreatic duct.
There is a division of the pancreatic tail into separate dorsal and ventral buds.
It is usually symptomatic,
incidentally detected.
MRCP shows a duplication of the major duct in the body of the pancreas and the presence of two pancreatic tails.
INTRAPANCREATIC ACCESSORY SPLEEN Fig. 18
Congenital foci of normal splenic tissue within the dorsal pancreas (pancreatic tail).
Intra-pancreatic accessory spleen can be misdiagnosed as a pancreatic neoplasm and subjected to unnecessary management.
At CT and MR,
shows similar imaging and enhancing characteristics to the proper spleen.
CONGENITAL HYPERINSULINISM Fig. 19
4)Systemic Disease with Pancreatic involvement
4.1)Cystic Fibrosis
CF is the most common genetic disease affecting European population and the most-often cause of exocrine pancreatic insufficiency in children.
It is caused by a defective function of the CFTR protein,
affecting the exocrine function of the lungs,
liver,
pancreas and small-bowel.
Pancreatic manifestations
- Exocrine and endocrine insufficiency: duct obstruction by protein-rich exocrine secretions,
which results in acinar ectasia and glandular atrophy
- Pancreatitis (acute and chronic)
- Fatty replacement: Fig. 20 increased echogenecity on US
- Fibrosis of the gland: hypoechogenecity on US
- Diffuse glandular cystosis: Fig. 21 complete replacement of the gland by multiple macroscopic cysts
4.2)Shwachman-Diamond Syndrome
It is a rare AR disease,
but the second most common cause of exocrine pancreatic insufficiency in children.
It may be associated with bone marrow dysfunction and skeletal anomalies.
Diffuse lipomatosis of the pancreas is commonly seen,
this results in complete fatty replacement and glandular enlargement,
but the islet cells of Langerhans and ductal architecture remain preserved.
4.3)Beckwith-Wiedemann Syndrome
Classic triad of omphalocele,
macroglosia and hemihypertrophy.
Variable degrees of organomegaly can be seen and pancreas may be involved.
Half of the patients have islet cell hyperplasia resulting in hyperinsulinemia and subsequent hypoglycemia.
Greater risk for development of abdominal visceral malignancies,
such as pancreatoblastoma.
5)Pancreatic cysts
5.1)True single congenital cysts
Single congenital cysts are extremely rare,
and have female predominance.
May present as an asymptomatic palpable epigastric mass or may be associated with symptoms related to compression of adjacent structures (pain,
jaundice,
vomiting).
These cysts are most commonly located in the tail and body of the pancreas,
and are typically unilocular thin-walled cysts,
ranging in size from microscopic to multiple centimetres.
5.2)Multiple congenital cyst
VON HIPPLE LINDAU DISEASE Fig. 22
VHL is an autosomal-dominant disorder,
characterized by retinal angiomas and central nervous system hemangioblastomas.
Pancreatic lesions include multiple cysts,
serous cystadenoma and islet cell tumours. Pancreatic cysts are common (up to 30% of patients),
being the first manifestation of disease,
the involvement can range from a single to multiple cysts.Cysts arte typically simple but may have peripheral calcification.
AUTOSOMAL DOMINANT POLYCYSTIC KIDNEY DISEASE
It is a hereditary disorder with 100% penetrance but variable expressivity.
Renal cysts are the major feature,
may be found in the liver,
spleen,
adrenal glands and pancreas(in 10% of the patients).
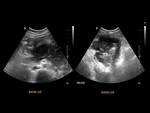
5.3)Gastrointestinal duplication cysts Fig. 23
Abnormalities of the foregut developing,
are spherical or tubular lesions lined by gastric or enteric epithelium and surrounded by a smooth muscle layer.
Most of these cysts contain gastric mucosa or pancreatic tissue,
they can manifest with internal haemorrhage or rupture with peritonitis.
Juxta-pancreatic duplication cysts typically originate from stomach or duodenum,
rarely may be found sequestered within the pancreas parenchyma.
A typical US finding is the double wall sign Fig. 24 (inner hyperechoic mucosa and outer hypoechoic muscular wall),
peristaltic activity can confirm the diagnosis.
6)Acquired Conditions
6.1)Acute pancreatitis
It has a wide spectrum of manifestations in children and adolescents,
and is a potentially life-threatening condition.
Patients typically present with upper abdominal pain,
nausea and vomiting.
The most common cause is abdominal trauma.
Other causes include,
biliary obstruction,
embryologic heaptobiliary system anomalies,
infection,
multisystem disease (vasculitis,
lupus,
sepsis,
shock),
hemolytic uremic syndrome,
medications (L-asperginasa,
steroids),
metabolic-toxic causes.
Two subtypes
- Interstitial oedematous pancreatitis
- Necrotizing pancreatitis
Radiological findings:
- US:Enlarged gland with abnormal echogenicity.
Dilatation of the pancreatic duct.
Peripancreatic fluid.
- CT:Enlarged gland with ill-defined margins,
with heterogeneous enhancement.
Peripancreatic fluid and inflammation of the peripancreatic fat planes.
6.2) Complications of acute pancreatitis
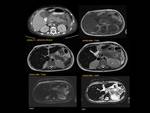
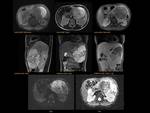
PANCREATIC FLUID COLLECTION Fig. 25
Are defined by presence or absence of necrosis:Fig. 26

Fig. 26: Pancreatic fluid collections are defined by presence or absence of necrosis
LIQUEFACTIVE NECROSIS
It is a severe form of disease that is usually associated with increased morbidity and mortality.
ABSCESS
Presence of infection without significant necrosis
VASCULAR COMPLICATIONS
- Haemorrhage
- Pseudoaneurysm splenic artery
- Splenic vein thrombosis
- Portal vein thrombosis
6.3)Chronic pancreatitis
An unusual condition,associated:
- Anatomic anomalies associated with ductal obstruction.
- Cystic fibrosis
- Shwachman-Diamond syndrome.
- Hereditary chronic pancreatitis: Fig. 27 Fig. 28 an autosomal dominant disease diagnosed in childhood or adolescence,
with a slow progression
- Autoimmune pancreatitis: Fig. 29 Inflammatory lymphoplasmacytic infiltrate.
The typical appearance is a sausage-shaped enlargement of the gland.
Imaging features:heterogeneous or atrophied pancreatic parenchyma,
irregular dilatation of the pancreatic or common bile duct and coarse calcifications.
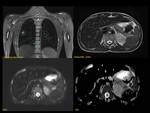
6.4)Pancreatic trauma
It is the 4th most common solid organ injury in the paediatric population.
Pancreatic trauma diagnosis is often delayed because of atypical symptoms.
However,
trauma is one of the most common causes of acute pancreatitis in children.
The mechanism of injury is attributed to the compression of the pancreas against the vertebral column.
Young children are prone to develop more pancreatic injuries than adults due to the flatter diaphragm,
thinner abdominal wall,
and higher costal margins.
Imaging findings:70% injuries in the pancreatic body with 10% occurring each in the head,
neck and tail.
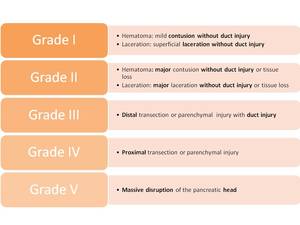
Fig. 30: Pancreatic injury scale (American Association for the Surgery of Trauma). Advance one grade for multiple injuries up to grade III. Proximal injury: to the right of the superior mesenteric vein.
-CT:Enlargement of the gland,
heterogeneous parenchymal enhancement,
peripancreatic fluid collections,
peripancreatic fat strading or fluid.
- Contusion:Fig. 31focal area of decreased attenuation
- Laceration:partial thickness tear
- Transection:full thickness tear Fig. 32
- Comminution(fracture):shattered pancreas
- Hematoma
- Pseudocyst Fig. 33
-MRCP:Better option to exclude pancreatic duct lesions
6.5)Pancreatic tumours
Primary tumours are rare.
EXOCRINE TUMOURS
-Pancreatoblastoma
<1% of all epithelial tumours of the pancreas (boy predilection).
The typical presentation is a large,
palpable abdominal mass,
although nonspecific symptoms may be present.
Alpha-fetoprotein are elevated in >55% of patients,
and the tumor may also secrete adrenocorticotropic hormone.
It is associated with Beckwith-Wiedemann syndrome.
Can be locally invasive but metastatic disease is rare.
Imaging findings: solitary,
large,
well-defined,
multilobulated mass in the pancreas.
- US:heterogeneous lesion with focal hypoechoic areas
- CT:heterogeneous mass with patchy areas of hypoattenuation,
mild-contrast enhancement and calcifications.
- MRI:high intensity on T2WI and low-to-intermediate intensity on T1WI.
-Adenocarcinoma
It can arise from pancreatic ducts or acini.
Types Fig. 34

Fig. 34: Types of adenocarcinoma
ENDOCRINE TUMOURS
-Nonfunctioning
-Functioning:20% of malignant pancreatic tumours.
May be associated with specific syndrome complexes such as multiple endocrine neoplasia type I (MEN1) Fig. 35 and may also be seen in Von Hippel-Lindau disease.
- Insulinoma:hyperinsulinemia.
>90% benign
- Gastrinoma:benign 50% Insulinoma and gastrinoma are typically small and very difficult to detect.
They are hypervascular
- Vasoactive intestinal peptide-producing tumour
- Glucagonoma
CYSTIC TUMOURS
-Solid-cystic papillary tumour Fig. 36 Fig. 37
Accounts for <3%.
It has female predilection,
and approximately 20% occur in children.
Imaging findings: 50% occur in the head
- US and CT:large,
well-defined lesion with variable solid,
cystic and necrotic components.
- MRI:central T1 hyperintense regions due to hemorrhagic necrosis or debris.
low signal rim corresponding to fibrous capsule or residual compressed pancreas.
Metastatic disease to the pancreas is uncommon,
but can be seen with neuroblastoma,
lymphoma (particularly Burkitt) Fig. 38 Fig. 39 ,
some sarcomas Fig. 40 Fig. 41 .